1. Since the pK values for aspartic acid are 2.0, 3.9 and 10.0, it follows that the isoelectric (pH) is (A) 3.0 (B) 3.9 (C) 5.9 (D) 6.0
Q: During pregnancy a special fetal hemoglobin was made with the following characteristics: It…
A: Hemoglobin is a blood protein which is resonsible for transport of oxygen and other gases in the…
Q: 2. Which of the following temperature best suits the growth and multiplication of transformed cells?…
A: Transformed cells are cells that have been genetically altered to acquire new characteristics or…
Q: Asp residue (both of which are essential for catalysis) with pK, values of 5.9 and 4.5,…
A: In simple terms, pH indicates whether a substance is acid or base. pKa determine how strong is an…
Q: Explain the discrepancy between the whole blood glucose concentration and the plasma glucose…
A: For the determination of Glucose, whole blood, serum, or plasma can be used.
Q: Complex 4 is also known as a) Cytochrome oxidase b) NADH dehydrogenase c) Succinate dehydrogenase d)…
A: Cytochrome oxidase, also known as Complex 4, is an enzyme complex found in the electron transport…
Q: 31. Explain Metabolic acidosis and Chloride shift
A: The body's acid-base balance is crucial for proper physiological functioning, and any imbalance can…
Q: A(n) _________ reaction converts glycerol to glycerol 3-phosphate. This reaction requires…
A: The conversion of glycerol to glycerol 3-phosphate is a key step in the metabolism of carbohydrates…
Q: Question 5 of 10 Among the given statements, which one(s) is/are correct about alpha keratin? 1. It…
A: Alpha-keratin is a protein found in vertebrates. It is found in nails, hairs, claws and epidermis of…
Q: Decide whether each molecule in the table below could be found embedded in the outer surface of a…
A: Cell membranes are mostly composed of phospholipids. An important characteristic of phospholipids is…
Q: Would you expect the glucose levels of the lactose solution to change appreciably after the addition…
A: Lactose is a disaccharide. It is made up of one monomer of glucose and one monomer of galactose. Its…
Q: Q1. What is the theoretical yield (product mass) of Aspirin if you started 2.50 g of Salicylic Acid?…
A: To calculate the theoretical yield (product mass) of Aspirin, we need to first determine the molar…
Q: Which of the following is required for crystallization and storage of the hormone insulin? (A) Mn++…
A: The question asks about the mineral required for the crystallization and storage of the hormone…
Q: In the protocol for extracting DNA, what is the purpose of the clear shampoo in the DNA extraction…
A: DNA is the biomolecule that carries genetic information from one generation to another. Shampoo is a…
Q: Glycogen phosphorylase in the liver... A. is the same enzyme that acts in the muscle. B. is mostly…
A: Glycogen phosphorylase, glycogen synthase, and protein phosphatase 1 (PP1) are all enzymes involved…
Q: Name and explain the sulphur containing amino acids. List two disorders of their metabolism.
A: Amino acids are biomolecules that have an amino group, a carboxyl group and another group called the…
Q: 4. Enzymes with a kcat / Km ratio of about 10³ M-¹s-1 are considered to show optimal catalytic…
A: The Kcat/Km ratio is a measure of the catalytic efficiency of an enzyme. Kcat (also known as…
Q: (b) The diagram on the right illustrates the change in the p50 (partial pressure of O₂ required to…
A: Hemoglobin is a protein found in red blood cells that is responsible for transporting oxygen from…
Q: Formula for % solubility of proteins: % solubility = [(C x V x F) / (1000 x 35%)] x 100% C =…
A: Percent solubility is a measure of the substance's ability to dissolve in solvent. It is the maximum…
Q: What are the metabolic pathways involved in glucose metabolism, and how are they regulated?
A: An integral mechanism in the human body is glucose breakdown and synthesis. The necessary substrates…
Q: The following compounds exhibit little to no adrenergic receptor function. Consider what components…
A: adrenergic receptors are G-protein coupled receptors that mediate the signal from agonist binding…
Q: Which of the following inactivates the 605 subunit of eukaryotic ribosomes? a) Ricin b)…
A: Protein synthesis is a fundamental process for all living organisms, and its inhibition can have…
Q: Lipids List the different groups of lipids presented in the course notes. Saturated: Unsaturated:…
A: Lipids are a diverse group of organic compounds that are insoluble in water but soluble in nonpolar…
Q: Explain about Cytochrome P450 in detoxification.
A: Cytochrome P450 (CYP) is a group of enzymes that plays a crucial role in the metabolism of…
Q: Which of the following most accurately describes the impact acetyl-CoA has in the cytosol of the…
A: Glycolysis is a metabolic pathway that breaks down one molecule of glucose into two pyruvate…
Q: 15. In neutral fats, the unsaponificable matter includes (A) Hydrocarbons (B) Triacylglycerol (C)…
A: The question asked about the unsaponifiable matter present in neutral fats and provided four…
Q: what are the formal chemical names for sucrose,lactose and maltose?
A: Sucrose, lactose, and maltose are all disaccharides, which are composed of two monosaccharide units…
Q: How many atoms are held into a geometric plane by a peptide bond? 6 7 4 5…
A: A peptide bond is a covalent bond formed between the carboxyl group (-COOH) of one amino acid and…
Q: 13. Explain about Air/H2O pollution.
A: Air and water pollution are major environmental issues that have significant impacts on human health…
Q: Question questions. Answer the following A) Compare and contrast triglycerides in fat and oils? Draw…
A: Triglycerides are esters derived from glycerol and three fatty acids. Fatty acids (FA) are long…
Q: Driven by the hydrophobic effect, certain______ molecules form bilayers. zwitterionic…
A: The bilayer nature of the membrane refers to the double layer of phospholipid molecules that make up…
Q: Why is necessary to completely grind the beef in an LDH purification experiment? How do you…
A: Since you have posted multiple questions, we will provide the solutiononly to the first question as…
Q: 1. What are the effects of pH and temperature to catalase? What is the optimum pH and optimum…
A: Hi! Thank you for the question. We have answered first four questions as it belonged to enzyme and…
Q: How do you identify the N vs C terminals? For the first part of the question, do you then list the…
A: Amino acids are the monomers of a polypeptide or protein. Amino acids contain an amino (-NH2) group…
Q: Which of the following condenses acyl and malonyl groups? a) Acetyl co-A ACP transacetylase b)…
A: β-ketoacyl ACP synthase is an enzyme that plays a critical role in fatty acid biosynthesis. It…
Q: 1. The number of milligrams of KOH required to neutralize the free and combined fatty acid in one…
A: The saponification number is the number of milligrams of potassium hydroxide (KOH) required to…
Q: Two amino acids are shown below. Name them in the blanks, and then draw the structure of the…
A: Amino acids are biomolecules that have an amino group and a carboxyl group linked to the same carbon…
Q: Which of the following co-enzymes are associated with the pyruvate dehydrogenase (PDH) complex?…
A: The pyruvate dehydrogenase (PDH) complex is mitochondrial multienzyme complex This complex catalyzes…
Q: In SDS page, the ________ proteins will move through the gel slowest and last, and in size exclusion…
A: SDS PAGE and Size Exclusion Chromatography are two biochemical analytical techniques used for the…
Q: What are the three states of raft assemblies? Describe each.
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Anti Lice Oil from Guyabano (Annona Muricata L.) Seed Extract
A: Infestations of lice are a widespread issue, and interest in all-natural cures is rising. The…
Q: 4. Indicate the polar and nonpolar portions of the following classes of lipids. a. triacylglycerol…
A: Lipids are organic molecules that are insoluble in water but are soluble in non-polar solvents like…
Q: Estimate the pl (isoelectric point) of a protein with the amino acid composition below. . Arg: 3…
A: The approximate pKa values for the amino acid residues are as follows: Arginine (Arg): pKa =…
Q: ATP Structure H- N NH₂ N Adenine, (Nitrogenous base) N N H -H H H CH, O H Alpha Beta phosphate…
A: ATP (adenosine triphosphate) is a molecule that plays a crucial role in cellular energy transfer.…
Q: compute the yield of ATP when one molecule of hexanoic acid (6:0 fatty acid) is completely oxidized…
A: Aerobic respiration is a metabolic pathway that breaks down organic molecules, such as glucose and…
Q: Which of the following gives rise to y-amino butyrate? a) Pyruvate decarboxylation b) Glutamine…
A: γ-Amino butyrate (GABA) is an amino acid derivative that is synthesized from glutamate through a…
Q: A Kf Kr B Say you have this reaction above. 3 f Keq is 1 10° without the enzyme, then what's Keq…
A: Enzymes are biological catalysts that increase the rate of biochemical enzymes. The enzymes are…
Q: 9. Classify acid-base disorders and explain them along with compensatory mechanisms.
