Q: CH3 CH3 -CH2-C-CH2-CH3 HNO3 KMNO4 А B H2SO4 H2SO/Heat CH3
A: Organic reaction mechanisms:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. N2 H2 init...
A: A question based on kinetics of reactions in solution that is to be accomplished.
Q: Question 13 Ultraviolet and visible molecular absorption spectroscopy is used primarily for quantita...
A: In UV visible best wavelength to use is the one with the highest molar absorptivity (λmax), provide...
Q: One atom has 15 protons and 16 neutrons in its nucleus, with 15 electrons traveling around it. Anoth...
A: Isotopes are the elements which have same atomic numbers but they have different mass number Ex. 1H...
Q: A gas mixture is composed of 2.0 moles Hz and 1 mole N2. Calculate (a) the average molar mass of the...
A:
Q: Does the addition of solvent change the molarity of a solution when a solution is diluted? Explai
A: The answer is as follows:
Q: A temperature inversion of the atmosphere was observed based on the data below as reported by probes...
A: Soln
Q: Three labs were contracted to measure the concentration of a 5.50 ppm Ca2+ standard solution. They e...
A: For the problem we have to find out which lab measurements is most accurate result and which one has...
Q: 1. Predict and label the conjugate acid/base pairs of the following acid-base reaction assuming it r...
A: The correct answer to the question is given below
Q: CECH 1. НО. CI
A:
Q: SO3H ? ČH2-CH3
A: The answer is given below
Q: the pH of
A:
Q: CH3CI A AICI, Sn B• HCI OH- C D NaNO2, HCI, 0°C CUCN E HNO3 -F H2SO4
A: The products are as follows:
Q: Indicate what specific electron will have the following quantum number indicated below: 8. 4, 3, +1,...
A:
Q: Nitroethylene, H,C-CHNO, is a sensitive compound that must be prepared with great care. Atempted pur...
A:
Q: Which is NOT true about Auxochromes O They are conjugated dienes O they are functional group attache...
A: A chromophore is a molecule's constituent responsible for a specific UV/visible absorption. The sub...
Q: Determine if the statements are true. The larger the surface area, the higher the dissolution. ...
A: True or false: As per our guideline we have to answer first three questions only:
Q: Concentrated aqueous HCIO4 has a concentration of 14.8 M. Calculate the concentrations of all ions p...
A:
Q: CH2-CH3 1. СО 2. H3O*
A: Given incomplete reaction is : Give the major product of the reaction = ?
Q: 1. Calculate the grams of solute that must be dissolved in 430g of H20 in the preparation of a 14% N...
A:
Q: A solution is prepared by dissolving 27.03 grams of FeCl3* 6H2O, and diluting to give a final volume...
A: Molarity is a concentration term for a solution. The molarity of a given solution is defined as the ...
Q: Lewis structures for CO2: one with C in the middle and one with O in the
A:
Q: The PHIVOLCS recently reported that Taal volcano emitted 15,500 tons of Sulfur Dioxide (SO,). How ma...
A:
Q: 0.508 g of KHP was mixed with 25 mL of H20 to titrate a NaOH solution. It took 32 mL of the KHP solu...
A: Given as 0.508 g of KHP was mixed with 25 mL of H2O KHP solution required to reach the endpoint is 3...
Q: What is the percent ionization of a 0.0289 M aqueous solution of cyanic acid, HCNO (K, = 3.5E-4)? i ...
A: The ionisation of weak acid are very low. The percentage of ionisation can be calculated as follows
Q: Draw the structural formula for lithium divinylcuprate and state whether it qualifies as an organome...
A: Structure of lithium divinylcuprate
Q: How many different 13CNMR signals does the following compounds exhibit?
A: 13-C NMR gives the information about the number of different environment carbon atoms.
Q: What is the Amax of this given structure?
A: We can calculate the maximum wavelength in ultraviolet- visible spectroscopy.
Q: What causes the oscilation of the covalent bond oscillates resulting to varying electromagnetic fi...
A: In a covalent bond, in natural states atoms in covalent bond vibrates at natural frequency which com...
Q: What is the Amax of this given structure?
A:
Q: Paraphrasing .ewriting Tool Car note Pirate Ship BLACKBOARD [Review Topics] [References] Indicate wh...
A: The first compounds are same conformation of a compound viewed from a different perspective.
Q: Which of the following are not correctly paired? a. Na, alkali metal b. Sn, lanthanide c. Br, hal...
A:
Q: The osmotic pressure of a 14.538 mM solution of LiCl is 343.095 mm Hg at 27.84 °C. How many ions per...
A: Given: Osmotic pressure = 343.095 mm Hg Concentration (C) = 14.538 mM Temperature = 27.84°C
Q: What is the general trend in first ionisation energies across Period 3? Explain why aluminium has a ...
