Q: What is the Lewis NH3 D) N Og
A:
Q: How many milliosmoles of sodium and how many milliosmoles of chloride are represented in 2 L of 3% h...
A: The milliosmoles of Sodium and chloride can be calculated as
Q: When prepoaring solution for UV spectroscopy, the more polar the solvent is the better.for higher cu...
A: In UV spectroscopy, we have to choose the solvent to prepare solutions in such a way that It should...
Q: A certain amount of benzene (Density y=0.985 g/mL1 was dissolved in 0.500 kg CCl4 to produce a solut...
A: (a) Solution -
Q: a) What is the advantage of very small particle sizes in LSC columns? (b) With reference to the Va...
A: a) Small particle sizes are used in the LSC columns.
Q: An aqueous solution with 15 g sulfuric acid is dissolved to make 0.2F solution. The specific gravity...
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for you...
Q: What is the oxidation state on Ec in the formula below? Ec4O18
A: Oxidation number: The covalent bond between the atoms of different electronegativity is polar. If we...
Q: 3. Calculate the mass, in grams, of 0.160 mol H,O2-
A: Detail mathematical calculation is given below
Q: In a lab you are measuring the heat of reaction for an unknown reaction that occurs in solution: A +...
A:
Q: What are the similar physical properties of open-chain hydrocarbons and closed-chain hydrocarbons?
A:
Q: Which one of the alkyl chlorides would you expect to undergo an SN1 process most readily and which o...
A:
Q: Calculate the pH of a 0.048 M solution of the following compounds. NH3. Kp(NH3) = 1.8E-5. eTextbook ...
A:
Q: Provide a detailed step-by-step mechanisms for the reaction shown. CH;CH2SH + Br, + NaOH
A:
Q: Exercise 18 Find the density of F2 gas at 20.0°C and 188 Torr. [Answer: 0.39 g dm]
A: Given 13) P1 = 90 kpa , V1 = 20 dm3 , T1 = 30°C = 303 K P2 = 250 kpa , V1 = ? , T1 = 20°C = 293 K...
Q: Use two concentration tables to calculate the concentrations of the species present in a 0.390 M sol...
A:
Q: A sample of Zn(s) is reacted with HCl(aq) to form hydrogen gas. The H2 gas bubbles out of aqueous so...
A: Zinc metal ( Zn ) reacts with Hydrochloric acid ( HCl ) to produce Zinc chloride ( ZnCl2 ) and Hydr...
Q: Problem 68 2266 IR Spectrum iquid im) 1749 2000 V (cm") 4000 3000 1600 1200 800 Mass Spectrum No sig...
A: Spectroscopy is the one of the important analytical techniques to confirm the structure of the compo...
Q: collision theory of reaction rates states that the rate of a reaction depends on the number of colli...
A: We should not be surprised that atoms, molecules, or ions must collide before they can react with ea...
Q: Read through each scenario. Under the scenario, write which lab safety rule is being broken and indi...
A:
Q: 32. Write the condensed structural formula for the C3H7C1 molecules you drew. (1) (2) elob 33. How d...
A: Isomers are those compounds which have same molecular formula but different structural formula.
Q: unit 3 Test - Energy in Which statement about the change in bond energy of this reaction is correct?...
A: Reaction :- CH4 + 4Cl2 ------> CCl4 + 4HCl
Q: Concentrated aqueous HCIO4 has a concentration of 14.8 M. Calculate the concentrations of all ions p...
A:
Q: What methods could be used to determine the structure of polyiodides? Discuss how could they identif...
A:
Q: What mass of CO is required to react with 25.13 g of F*e_{2} * O_{3} according to the equation : Fe ...
A:
Q: Some measurements of the initial rate of a certain reaction are given in the table below. N2H2 initi...
A: For a reaction aA + bB →cC + dD rate = K [A]a [B]b
Q: Using the compound elements below. Find the following i. Non-Bonding and Bonding Domain ii. Molecu...
A: VSEPR theory: The structure of a molecule can be determined by using the VSEPR theory. According to ...
Q: When the equation below is balanced , H 2 O 2 (aq)+H^ + (aq); +Fe^ 2+ (aq) H 2 O(l); +Fe^ 3+ (aq)
A: H2O2(aq) + H+(aq) + Fe+2(aq) -------> H2O(l) + Fe+3(aq) We have to balance the above equation.
Q: 1. An organic compound was found to contain 54.5% C, 9.2% H, and 36.3% O. a. What is the empirical f...
A:
Q: (Q-2) Estimate the thermal conductivity of liquid acetone in (Btu/hr.ft.F),at (25 C) given; Isotherm...
A: Soln
Q: what is the ΔG when - K constant = 1.2x103 - temperature = 40 degrees - R = 8.31
A: Gibbs Free Energy Equation, ∆G = ∆G° + RT(lnK) ∆G = Gibbs Free Energy ∆G° = Standard Gibbs Free Ener...
Q: Benzene is one of the Toxic Air Pollutants (TAPS) found in car exhaust fumes When a 50.00 ml. aqueou...
A: It is given that the fractional distillation of 50.00 mL of aqueous solution of 0.05000 M benzene is...
Q: Find the volume in liters of 8.00 g O2 at STP
A:
Q: Indicate the type of IMF of the each and rank them based on increasing intermolecular forces
A: As per the guideline, since you have asked multiple questions, we have solved the first question for...
Q: HSO4 OH So,2 H20 +
A: Proton (H+) donors are acids and Proton acceptors are bases. HSO4- ------> SO42- HSO4- act as ac...
Q: e the molar mass of each compound. Show yo
A:
Q: e Van Deemter equation,
A: VAN DEEMTER EQUATION ANALYSIS
Q: A solution is prepared by dissolving 27.03 grams of FeCl,* 6H;O, and diluting to give a final volume...
A: The molarity (M) of a solution is the ratio between the number of moles (n) of solute to the solutio...
Q: Devise 2 synthesis for the given compound starting from BENZENE and other pertinent inorganic or org...
A:
Q: a. Mg I Mgaa) l Cuag) I Cus) b. Nice I Niaa) | Agiaa) I Ag) 3. For the cell notations in the previou...
A: Here from the given Galvanic cell notation we have to write the cell reaction. For this we have to w...
Q: In the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses...
A: Given that, lysergic acid diethylamide (LCD, C20H25N3O). It is a weak base, having Kb = 7.6×10-7. Th...
Q: What are the uses of polyiodides? What are the importance of polyiodides?
A: The answer to the following question is-
Q: c. 20.45 g Al(NOsla 5. A saline solution with a mass of 233 g has 34.5 g NaCl dissolved in it. What ...
A:
Q: Given the following information, calculate the missing values for multiple different solutions of Ba...
A:
Q: (0.1mol) of NaoH was added to 1L of a solution containing a weak acid and its base salt had the same...
A:
Q: Use Hess's Law and the three reactions, below, to calculate the standard enthalpy of reaction (AH°rx...
A: The reaction for the coal gasification is, 2C(s) + 2H2O(g) ---> CH4(g) + CO2(g) The three re...
Q: 1. Methyl Alcohol and ethyl alcohol form ideal solutions. At 500C, the vapor pressure of pure methyl...
A: 1) Raoult's law: According to the Raoult's law at any particular temperature the partial pressure of...
Q: Match column A with Column B. Given: Samples were spotted on the TLC plate and after performing the ...
A: This question is related to Thin Layer Chromatography. Thin-layer chromatography (TLC) is a techniq...
Q: Mass of NaOCl in commercial bleach titrated. ?
A: Moles help to calculate mass
Q: Intravenous, or IV, solutions used in medicine must exert the same osmotic pressure as blood to prev...
A: Mass of NaCl = 0.9g Mol. Mass of NaCl = 58.5g/mol Volume of solution = 100ml i = Van't Hoff factor =...
Q: 1.75 g of C:H6 and 6.75 g of Oz are placed in a vessel where they react according to the following b...
A: Given, 2C2H6 + 7O2 ➝ 4CO2 + 6H2O Mass of C2H6 reacts = 1.75 g Mass of O2 reacts = 6.75 g Note: Mola...
