Q: ng from 3-hexene? Br Br a. CH,CH,CHCHCH,CH; b. CH,CH,CH,CH,CH,CH, Cl c. CH,CH,CH,CHCH,CH, с. ОН d. C...
A: Reaction a. Reaction of alkene with Br2 reduces the double bond and forms a dibromo product Reacti...
Q: Give the name of the molecule
A: The given molecule contains a five-carbon atoms chain and a thiol (SH) functional group attached wit...
Q: How many significant figures are in each of these measurements? 71. 93 g, 0.420210 kg
A: The significant figure refers to particular digits in a number. According to the measurement rule, o...
Q: Are all atoms of an element exactly the same? Maybe No HUH? Yes
A: We need to determine if the atoms of same element are same.
Q: Degradation of an airport's aging fuel tanks is suspected, resulting in both rust and water contamin...
A: A question based on properties of liquids that is to be accomplished.
Q: Objectives: a) Define Aufbau principle, Pauli Exclusion Principle, and Hund's Rule. b) List and desc...
A: A question based on structure of atoms that is to be accomplished.
Q: INTRODUCTION LABORATORY SIMULATION 10 8.75 7.5 6.25 5 3.75 2.5 1.25 12 16 18 22 Volume of NaoH Added...
A: Pka can be calculated as
Q: Enter electrons as e¯. The following skeletal oxidation-reduction reaction occurs under basic condit...
A: The basic steps to balance redox reaction in basic medium are- 1. Find oxidation no of each atom and...
Q: 2. Consider the reaction below: Al½O3 (aq) + 6 HC1 (aq) → 2 AlCl3(aq) + 3 H2O(1) Using the method of...
A:
Q: Give the oxidation state of following compounds and tronegativity values in Appe (a) CaO; (b) H;O; (...
A: Since you have asked multiparts questions, we will solve the first three sub parts questions for you...
Q: A student proposed the following mechanism for the gas phase reaction of fluorine with chlorine diox...
A: Step 1.....fast:......2 ClO2 <==> Cl2O4 Step 2.....slow:....Cl2O4 + F2 ==> 2 FClO2
Q: What is the charge of an iron ion with 24 electrons? Is this a cation?
A:
Q: Name of Test Positive result Sodium Metal test Production of effervescence Ester Fruity smell or men...
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for you...
Q: 3. following reactions. (a) Draw the product(s) including its(their) stereochemistry in each of the ...
A: We have to do reactions.
Q: Why are the chiral carbons (S) and (R)? Shouldn't they be both (S)
A: The answer is as follows:
Q: thermometer A sample of polystyrene, which has a specific heat capacity of 1.880 J-g.°C, that contai...
A:
Q: 3. The two most important inorganic buffer systems in the body are the phosphoric acid and carbonic ...
A:
Q: "solid potassium chlorate reacts to form solid potassium chloride and gaseous oxygen."
A: Moleculer formula of Potassium chlorate is KClO3 Moleculer formula of potassium chloride is KCl Mo...
Q: There are two naturally occurring isotopes of chlorine. 35 Cl has a mass of 34.9689 u. 37 Cl has a m...
A: We have to find the percentage abundance of 35Cl and 37Cl.
Q: QUESTI UN 15 . Which of the following approximate pka ranges for the following acids is correct? You...
A:
Q: What reagents are needed to convert (CH3CH2)3CCH=CH2 to each of the following compound? а. b. CI CI ...
A:
Q: e rate of a
A:
Q: The average linebacker in the NFL can run 40.0 yards in 4.76 seconds. What is the average speed of t...
A:
Q: assification are possible. These prefixes are underlined italics and separated from the rest of the ...
A: Answer are as follows:
Q: 4. Calculate the work done (in J) when a gas condenses from 971 mL to 264 mL at a constant pressure ...
A: Given :- initial volume of gas = 971 mL final volume of gas = 264 mL pressure = 4.00 atm To cal...
Q: Calculate the coefficient of compressibility for a mixture containing 40 mol% of carbon dioxide and ...
A:
Q: The rate of a certain reaction is given by the following rate law: rate=k[N,][H,] Use this informati...
A: Chemical kinetics can be defined as the branch of chemistry that deals with rates of chemical reacti...
Q: What is the identity of the orange-red complex ion formed by the reaction of ferrous ions with o-phe...