A: A question based on periodicity properties that is to be accomplished.
Q: Use Hess's Law and the three reactions, below, to calculate the standard enthalpy of reaction (AH°rx...
A: The reaction for the coal gasification is, 2C(s) + 2H2O(g) ---> CH4(g) + CO2(g) The three re...
Q: Fill up the empty boxes with the correct chemical structures. CH3 Br CO,H
A: Organic reaction mechanisms:
Q: Most molecules/compounds that have a mass similar to water are gaseous at room temperature, what mak...
A: Water is liquid at room temperature.
Q: エ 2. Determine which of the following would effectively deprotonate an acid with a pKa of 30. (hint:...
A: Given-
Q: At 24 °C and 745 mmHg, the density of an unknown gas is determined to be 7.76 g/L. What is the molar...
A:
Q: 4 What is the freezing point of a solution that contains 64.3 g of sucrose in 200 g of water? Selec...
A: mass of the sucrose=64.3g molar mass of sucrose=342.3 g/mole Number of moles of sucrose=64.3/342.3 =...
Q: What causes the oscilation of the covalent bond oscillates resulting to varying electromagnetic fiel...
A: covalent bond between atoms behave as a elastic spring ( on the basis of IR spectra, we can say this...
Q: 10. NHẠI 11. KrBr2 12. KNO3
A: Since you asked multiple questions so,as per Q&A guidelines of portal I solved first three parts...
Q: Consider the initial‑rate data at a certain temperature in the table for the reaction described by ...
A:
Q: SO3H CH3CI AICI, CH2-CH3 3-ethylbenzenesulfonic acid
A: Given example is alkylation reaction in presence of lewis acid. For Alkylation, the ring should be e...
Q: A gas mixture is composed of 2.0 moles N2 and 1 mole H2. Calculate the average molar mass of the gas...
A: Given: 2 moles of N2 1 mole of H2 2 moles of N2 has mass 2*28 = 56 gram 1 mole of H2 has mass 2 gram...
Q: A solution is prepared by dissolving 27.03 grams of FeCl,* 6H;O, and diluting to give a final volume...
A: The molarity (M) of a solution is the ratio between the number of moles (n) of solute to the solutio...
Q: Benzene is one of the Toxic Air Pollutants (TAPS) found in car exhaust fumes When a 50.00 ml. aqueou...
A: It is given that the fractional distillation of 50.00 mL of aqueous solution of 0.05000 M benzene is...
Q: Calculate the mass of excess reagent remaining at the end of the reaction in which 180.0 g of SO2 ar...
A:
Q: . magnesium selenide
A:
Q: 11. gold (III) acetate 12. barium hydroxide 13. hydrochloric acid 14. calcium bicarbonate 15. perchl...
A: Naming is done by looking at the cation and anion
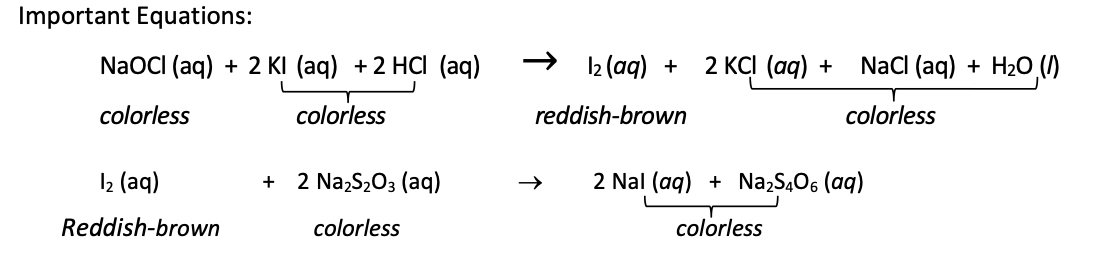
Molarity of Na2S2O3 (stock solution). 0.100 M
Volume of diluted bleach titrated (taken from the volumetric flask). 10ml
Initial Burette Reading. 14.80
Final Burette Reading 25.60
Volume of Na2S2O3 used in the titration (mL). 10.80
Moles of Na2S2O3 used in the titration. 1.71
Moles of S2O3–2 used. 1.66
Moles of I2 initially present in titration mixture 0.83
Moles of NaOCl in the diluted bleach titrated. 0.415
Moles of OCl– in the diluted bleach titrated. ?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- The concentration of gadolinium was measured in the discharge from a wastewater treatment plant near a large medical facility. Eight measurements were obtained: 1.411 ppm, 1.411 ppm, 1.401 ppm, 1.409 ppm, 1.979 ppm, 1.408 ppm, 1.419 ppm, 1.402 ppm. Use the Grubbs test to determine if one of these values is an outlier. What is the value of ?cal?What are the reactions (with phases) and their balanced equation for a.)AgNO3+Na2SO4? b.)Pb(NO3)2+Na2SO4? c.) CaCl2+Na2SO4? AgNO3+Na3PO4? Pb(NO3)2+Na3PO4? CaCl2+Na3PO4? AgNO3+Na2CO3? Pb(NO3)2+Na2CO3? CaCl2+Na2CO3? AgNO3+CuSO4? Pb(NO3)2+CuSO4? CaCl2+CuSO4?2 CoCl2(aq) + 8 NH3(aq) + H2O2(aq) + 2 NH4Cl(aq) 2 [Co(NH3)5Cl]Cl2(s) + 2 H2O(l) In a 50-mL Erlenmeyer flask, cobalt(II) chloride hexahydrate (1.075 g), an aqueous solution of ammonia (1.2 mL, 15 M), ammonium chloride (0.505 g), and hydrogen peroxide (1.0 mL, 30.0% (w/w), density = 1.10 g/mL) were combined. After completion of the reaction, [Co(NH3)5Cl]Cl2 (0.450 g) was isolated. What was the percent yield of the product?