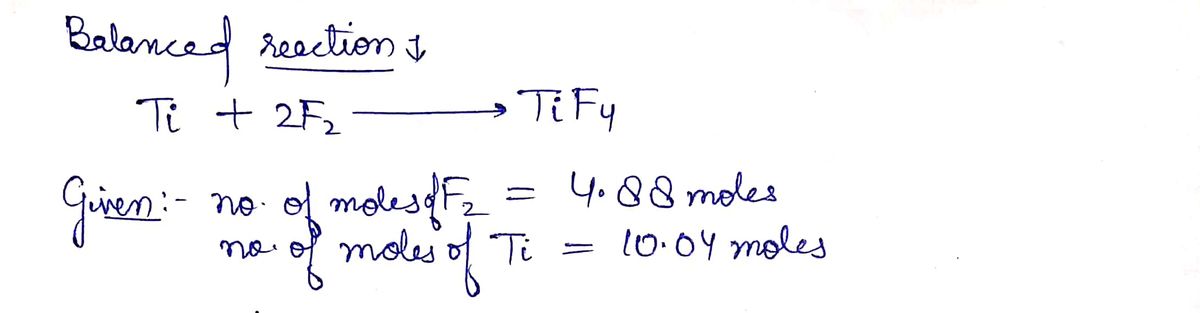
Step by step
Solved in 2 steps with 2 images

- Fe in ore can be analyzed by oxidizing Fe to Fe2+ then titrating Fe2+with K2Cr2O7 . See equation below. K2Cr2O7(aq) + 6Fe2+(aq) + 14H+(aq) → 2Cr3+(aq) + 2K+(aq) + 6Fe3+(aq) + 7H2O(l) What is the stoichiometry of the analyte and titrant ? a. 6 mol titrant to 1 mol analyte b. 6 mol analyte to 1 mol titrant c. 2 mol titrant to 1 mol analyte d. 2 mol analyte to 1 mol titrantAll arsenic in the mineral containing 20 g of arsenic is dissolved by converting it into As3+. Calculate the percentage of arsenic in the mineral since the solution is titrated with 43.8 mL of decinormal KBrO3 solution.0.683 grams of iron mineral dissolves with acid. Fe2+ is titrated with Ce(SO4)2 solution. Calculate the percentage of iron since 38.5 mL of 0.161 M titrant is consumed in the titration. ( Fe:56 g/mole)
- 1. What is the oxidation state of Ti in TiO2? Group of answer choices 2- 2 4 0 4- 2, 2.Which element is oxidized in the reaction below? Fe2+ + CrO42- + H2O→ Fe3+ + Cr3+ +OH- Group of answer choices A, Cr6+ B, O2+ C, Cr3+ D, Fe2+ E, Fe3+25.00 mL of a solution containing KIO4 is treated with excess potassium iodide. IO4- + 8H+ + 7I- → 4I2 + 4H2O The resulting solution is titrated with 0.15 M sodium thiosulfate pentahydrate. The average titre was 21.76 mL. I2 + 2S2O32- → 2I- + S4O62- What mass of KIO4 (in g) was dissolved in water to make the original solution?A sample of an iron ore is dissolved in acid, and the iron isconverted to Fe2+. The sample is then titrated with 47.20 mLof 0.02240 M MnO4- solution. The oxidation-reduction reactionthat occurs during titration isMnO4-1aq2 + 5 Fe2+1aq2 + 8 H+1aq2¡Mn2+1aq2 + 5 Fe3+1aq2 + 4 H2O1l2(a) How many moles of MnO4- were added to the solution?(b) How many moles of Fe2+ were in the sample? (c) Howmany grams of iron were in the sample? (d) If the samplehad a mass of 0.8890 g, what is the percentage of iron in thesample?
- The oxidizing agent in the reaction below is 3CuCl2(aq)+2Al(s) →3Cu(s)+2AlCl3(aq) Cu2+ Al Cu Al3+Iron can be extracted from haematite, Fe2O3, using either C or CO2 as the reducing agent.The reactions are shown below.Fe2O3(s) + 3 C(s) → 2 Fe(s) + 3 CO(g) ΔH = +492.7 kJ /molFe2O3(s) + 3 CO(g) → 2 Fe(s) + 3 CO2(g) ΔH = - 24.8 kJ/molSubstance S° (J/ K·mol)Fe2O3 (s) 87.4Fe (s) 27.3C (s) 5.7CO (g) 197.6CO2 (g) 213.6Use available data given to:1. Calculate the minimum temperature at which reduction with carbon is feasible2. Prove by calculation that reduction with carbon monoxide is feasible at all temperaturesWhich substance is the oxidizing agent in the reaction below?Fe(CO)5 (l) + 2HI (g) →→ Fe(CO)4I2 (s) + CO (g) + H2(g) HI Fe(CO)4I2 Fe(CO)5 H2 CO
- 0.1745 g of primary standard Na2C2O4 is used to set a freshly prepared KMnO4 solution and 30.24 mL of KMnO4 solution is spent at the turning point. Since 36.92 mL of this KMnO4 solution is consumed for the determination of iron in 0.5618 g ore sample, calculate the amount of iron in the ore in terms of % iron (III) oxide and % iron (III) chloride. (Fe: 56, K: 39, Mn: 55, O: 16, Na:23, C: 12, Cl: 35.5 g/mol) (Please type answer)The reaction of hydroxylamine and Fe(III) produces nitrous oxide, Fe(II) and water: 2H2NOH + 4Fe3+ N2O(g) + 4Fe2+ + 4H+ + H2O. A 50 ml sample of hydroxylamine was treated with Fe(III) solution and the released Fe(II) was titrated with 0.0325 M K2Cr2O7 solution, which consumed 19.83 ml. Calculate the hydroxylamine concentration of the sample.0.683 grams of iron mineral dissolves with acid. Fe2+ is titrated with Ce(SO4)2 solution. Calculate the percentage of iron since 38.5 mL of 0.161 M titrant is consumed in the titration (Fe:56 g/mol)

