Q: 6. Iron reacts with oxygen gas to form iron (III) oxide according to the chemical equation shown 4イF...
A:
Q: Which statement best describes the properties of NaCl? Question 1 options: It is an inorganic ...
A: NaCl is an inorganic compound commonly named as table salt. It is an ionic compound formed by donati...
Q: A crude nonacidic product mixture dissolved in diethyl ether contains acetic acid. Describe an extra...
A: We have a crude non acidic product mixture dissolved in diethyl ether contains acetic acid. We have ...
Q: 3. Assign and label R/S configuration for all stereogenic centers and assign E/Z to all alkenes on t...
A: R/S configuration: If 4th group is present on wedge then R convert to S or vice versa. E/Z: E--->...
Q: 1. 50.0 ml of 0.1 M NaOH is added to 100.0 ml of 0.2 M HCN (Ka=6.2x10-10). What is the pH of the sol...
A: Since you have posted multiple questions, we are entitled to answer the first only. 1) Given: Concen...
Q: A proposed mechanism for the gas phase reaction of chloroform (CHCl3) and chlorine is as follows: 1....
A: The relation between the concentration of the reactants and the rate of reaction is expressed by the...
Q: not exist in water. For example: Na2O(s) + H2O → 2 NaOH(aq) 6. All salts containing PO43-, CO32-, S...
A: Soluble salts dissolve in aqueous solution and insoluble salts form precipitate in solution.Here we ...
Q: Consider the following reaction and initial rate data: 2H2 (g) + 2NO (g) → 2H,0 (g) + N, (g) [NO]ini...
A:
Q: A precipitate of PbI, is formed when 5 mL of 0.012 M Pb(NO,), is added to 5 mL of 0.030 M KI. It was...
A:
Q: Chemists have defined the pH (Hydrogen Potential) of a solution by pH = -log[H+] where [H+] represen...
A: • The values provided in the question are:- i) pH of a destructive acid rainfall = 2.7 • We n...
Q: II. Calculate AH, for each of the following (using Table 1). + CHa from the reaction CH.(8) • 20:(8)...
A:
Q: What is the rate constant for this reaction? Do not report units. Report the number with 2 places pa...
A:
Q: Which of the following equilibrium constants corresponds to a situation where AG = 0? OK>1 ОК»»»1 OK...
A:
Q: Which type of bonds are present in a double bond? Two pi bonds One sigma and one pi bond Two sigma b...
A:
Q: 4. Apply the real rule of significant figures to express the final result for each of the values giv...
A: The result of addition or subtraction in the numbers having different precision should be reported t...
Q: Use the equation below to determine the amount of nitrogen needed:N2 + 3 H2 ? 2 NH3 A student in l...
A:
Q: (a) To what temperature must a 1.0 L sample of a perfect gas be cooled from 25 °C in order to reduce...
A: Given: Initial volume = 1.0 L = 1000 cm3 Initial temperature = 25 oC And final volume = 100 cm3
Q: A molecule absorbs radiation of frequency 3.00x1014 Hz. What is the energy difference between the mo...
A:
Q: how many grams of CO are formed from 0.184 g of O2
A: Recall the reaction that will occur between C and O2 to form CO 2C(s) + O2(g) ---> 2CO(g) We hav...
Q: Give the balanced chemical equation for each of the following reactions. 1. Identification of chromi...
A: Here we have to identify the following ions.
Q: rate of reaction =
A:
Q: In each of the following molecules 1-6, identify the relation between the circled hydrogen atom as h...
A: On the basis given questions , I have find the relationship between circle Hydrogen.
Q: The vapor-liquid reactive equilibria for ethyl lactate synthesis follows the expression log K = 7.89...
A: The vapour-liquid reactive equilibria equation given is, => log(K) = 7.893 - 2.4312 × 103T And te...
Q: . explain the results of the experiment Table 1 Absorption Spectrum of Cobalt Chloride Using a Spe...
A: Solution: We know in spectrophotometric/ colorimetric experiment, concentration of the unknown compo...
Q: How many moles of oxygen (O2) are needed to react with 56.8 grams of ammonia (NH3) by this reaction?
A:
Q: sample of rainwater was determined to have a pH of 6.35. What were the H+ concentrations of the samp...
A: The PH of the solution can be determined by using a formula that is PH = - log[H+]
Q: Given this information, what are the 2 compounds? (Second image has a molecular formula of c4h8o2)
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: Fuel used in nuclear fission is U-235. In the process, neutron particle is used to split U-235 into ...
A: We to write the nuclear reaction for nuclear fission is U-235 which undergo by neutron particle whic...
Q: 1. 50.0 ml of 0.1 M NaOH is added to 100.0 ml of 0.2 M HCN (Ka=6.2x10-10). What is the pH of the sol...
A:
Q: Determine the value of the equilibrium constant, Kgoal, for the reaction N2(g)+O2(g)+H2(g)⇌12N2H4(g...
A: Answer: Value of equilibrium constant is the ratio of the molar concentration of products and reacta...
