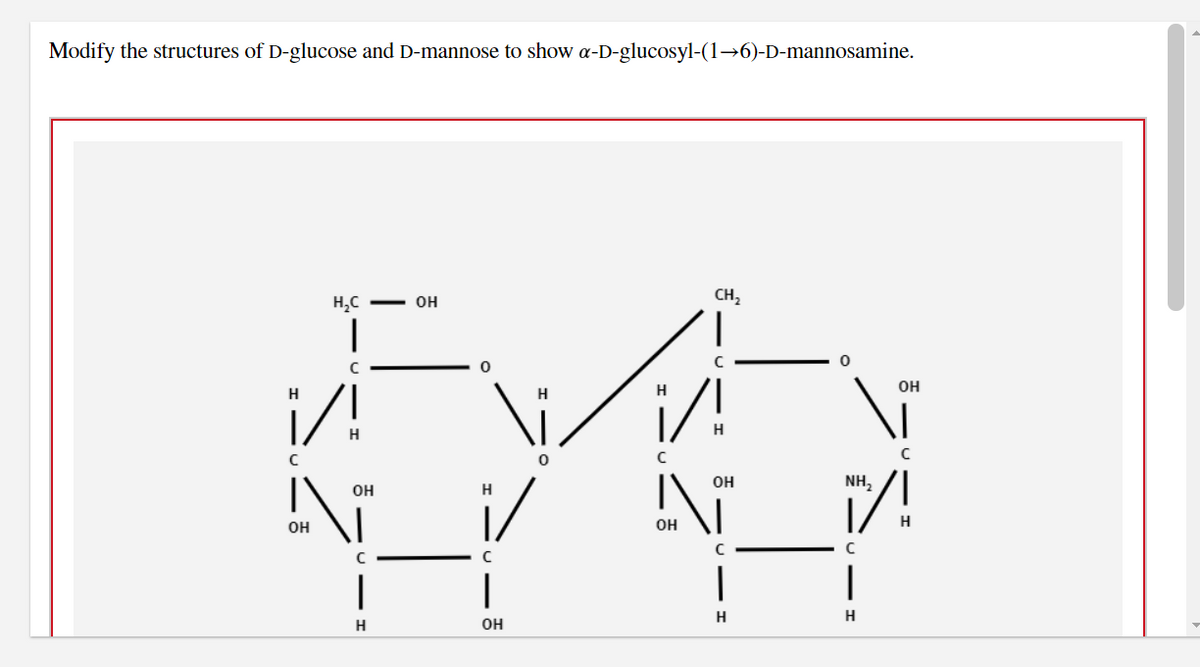
Modify the structures of D-glucose and D-mannose to show a-D-glucosyl-(1→6)-D-mannosamine. H с H C I с H ОН N OH C I H OH 0 H i/ | ОН V с CH₂ ОН H ОН \i C I H NH₂ с I Н ОН с Н
Q: we aimed to separate lysozyme from the Hen egg white proteins. Which of the following is the correct…
A: The correct observation/result from the lab is: Load contains positive, negative, and neutral…
Q: Are the Embden-Meyerhoff-Parnas pathway, transition reaction and the Krebs cycle connected?? If so,…
A: Cellular respiration is the process how biochemical energy is generated from food. It involves the…
Q: 15. A fatty acid which is not synthesized in the body and has to be supplied in the diet is (A)…
A: Regarding a fatty acid that is not synthesized in the body and must be obtained from the diet. The…
Q: Looking at the structure of alpha linolenic acid and knowing the electron carriers produced in each…
A: Alpha-linolenic acid is an omega-3, essential fatty acid. Looking at its structure, it is also…
Q: Which of the following is a heteropolysaccharide?
A: Polysaccharides are complex carbohydrates made up of long chains of monosaccharides. They can be…
Q: 2. A DRUG WHICH PREVENTS URIC ACID SYNTHESIS BY INHIBITING THE ENZYME XANTHINE OXIDASE IS (A)…
A: The answer to the question of which drug prevents uric acid synthesis by inhibiting the enzyme…
Q: Based on your knowledge of metabolism, describe why eating a fat free but high calorie diet can lead…
A: Whenever we intake food, the left over macronutrients in our food (like sugars, proteins, etc) which…
Q: Explain about Renin-Angiotensin system and Hyponatremia
A: The Renin-Angiotensin System (RAS) is a physiological pathway that helps regulate blood pressure,…
Q: How does extracellular pH, NADH, and ATP supply can affect catabolic processes in heterotrophs?
A: Catabolic reactions are metabolic pathways that break down big molecules into smaller ones while…
Q: How is this equation derived? from pre-existing michaelis menten kinetics assumptions?
