Melting and Boiling Points of Group 4 elements 5000 |MPt C) |BPt C) 4000 3000 2000 1000 c' Si 'Ge Sn Pb Figure 5: Diagram showing the melting and boiling points for the elements in Group IV of the Periodic Table. Figure taken from www.bing.com; accessed 19 May 2021. What the general trend observed in Figure 5? Explain the variation in the melting and boiling points of the element in Group IV of the Periodic Table, shown in Figure 5, in terms of structure and bonding.
Melting and Boiling Points of Group 4 elements 5000 |MPt C) |BPt C) 4000 3000 2000 1000 c' Si 'Ge Sn Pb Figure 5: Diagram showing the melting and boiling points for the elements in Group IV of the Periodic Table. Figure taken from www.bing.com; accessed 19 May 2021. What the general trend observed in Figure 5? Explain the variation in the melting and boiling points of the element in Group IV of the Periodic Table, shown in Figure 5, in terms of structure and bonding.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 32QAP: Follow the step-wise process outlined in Problem 31 to calculate the amount of heat involved in...
Related questions
Question
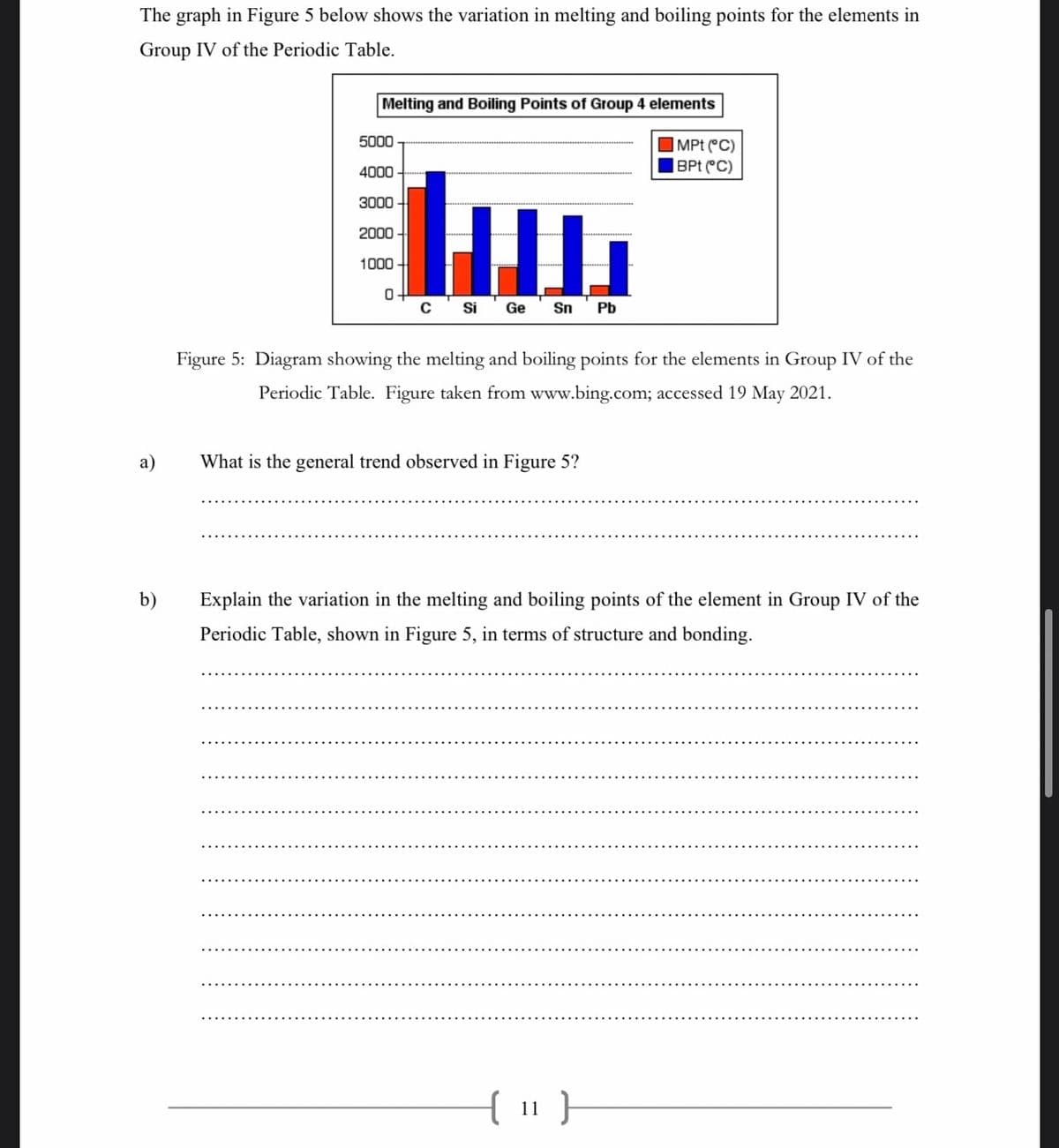
Transcribed Image Text:The graph in Figure 5 below shows the variation in melting and boiling points for the elements in
Group IV of the Periodic Table.
Melting and Boiling Points of Group 4 elements
5000
|MPt (°C)
|BPt C)
4000
3000
2000
1000
0+
Si
Ge
Sn
Pb
Figure 5: Diagram showing the melting and boiling points for the elements in Group IV of the
Periodic Table. Figure taken from www.bing.com; accessed 19 May 2021.
a)
What is the general trend observed in Figure 5?
b)
Explain the variation in the melting and boiling points of the element in Group IV of the
Periodic Table, shown in Figure 5, in terms of structure and bonding.
{ 11 }
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning