Consider the initial‑rate data at a certain temperature in the table for the reaction described by 2NO2(g)+O3(g)⟶N2O5(g)+O2(g)2NO2(g)+O3(g)⟶N2O5(g)+O2(g) [NO2]0 (M) [O3]0 (M) Initial rate (M/s) 0.650 0.800 2.99×1042.99×104 1.10 0.800 5.06×1045.06×104 1.76 1.40 14.17×10414.17×104 Determine the value and units of the rate constant, ?.
Consider the initial‑rate data at a certain temperature in the table for the reaction described by 2NO2(g)+O3(g)⟶N2O5(g)+O2(g)2NO2(g)+O3(g)⟶N2O5(g)+O2(g) [NO2]0 (M) [O3]0 (M) Initial rate (M/s) 0.650 0.800 2.99×1042.99×104 1.10 0.800 5.06×1045.06×104 1.76 1.40 14.17×10414.17×104 Determine the value and units of the rate constant, ?.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter12: Chemical Kinetics
Section: Chapter Questions
Problem 100AE: Consider a hypothetical reaction between A and B: A + B products Use the following initial rate...
Related questions
Question
100%
Consider the initial‑rate data at a certain temperature in the table for the reaction described by
2NO2(g)+O3(g)⟶N2O5(g)+O2(g)2NO2(g)+O3(g)⟶N2O5(g)+O2(g)
| [NO2]0 (M) | [O3]0 (M) | Initial rate (M/s) |
|---|---|---|
| 0.650 | 0.800 | 2.99×1042.99×104 |
| 1.10 | 0.800 | 5.06×1045.06×104 |
| 1.76 | 1.40 | 14.17×10414.17×104 |
Determine the value and units of the rate constant, ?.
Expert Solution
Step 1
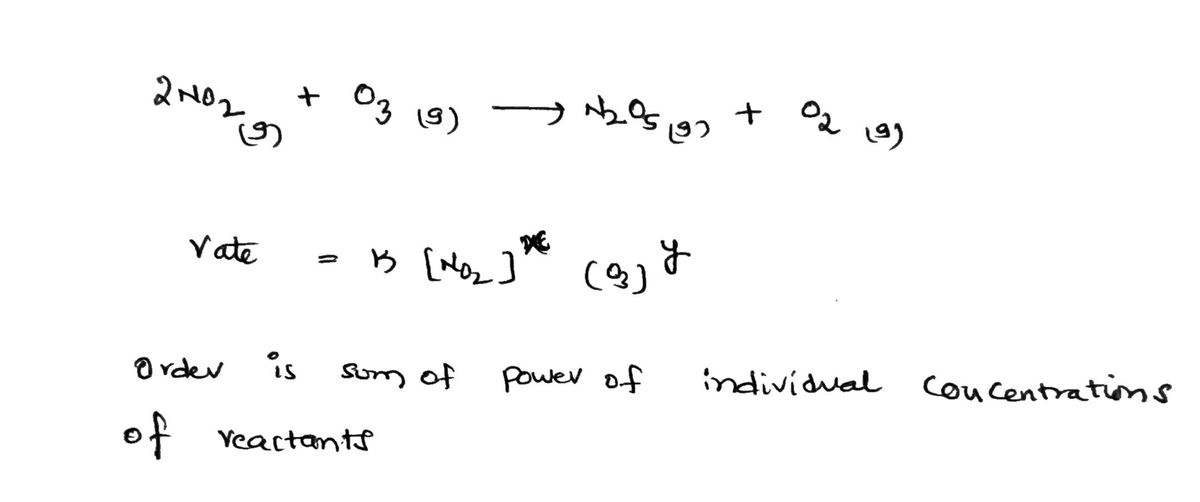
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning