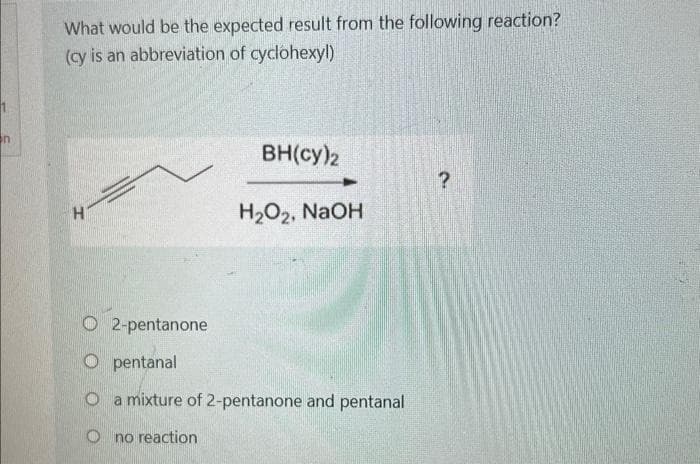
n What would be the expected result from the following reaction? (cy is an abbreviation of cyclohexyl) BH(cy)2 ? H H₂O2, NaOH 2-pentanone Opentanal O a mixture of 2-pentanone and pentanal Ono reaction
Q: What is the solubility of SrCO₃ in water? (Ksp of SrCO₃ is 5.6 × 10⁻¹⁰)
A:
Q: <HW 4: Chapter 14 (Aldehydes and Ketones) - Attempt 1 Question 4 Part A Spell out the common name of…
A: Step 1: Information : As the given example having functional group RCOR It indicates the presence…
Q: None
A: To obtain 145 mL of Cl2(g) at 25 °C and 725 Torr by the reaction of MnO2(s) with excess HCl(aq),…
Q: Question 5 Which set of Newman projections represents correct the most stable and the least stable…
A: Question 5: The most stable is when they CH3 are anti to each other180∘. Whereas the least stable…
Q: The following reaction involves an CI A O a. intermolecular SN1 reaction. O b. intramolecular SN2…
A: Step 1:Q.3 option d not clear in image and options d is correct if this question contains four…
Q: Identify the Haworth structure showing the ẞ-isomer of the following Fischer projection. H (Hint:…
A: Beta form -OH group is above the plane, here there are two form in which OH is above the palne . A…
Q: Consider the reaction 2NO(g) + O2(g)->2NO2(g) Using the standard thermodynamic data in the tables…
A: The prompt describes a scenario where a chemical reaction takes place between NO, O2, and NO2 gases.…
Q: Question 12 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: Circle If the compound below is chiral, and provide R/S assignments where appropriate: اف + H CH3
A: Step 1: Step 2: Step 3: Step 4:
Q: 2. Tetrodotoxin (TTX) is a potent neurotoxin. Its name derives from Tetraodontiformes, an order that…
A: Objective: Inspired by the structure and function of tetrodotoxin (TTX), the proposed research…
Q: Consider the following system at equilibrium where K = 1.80×102 and AH° = 10.4 kJ/mol at 698 K. 2 HI…
A: First Question: 1.) TRUE. The enthalpy change (ΔH) of the reaction is positive, which indicates that…
Q: Draw the most stable chair conformation of each of the following compounds. (a)cis-1,1,4 -…
A: Step 1: Step 2: Step 3: Step 4:
Q: 53) The valence electron configurations of several atoms are shown here. How many bonds can each…
A: Step 1:Valance electron:The electrons which are present in the outer most energy level are known as…
Q: Determine the predominant type of reaction: SN1, SN2, E1 and E2. Draw the major products only. Show…
A: Step 1: Step 2: Step 3: Step 4:
Q: Part a through b please Part a) In the following alum synthesis reaction 1.50 grams of aluminum…
A:
Q: Drawing Lewis Structures For each molecule, calculate the total number of valence electrons. Then,…
A: Detailed explanation:Examples:H2SO4
Q: 1. Compare ... M 2. Standard ... 3. AS surroundi... 4. AG° = AH°... 5. AG: Pre... 1req 6. AG:…
A: Step 1:First, we need to find the standard free energy change of the reaction, ΔG°. We can do this…
Q: Show work, thank you!