A: The pH of the blood is an essential factor for the proper functioning of the human body. Any…
Q: Which of the following statements about ion channels is incorrect? Channel proteins are used…
A: Channels are membrane proteins that allow biomolecules such as glucose or water or ions to pass…
Q: 1. Where inside the cell does the degradation of long-chained fatty acids occur? a. mitochondria b.…
A: Since you have posted multiple multiple-choice-questions, we will provide the solution only to the…
Q: Which of the following statements is correct (note: there may be more than 1 correct response)?…
A: Cellular respiration is the process how biochemical energy is generated from food. It involves the…
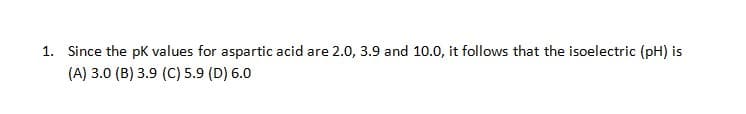
Step by step
Solved in 4 steps with 2 images

- 1. a. Explain why the melting point of palmitic acid (16 carbons, no double bonds) is slightly lower thanthat of stearic acid (18 carbons, no double bonds). Explain why the melting point of oleic acid (18carbons, one double bond) is lower than that of stearic acid b. A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, phosphatidylserine, andphosphatidylethanolamine was applied to a hydrophobic interaction chromatography column. Thecolumn was washed with a high salt buffer, and the lipids were then eluted with decreasing saltconcentrations. In what order would the lipids be eluted from the column? Explain your answer.1. What is the possible identity of the AA? a. Aspartic acid b. Lysine c. Alanine d. Proline 2. What is the isoelectric point of AA? 3. What is the pKa responding to the dissociation of the alpha-carboxylic group?The normal concentration of Ca2+ in blood is 5.0 mEq>L. How many milligrams of Ca2+ are in 1.00 L of blood?
- 4. Why is PHC still relevant today?The concentration of cholesterol (C27H46O) in blood is approximately 5.0 mM. How many grams of cholesterol are in 250 mL of blood?In a TPN bag contains: Kcal of 1000 ml of D50 W, 1000 ml of 7% amino acids, and 650 ml of 20% lipids. A. How many total kcal is provided by this bag of TPN?
- Ty is three years old and as a result of a stomach bug has been vomiting for about 24 hours. His blood pH is 7.48. What does this mean? Tys blood is slightly acidi Tys blood is slightly alkaline. Tys blood is highly acidi Tys blood is within the normal rangeWhich of the following is TRUE, if the protein pH is electrically neutral? Refer to the graph below. a. The denaturation of protein is lower than 100% b. The denaturation of protein is greater than 100% c. The protein is in native form d. The protein is completely denatured3. Diet Big Red soda contains citric acid, as shown in the ingredient list below, Carbonated Water, Citric Acid, Natural and Artificial Flavors, Sucralose, Sodium Benzoate, Caffeine, Malic Acid, Red 40, Acesulfame Potassium, Phosphoric Acid, and Acacia Gum What are two obvious reasons that we did not try to determine the citric acid content in a "flat" sample of Diet Big Red soda?
- 1. What are the differences between unsaturated and saturated fatty acids? 2. What is Biological Value (protein)? 3. What is Chemical Score (protein)? 4. What temperature is your beverage optimal for rehydration?1. In Clinical Chemistry, are enzymatic methods for determining glucose, cholesterol, and uric acid more preferred than non-enzymatic methods?What are the possible effects of taking multivitamin tablets to your overall health? Explain in terms of biochemistry.