A: Michaelis Menten kinetics shows the relation between reaction velocity and substrate concentration.…
Q: his is the most abundant carbohydrate found in plants Select one: a. starch b. sucrose c. Rubisco d.…
A: Plant are made up of complex cells consisting of a variety of biomolecules, some of which are unique…
Q: 9. Classify acid-base disorders and explain them along with compensatory mechanisms.
A: The pH of the blood is an essential factor for the proper functioning of the human body. Any…
Q: Which of the following statements is falseconcerning acetyl CoA carboxylase (ACC)? ACC is more…
A: Acetyl CoA carboxylase (ACC) is an enzyme involved in the synthesis of fatty acids. It catalyzes the…
Q: Consider the amino acid valine. Note: Reference the Part: 0/4 Part 1 of 4 Naturally-occurring amino…
A: Valine is one of the 20 amino acids that are used to build proteins in the body, and it has a…
Q: What changes in ATP production can be observed when the pH changes in the intramembranous matrix…
A: Oxidation of glucose via the glycolytic pathway and citric acid cycle generates electrons that…
Q: Which of the following is not a lipid link that anchors membrane-associated proteins to the bilayer?…
A: Membrane-associated proteins can be anchored to the cell membrane by a variety of lipid anchors.…
Q: Glucose-6-phosphate dehydrogenase catalyzes the first step of the pentose phosphate pathway. This…
A: Km of an enzyme is the measure which gives an idea of a substrate's affinity towards its enzyme. It…
Q: 21. The aldose sugar is (A) Glycerose (B) Ribulose (C) Erythrulose (D) Dihydoxyacetone
A: Here provided explanations for each of the answer options in regards to which one is an aldose…
Q: Block diagrams representing the general structures of two types of lipids are drawn. Which terms…
A: The lipid given to us is composed of 2 types of molecules; the one GREEN bar represents a type of…
Q: 11. How many amino acid residues are there in ubiquitin? a) 70 b) 72 c) 74 d) 76
A: Ubiquitin is a small but highly versatile protein found in all eukaryotic cells. Its primary role is…
Q: phosphatidylethanolamine sphingomyelin galactosylcerebroside ganglioside cholesterol Hydrophobic…
A: Amphipathic lipids are those that have a water loving (hydrophilic, polar) end and a water fearing…
Q: Protein from a muscle is being used as a source of energy. Follow the catabolism of a single…
A: The myoglobin first undergoes extensive proteolysis to produce its constituent amino acids , one of…
Q: 3. The structure of phenylalanine is shown to the right. (a) Will phenylalanine test positive or…
A: Phenylalanine is an aromatic amino acid. It has a phenyl group for side chain. Different…
Q: Some reaction components are shown on the left. Match them to the reactions catalyzed by glutamine…
A: Glutamine is a polar, uncharged amino acid. Glutamate is a negatively charged, acidic amino acid.…
Q: A) Please explain the differences between these isozymes as shown by the Michaelis- Menten plot. B)…
A: Isoenzymes : The enzymes that are characterised by the same function but possess different…
Q: 12. Explain about Green House effect.
A: In this answer, I will explain the greenhouse effect, a natural process that occurs when certain…
Q: Structure of direct bilirubin, its value
A: Haemoglobin is a heme prosthetic group-bearing protein that helps transport oxygen from the lungs to…
Q: Explain about Deamination and transdeamination.
A: Amino acids are fundamental building blocks of proteins and play crucial roles in numerous…
Q: 1. Provide the best coenzyme(s) for each step shown below. Give a 1–2 sentence rationale for your…
A: Enzymes are biological catalysts that catalyse biochemical reactions. Most enzymes are made up of…
Q: All the following are sulphur containing amino acids found in proteins except (A) Cysteine (B)…
A: The correct answer is (D) Threonine, which is not a sulphur-containing amino acid found in proteins.…
Q: 1. Since the pK values for aspartic acid are 2.0, 3.9 and 10.0, it follows that the isoelectric (pH)…
A: Amino acids are the main units of protein. Each amino acid has a central carbon atom called Cα. To…
Q: b) The hydrolysis of ATP is directly coupled to H* transport and the stoichiometry of the transport…
A: To determine whether the transport of H+ into the stomach is thermodynamically favorable at 37 °C,…
Q: An experiment was performed to determine the effects of an inhibitor on the breakdown of glycogen by…
A: In the enzyme assay conducted here, we are trying to figure out how the kinetics of the reaction is…
Q: Which type of supercoiling takes the form of extended right-handed coils? a) Positive supercoiling…
A: Supercoiling refers to the twisting and coiling of DNA strands upon themselves, and it is a…
Q: 6. Pathochemistry of jaundice: 6.1. prehepatic (hemolytic) jaundice; 6.2. hepatic (parenchymal)…
A: "Since you have posted a question with multiple sub-parts, we will provide the solution only to the…
Q: Which of the following statements about ion channels is incorrect? Channel proteins are used…
A: Channels are membrane proteins that allow biomolecules such as glucose or water or ions to pass…
Q: Select all the correct statements. Note there might be more than 1 correct statement.…
A: Glycolysis & gluconeogenesis are opposite events. The term genesis means synthesizing while…
Q: 9. Sulphur containing amino acid is (A) Methionine (B) Leucine (C) Valine (D) Asparagine
A: The above answer explains why Methionine is the correct answer to the question "Sulphur containing…
Q: Describe thyroid function tests along with its clinical interpretation.