A: The attached image mostly shows gray screen. No question was asked. It would be better to…
Q: ABCD Question 4 Please predict the product for each of the following reactions. Make sure to clearly…
A:
Q: Please don't provide handwritten solution. Show how the following synthesis can be performed.
A: Step 1: Step 2: Step 3: Step 4:
Q: Indicate the name of the compound in the figure and indicate how it is obtained from 1-(3-…
A: Sure, I can help you with the question based on the image you sent.The question asks you to identify…
Q: If, in Part 2 (high temperature), a student uses tap water to create the solution rather than DI…
A: If a student uses tap water containing calcium ions (Ca²⁺) in Part 2 (high temperature) instead of…
Q: Choose the structure below that correctly represents the intermediate that is observed after step 1…
A: The image you sent is a question about aminoacylation by GlyRS, not aminooxidation by…
Q: B HO HỌ HỌ HO OH A но. HỌ HỌ HO OH
A: The question asks:The researchers wanted to find out if there is a sugar alcohol isolated from the…
Q: Write the systematic name of each organic molecule: structure CH3 CI- CH-CH2- CH=CH2 CH2 CI C-CH2- -…
A: Thank you.
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: The process depicted in the image is an example of an electrophilic addition reaction. To understand…
Q: Show work, thank you!
A: There is no way to answer this question with out proper data. Please provide the proper information…
Q: Name these organic compounds please:
A: Step 1: Notice that all the given compounds only have single bonds. This means that all three…
Q: What is the synthetic scheme that can be used to produce the target molecule. Pay attention to stero…
A: Step 1: Step 2: Step 3: Step 4:
Q: (c) ? Br OEt OH O
A: Step 1:The diagram represents the reaction of an alcohol (OH) with a strong base (Br) to form an…
Q: Question 8
A: Step 1: Step 2: Step 3:
Q: :$:$;$;$;$;$ draw out neatly
A:
Q: None
A: Step 1: IUPAC (International Union of Pure and Applied Chemistry) nomenclature is a systematic…
Q: None
A: Given:…
Q: Predict the products of the following reactions. Show stereochemistry when applicable.
A: #1 is a hydroboration-oxidation reaction. it is a reaction that uses anti-Markovnikov addition of…
Q: The formation constant* of [M(CN)2] is 5.30 x 1018, where M is a generic metal. A 0.150 mole…
A: Given: molM+=0.150mol;[CN−]=0.790M;V=1.00LKf(M(CN)2−)=5.30x10−18;[M+]=???MStep 1: Write the…
Q: Consider the reaction CO(g) + H,O(l) _CO,(K) + H(8) for which AH° = 2.800 kJ and AS° = 76.80 J/K at…
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. How much heat energy, in kilojoules, is required to convert 74.0 g of ice at −−18.0 ∘C to water…
A: Given:mass of ice = 74.0gInitial temperature of ice = -18°Cfinal temperature = 25.0°CSpecific heat…
Q: 15 1/1 point Which of the following reactions has a positive AS? Select all correct answers. Na(s) +…
A: A ( g ) + B ( g ) → 3 C ( g ) : There are 3 moles of products and 2 moles of reactants, so this…
Q: Show work, thank you!
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Help me identify the propagation, initiation, and termination
A: Step 1: Step 2:Step 3: Step 4:
Q: Question 20 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A: Reaction of dihalide with excess strong base forms an alkyne.Reaction of alkyne with n-BuLi and a…
Q: ABCD Question 1 Please predict the product for each of the following reactions. Make sure to clearly…
A:
Q: Question 27 Predict the FINAL (?) product (or a mixture of products) for the following synthetic…
A:
Q: વિનાનું Propose a chemical stepwise process to achieve the conversion Shown below. State the…
A: In case of any doubt please feel free to ask.