A:
Q: a. What is the maximum amount of crude RDX the student can purify given this volume of acetone in o...
A: As per the given information- 1 g RDX : 100 mL cold acetone 1 g RDX : 25 mL boiling acetone As the p...
Q: Electrophilic addition of HI
A:
Q: A polluted stream flows at 1 MGD carrying a concentration of 1 mg/L of a conservative pollutant such...
A: Given that, a polluted stream flows at 1 MGD (million gallons per day) carrying a concentration of 1...
Q: Obtain the infrared spectrum of your (dry powder) polymer product. n.b. : Attach a copy of your infr...
A: A question based on IR spectroscopy that is to be accomplished.
Q: 8. The density of nickel is 8.908 g/cm3. If the metallic radius of nickel is 125 pm, what is the str...
A: Concept n= 1 then Simple cubic structure n= 2 then Body centered structure n = 4 then Face centered...
Q: The negative ion mode electrospray ionization (ESI) mass spectrum of an oligonucleotide shows peaks ...
A:
Q: mass
A:
Q: 1.Write the balanced, net ionic equation for the reaction occurring when (a) Cl is added to a soluti...
A:
Q: created x10 Is C,H,Cl being created or destroyed by the chemical O destroyed reaction? neither creat...
A: Given that, at time t = 0 sec the concentration of C4H9Cl is 0.266 M. From the graph, we observed th...
Q: Would you expect 2-Chlorobutanoic acid to have higher or lower pka than 4-Chlorobutanoic acid?
A: Does 2-Chlorobutanoic acid to have higher or lower pka than 4-Chlorobutanoic acid?
Q: Entropy Change Suppose we have 1.50kg of water at a temperature of 0 degree Celsius. We put the wate...
A: Mass of water = 1.50kg or 1500 g Temperature = 0°C or 273.15 K
Q: Briefly discuss the importance and principles of non-contact laser profilometry.
A: The non-contact laser profilometry is based on the principle of optical light interference to scan a...
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are ...
A: whether a precipitate forms when aqueous solutions A and B are mixed.
Q: Complete the table below by deciding whether a precipitate forms when aqueous solutions A and B are ...
A: solution A solution B precipitate form when A and B are mixed empirical formula of precipitate Sod...
Q: 85 11. For the Isotope 38Sr A. What is the atomic number? B. What is the mass number? C. What is the...
A: The given symbol for isotope is 8538Sr A) The atomic number is represented by the subscript. The...
Q: The red arrow (which we use in organic chemistry to indicate the movement of one or two electrons du...
A:
Q: Use standard enthalpy and entropy data value from the Table: to calculate the standard free energy c...
A:
Q: C7H12O4 I need help answering 2 3 4 5 For IHD please show step by step ir analysis: absorption...
A: Given that, the molecular formula of the molecule is C7H12O4. 2. So, the required DU (IHD) is DU = ...
Q: In the equation (red + white à pink), which equation below allows you to determine the equilibrium c...
A:
Q: For each of the following pairs or reactions, which is correctly labeled as having the larger equili...
A:
Q: Heptane: yellow flame
A: This is a multipart question.According to the guidelines, in multiple parts question, we can solve o...
Q: mass
A:
Write the products and balance the following: C3H8 + O2 ---------->
What is the maximum number of water molecules produced 0.25 moles of propane combust with excess oxygen
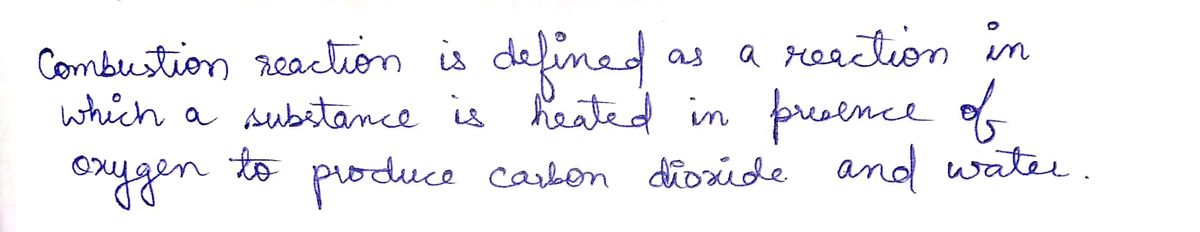
Step by step
Solved in 3 steps with 3 images

- A typical formulation for window glass is 75% SiO₂, 15% Na₂O, and 10.% CaO by mass. What masses of sand (SiO₂),sodium carbonate, and calcium carbonate must be combined toproduce 1.00 kg of glass after carbon dioxide is driven off bythermal decomposition of the carbonates?A commercial 737 jet transporting 143 passengers and 5 crew members from Kansas city (MCI) to Baltimore (BWI) burned 11800 lb. of Jet A fuel. Jet A fuel is kerosene based, consisting primarily of CnH2n+2 hydrocarbons, with n = 6 to 16, so the Carbon:Hydrogen ratio is close to 1:2. During this flight , how much CO2 (in kg) was released into the atmosphere? (assume the combustion of fuel was complete, so all fuel was converted into H2O and CO2).0.004 C8H7NO3, 15mL ethanol, 0.002mol NaBH4 was used. HCl was used to acidify. It was then extracted with ether. Then it was dried and a mass of 0.32g was obtained. What is the balanced equation of this synthesis, and what is the limiting reagent?