A: The thyroid gland is a small, butterfly-shaped gland located in the neck that produces hormones that…
Q: The redox carriers are grouped into respiratory chain complex (A) In the inner mitochondrial…
A: The answer explains the location of redox carriers involved in oxidative phosphorylation in…
Q: Draw the skeletal structure of a triacylglycerol that contains three molecules of caproic acid.
A: Tri acyl glycerol : The lipid molecules which consist of glycerol with three fatty acids attached…
Q: Looking at the structure of alpha linolenic acid, how many rounds of beta oxidation will occur to…
A: Introduction: In biochemistry and metabolism, beta-oxidation is the catabolic process by which fatty…
Q: The brain is fueled almost exclusively by glucose. Using this fact, would it be a good idea to…
A: Glucose-1-phosphatase belongs to the family of hydrolases. This enzyme removes a phosphate group…
Q: Provide for each amino acid the names that will uniquely identify all ionisable groups at pH 11.0…
A: Amino acids are organic compounds that are the building blocks of proteins. They contain an amino…
Q: Please draw all of the structures of the intermediates and names of all reactants, intermediates and…
A: Glycolysis is the metabolic pathway that stepwise oxidises glucose to pyruvate to produce ATP and…
Q: In the DNA extraction protocol, what is the purpose of squashing the fruit? In the DNA extraction…
A: DNA extraction is necessary because DNA is found inside cells, tightly packed with other molecules…
Q: The importance of phospholipids as constituent of cell membrane is because they possess (A) Fatty…
A: The question asks about the importance of phospholipids as a constituent of cell membrane and which…
Q: Which of the following is a mechanism of catalysis by co-enzymes? 1. They form intermediate covalent…
A: Co-enzymes are small, non-protein organic molecules that are required by certain enzymes to carry…
Q: Which of the following catalyzes reactions that incorporate nitrogen derived from glutamine? a)…
A: The enzyme that catalyzes reactions that incorporate nitrogen derived from glutamine is b) Glutamine…
Q: A.Define and classify jaundice .Describe laboratory tests for jaundicealong with its clinical…
A: Jaundice is a common condition that is characterized by the yellowish discoloration of the skin and…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

- Name the α-ketoacid that is formed by the transamination of each of the following amino acids: (a) Alanine (d) Leucine (b) Aspartate (e) Phenylalanine (c) Glutamate (f) TyrosineWhy is B-D-Glucopyranose in a chair conformation form is the most stable form of glucose present in our body?Which of an alpha-D-2,3-di-O-methylglucopyranose or alpha-D-2,3, 6-tri-O-methylglucopyranose represents a glucose unit in glycogen which was originally carrying an (alpha 1-->6) glycosidic bond?
- Indicate which of the following amino acids are ketogenic and which are glucogenic: a. tyrosine b. lysine c. glycine d. alanine e. valine f. threonineWhy is it that only 2ATP molecules are required to store one glucose molecule as glycogen?What is the structural difference between glucose and (a) β-D-glucuronate,(b) β-D-glucosamine, (c) N-acetyl-β-D-glucosamine?
- Draw the α-l-glucopyranose sugars using Haworth projections:What does this important observation imply about the relation between the amino acid sequence of insulin and its three-dimensional structure?Why is it advantageous that breakdown of glycogen gives rise to glucose-6-phosphate rather than to glucose?
- What is a glucogenic amino acid? Give three examples.Can an amino acid be both glucogenic and ketogenic? Explain why or why not.Will an amino acid be glucogenic or ketogenic if it is catabolized to the following molecules?(a) Phosphoenolpyruvate(b) -Ketoglutarate (c) Succinyl-CoA(d) Acetyl-CoA(e) Oxaloacetate(f) Acetoacetate