Q: For each of the following sets of experimental data, using a spreadsheet, determine From the slope,...
A: To find: Activation energy, A and K at different temperatures.
Q: e concentration of M
A:
Q: 4 Al (s) + 3O2 (g) → 2 Al2O3 (s) ΔHo = -3351 kJ/mol Consider the following statements about the...
A:
Q: In the three-dimensional structure of methane CH4 , the hydrogen atoms attached to a carbon atom are...
A:
Q: Explain why it is necessary to titrate the vinegar and milk.
A: Vinegar is acetic acid.It has formula CH3COOH.Milk is mainly protein,along with it has fat,lactose,m...
Q: 9. Draw the following compounds a. 3-methylbutanal b. 3-methyl-2-butanone C. 4,6-dimethyl-2-nonanone...
A:
Q: Why is CaO is not named Calcium (ii) Oxide?
A: The Roman numeral is needed to mention when the metal ion has multiple possible charges or oxidation...
Q: Write a balanced net ionic equation a for the reaction of the following weak base with water: acetat...
A:
Q: Question 8 please
A: mass of methanol = 48.9g Initial temperature, Ti = 82.5°C Final temperature, Tf =0°C Boiling te...
Q: Using Faraday constant, how many grams of copper will attach to the cathode of an electrolysis cell ...
A: The Faraday first law states that the amount of material deposited at cathode from electrolytic solu...
Q: The solubility of the hydroxide of a hypothetical metal, Me, in water is 94.89 g MeOH per 64.88 mL w...
A: The amount of water is needed 42.82 mL.
Q: cis/trans isomerism can identify the rotation of a compound based on a reference double bond. a T...
A:
Q: Three moles of NaCl is dissolved in pure water. How many moles (to the nearest whole number) of solu...
A: The dissociation reaction of NaCl is given as, => NaCl (s) → Na+ (aq) + Cl- (aq) Given: Moles of ...
Q: A point group has the following sets of elements E, 8 C3, 3C2, 6 S4, 6 od. The order and number of i...
A: We know tetrahedral point group has E, 8 C3 , 3 C2 , 6 S4 and 6 σd. Where the numbers before symmetr...
Q: Classify each of the following as a binary ionic compound, ternary ionic compound, binary molecular ...
A:
Q: The vapor-liquid reactive equilibria for ethyl lactate synthesis follows the expression log K = 7.89...
A: The vapour-liquid reactive equilibria equation given is, And temperature = 85.0 oC
Q: What is the entropy change when 1.55 moles of CHCl₃ vaporizes at 61.0 °C? [∆H(vap) = 29.6 kJ/mol at ...
A:
Q: Consider the reaction 2H2 + O2 = 2H2O + energy Which will have higher energies, (H2 + O2) mixture o...
A: Given: Bond enthalpy for C-C = 3.47 ×105 J/mol Bond enthalpy for C=C = 6.11 × 105 J/mol Bond enthal...
Q: 526 g of NaCl (m.m. = 58.44 g mol–1) is dissolved in pure water. How many moles (to the nearest whol...
A: Number of moles of solute is calculated by dividing the given mass with molecular mass.
Q: 3,369 of dia tome gas occupind dia tomic upled a vo lume 2,00 dm3 orcupies at 'an°c of OC and 150 k ...
A: Using ideal gas equation. calculate the number of mole of the gas. Then by using the given mass of g...
Determine each structure to be chiral or achiral.
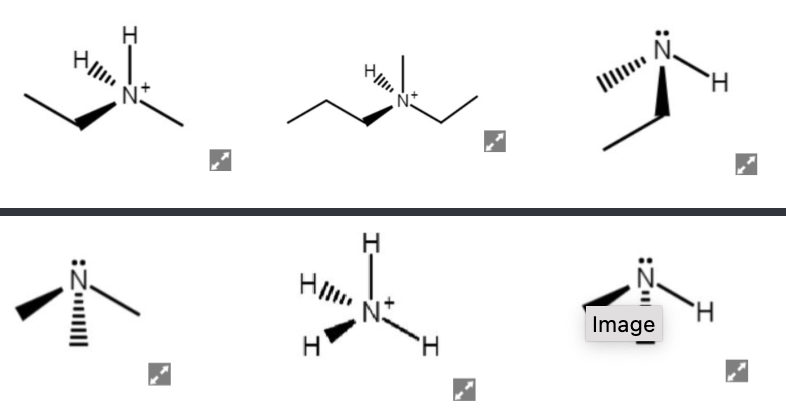
Step by step
Solved in 2 steps with 1 images

- Kf kb eq..Real life applications of standardization of NaOH.The average bond enthalpies of the C—F and C—Cl bondsare 485 kJ/mol and 328 kJ/mol, respectively. (a) What isthe maximum wavelength that a photon can possess andstill have sufficient energy to break the C—F and C—Clbonds, respectively? (b) Given the fact that O2, N2, andO in the upper atmosphere absorb most of the light withwavelengths shorter than 240 nm, would you expect thephotodissociation of C—F bonds to be significant in thelower atmosphere?