- in a common medical laboratory determination of the concentration of free chloride ion in blood serum, a serum sample is to treated with a Hg(NO3) solution. 2CL-(aq) + Hg(NO3)-(aq) --> 2NO3-(aq)+HgCl2(s) what is the Cl- concentration in a 0.25mL sample of normal serum that requires 1.64mL of 8.35 x 10(negative 4th power) M Hg(NO3)2(aq) to reach the end point?Rx lithium carbonate 300mg Dispense #60 Sig: take 1 capsule by mouth 4 times a day How many milliequivalents of litium are provided daily to the patient by the prescription above? (Lithium carbonate, LiCO3, MW=74) (Li, MW=7)Use the data provided for stock concentration and mass to calculate the concentrations of each solution. Solution A) A stock solution of 0.0100 M solution of K3[Fe(CN)6] is provided. Calculate the volume of the stock solution needed to prepare 100.0 ml of .00200 M K3[Fe(CN)6]. Deionized water should be used to dilute the solution. Solution B) Calculate the volume of solution A needed to prepare 50.0 ml of .0010 M K3[Fe(CN)6]. Deionized water should be used to dilute this solution.
- In electrochemistry, specific conductance is defined as (1000/N)* k, where N is the normality of the salt solution and k is the specific conductance. Current is carried by both anions and cations to a different degree. Thus, the equivalence of a salt is the sum of the ionic conductances of the cation and anion. If the ionic conductance of Na+ is 50.1 and that of Cl- is 76.6. What is the estimated concentration of NaCl in mg/L. The specific conductance of NaCl is 125x106. A) 49 B) 58 C) 29 D) 38 Note: This is not a graded question. This question is from my reviewer.Magnesium Chloride is available as magnesium chloride hexahydrate in an injectable solution that supplies 1.97 mEq of magnesium per milliliter. What is the percent strength of magnesium chloride hexahydrate in this solution?Analytical chemistry is a particular field within the broader spectrum of the chemical sciences, in which many times the focus of analytical experiments is to develop new methods to analyze compounds, either structurally or by determining concentrations of compounds. One of the processes analytical chemists use to determine the exact concentration of a working solution is called standardization. In your own words, describe the differences between a primary, secondary, and tertiary standard, and describe the underlying concept behind the standardization process. Why is it done?
- Some wastewater stream contains 0.015 M Hg22+. Some chemical engineer decides to remove it by precipitating it as Hg2CO3(s). He mixes 100.00 ml of the wasterwater with 100.00 ml of a 2.00 M solution of Na2CO3 and filters the result to remove the solid. How much residual Hg22+ should he expect to find still in the water? (Hint: it should not be 0) Ksp of. Hg2CO3 = 9.0 x 10-15. Also keep in mind that Hg+ is the only ion that exists in solutions as a dimer, i.e. Hg22+, not 2 Hg+ (Hg2CO3→ Hg22+ + CO32-)Mr. Roy, the principal of a reputed school organized a seminar in which he invited parents and principals to discuss the serious issue of diabetes and depression in students. They all resolved this issue by strictly banning the junk food in schools and to introduce healthy snacks and drinks’like soup, lassi, milk, etc. in school canteens. They also decided to make compulsory half an hour physical activities for the students in the morning assembly daily. After six months, Mr. Roy conducted the health survey in most of the schools and discovered a tremendous improvement in the health of students. After reading the above passage, answer the following: What are the values (at least two) displayed by Mr. Roy? As a student, how can you spread awareness about this issue? What are tranquilizers? Give an example. Why is the use of aspartame limited to cold foods and drinks?5. Permanganatometry is used to quantify?A) Iron (II) sulfateB) Zinc sulfateC) Silver nitrateD) Basic bismuth nitrateE) Copper sulfate