- One of the sources of iron is magnetite, Fe3O4 (FeO and Fe2O3 mixed), which reacts with coke (carbon) to produce iron in the liquid state and carbon monoxide. If we assume that coke is pure carbon, how much is supplied to produce 10,000 metric tons of iron?Urea, CH4N2O, is produced by the reaction between ammonia and carbon dioxide, as shown by the followingequation. 2NH3 + CO2 → CH4N2O + H2O a. Calculate the quantities, in kilograms, of NH3 and CO2 that should combine to produce 6.0 kg of urea.NH3 _______________ kgCO2 _______________ kg b. How many kilograms of water will be produced as the side product in this process?H2O _______________ kg42. The primary use of 1,2‐dichloroethane, ClCH2CH2Cl, is to make vinyl chloride, which is then converted into polyvinyl chloride (PVC) for many purposes, including plastic pipes. Balance the following equation, which describes the industrial reaction for producing 1,2‐dichloroethane. C2H4 + HCl + O2 → ClCH2CH2Cl + H2O 50. Gaseous propane and liquid diethyl ether mix to form a solution. Which of thesesubstances is the solute and which is the solvent? 51. Consider a solution of 10% liquid acetone and 90% liquid chloroform. Which of these substances is the solute and which is the solvent?
- Ammonia (NH3) is naturally produced through the decomposition of organic material as wellas being high in wastewater discharged to natural waters. In the environment it is oxidizedto form nitrite (NO2-), and nitrite is then oxidized to form nitrate (NO3-) in a process callednitrification. The balanced equations are given below:2NH3 + 3O2 → 2NO2- + 2H+ + 2H202NO2- + O2 → 2NO3-a) Assuming complete oxidation, what is the theoretical oxygen demand to convert100mg/L of ammonia to nitrate (i.e., how much oxygen is necessary)? Report youranswer as mg/L of O2.Answer: 376 mg/L O2b) This conversion process makes water more acidic. With reference to the equations forpH, explain why this occursThe sugar glucose (C6H12O6) is broken down in a decomposition reaction to ethanol (C2H6O) and bubbles, CO2. Write the balanced chemical equation for the reaction.Balance the equation below such that it has the smallest, whole-number coefficients. CH3CH2CH2CH2OH + O2(g) ® CO2(g) + H2O(g)
- The fat stored in a camel’s hump is a source of both energy andwater. Calculate the mass of H2O produced by the metabolismof 1.0 kg of fat, assuming the fat consists entirely of tristearin(C57H110O6), a typical animal fat, and assuming that during metabolism,tristearin reacts with O2 to form only CO2 and H2O.The Bayer process is the formation of aluminum hydroxide from metallic aluminum and sodium hydroxide heated to high temperatures. Al(s)+ 3NaOH(aq) -->Al(OH)3(s) + 3Na A manufacturing chemist observes that two distinctly different solids are present in the solutions after the reaction has come to completion. The chemist collected the following data: data table From the above information, identify the limiting and excess reagent, the supporting evidence, and suggest a method to increase the amount of product produced.Knowing that the reaction yield: FeS2 + O2 → Fe2O3 + SO2 is 75%, from 360 g of iron disulfide, calculate: a) The amount of iron (III) oxide produced. b) The volume of SO2, measured at 0 ºC and 1 atm, that will be obtained.