Q: Draw a Haworth projection of a common cyclic form of this monosaccharide:
A: The sugar in open chain form is a 5-carbon aldose (aldopentose) known as D-Arabinose.It is…
Q: Organic Chemistry problem. Please help solve. Suggest reaction that will allow the transformation…
A:
Q: I really need help on how to solve this problem ! I would appreciate any help :)
A: Step 1:Enthalpy change value for any reaction…
Q: Draw the product of the reaction below. Ignore inorganic byproducts. OH OH OH H H H H H OH OCH 3 Q…
A:
Q: Using the retrosynthetic analysis propose a logical route for the following transformation. Draw all…
A: In the given question, the starting reactant is given i.e. ethylidenecyclohexane and the final…
Step by step
Solved in 2 steps with 1 images

- Draw the major product of the reaction between 1-butanol and Na2Cr2O7, H2SO4, H2O.How would you synthesize triphenylmethanol starting from the following class of compound? Draw the structure of the starting compound and list the other reagent(s) used for the reaction. a. Ester b. Ketone c. Aldehyde eduction dd H IT More "0" Oxidiz strong leak ReduDraw structures corresponding to the following names: (a) 3-Methyl-1, 2-benzenediamine (b) 1, 3, 5-Benzenetriol (c) 3-Methyl-2-phenylhexane (d) o-Aminobenzoic acid (e) m-Bromophenol (f) 2, 4, 6-Trinitrophenol (picric acid]
- The reagent NaBH4 acts as an ____ 1. H+ (hydride), 2. H- (hydride), 3. H+ (proton), 4. H- (proton) delivery agent. In a reaction with NaBH4 and methanol, the carbonyl (C=O) carbon atom of a ketonre is reduced or oxidized? to give 1. ether 2. alcohol 3. alkane 4. aldehyde 5. an ester product?2. What is the organic by-product in a haloform reaction? A) an ether B) a haloform C) an alcohol D) an ester E) an amideDraw a structural formula for the product of the reaction of acetophenone with reagent Q. N2H4, KOH at 250°C
- What carboxylic acid is formed from attached alkyl halide on treatment with [1] Mg; [2] CO2; [3] H3O+?3117 Is the starting compound an ester and an OMe? Where do you add the CH3CH2 of the Grignard reagent on to the product? What is the right answer? Is it right to show the MeOH as one of the products?Draw the carbon containing products of the following reaction: Reactant = 2-bromo-2-methyloctane Reagent = CN minus, room temperature
- (A true story.) A drug user responded to an ad placed by a DEA informant in a drug-culture magazine. He later flew fromColorado to Maryland, where he bought some 1-phenyl-2-propanone (P2P) from the informant. The police waited nearlya month for the suspect to synthesize something, then obtained a search warrant, and searched the residence. They foundthe unopened bottle of P2P; apparently, the suspect was not a good chemist and was unable to follow the instructions theinformant gave him. They also found pipes and bongs with residues of marijuana and cocaine, plus a bottle of methylamine hydrochloride, some muriatic acid (dilute HCl), zinc strips, flasks, and other equipment.(a) Assume you are consulting for the police. Show what synthesis the suspect was prepared to carry out, to provideprobable cause for the charge of attempting to manufacture a controlled substance.(b) Assume you are a member of the jury. Would you convict the defendant of attempting to manufacture a…What is the product formed when 1-ethynylcyclohexane is reacted using mercury (II) acid catalyzed hydration?How do you produce an ester from a carboxylic acid? 1. Heat the acid with an alcohol and catalytic acid 2. Deprotonate the acid with a base and react with an alcohol 3. Deprotonate the acid with a base and react with an alkyl halide A) 1 only B) 2 only C) 3 only D) 1 and 2 E) 1 and 3



