Q: A. Write letter A before each number if the chemical name matches the chemical formula and B if it d...
A: A chemical formula defines the number of atoms of each element in a compound.
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients ...
A: Cr3+ + HClO ....> Cr2O72- + Cl2 In above reaction, Cr is oxidised from +3 to +6 oxidation state. ...
Q: is nitrogen trioxide same with nitrate ion?
A: They are not same.
Q: How many grams of boric acid should be used in compounding the following prescription? Report to the...
A: Prescribed drug is isotonic solution. From theoritical value,nacl equvalent of proparcaine hydrochl...
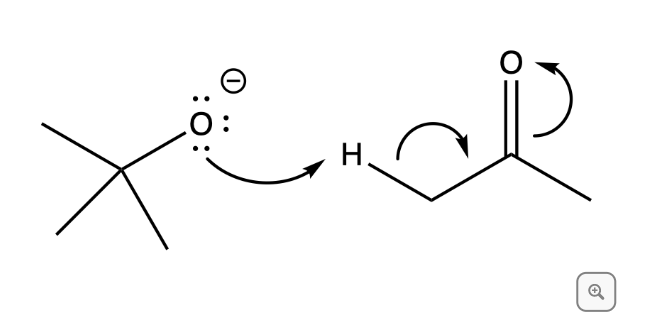
Q: Using the arrows provided in the image, predict the product of this reaction. Include all lone pairs...
A: Hydride ion acts as a base here and deprotonate OH
Q: During Experiment 2, what component of the prepared buffer solution reacted during the addition of t...
A:
Q: Complete the blanks in each column, as in the first example: Name of the Nitride Chloride ion Carbon...
A:
Q: II. Von Weirm an ratio. Fill in the Blanks With the given conditions during precipitation, conclude ...
A: van-Weirman ratio is expressed as: Relative supersaturation=Q-SS (1)where,Q: Concen...
Q: gram, show the similarities and differences between the atomic models of Rutherford, Bohr and the qu...
A:
Q: Use the data reported in Table 1 and plot two separate graphs of measured pH against the volume of N...
A: We need to plot graphs for pH against volume of NaOH added.
Q: Heterolytic reactions involve: A) Bond-breaking where each of the fragments retains one of the orig...
A: What is heterolytic reaction is given below.
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of potassium hydroxid...
A: In acid base neutralization reaction, salt and water is formed. Here NaCl is produced as a salt. The...
Q: Based on the two graphs , draw conclusions regarding the buffer and water solutions ability to resis...
A:
Q: A person's blood alcohol (C2H5OH) level can be determined by titrating a sample of blood plasma with...
A: Mass percent :- The mass of solute in grams present in 100 grams of solution is defined as mass perc...
Q: QUESTION 7 WHEN DO WE USE SIMPLE DISTILLATION AND WHEN DO WE USE FRACTIONAL DISTILLATION?
A: Distillation is the process of purifying the liquid. In this process, the liquid is converted into v...
Q: Below is the graph for the melting point of the period 6 elements versus atomic number. Determine th...
A:
Q: of elementary step based on the mechanistic
A: It is Tertiary Alkyl halide. So, it follows Sn1 mechanism. And it is two step mechanism.
Q: Arrange the following molecules according to increasing ease of evaporation. CH3 H3C-CH3 1 H3C 3 H3C...
A: lower the boiling point, more easily it evaporates. in propanoic acid or in compound 2, there is int...
Q: Gaseous butane (CH3(CH,) CH;) reacts with gaseous oxygen gas (O2) to produce gaseous carbon dioxide ...
A:
Q: Classify the following compounds based on their functional groups. CH3 H3C A H3C CH3 H3C H3C CH3 I-
A:
Q: 1. Place 5 mL of starch solution in each of 2 test tubes. 2. Heat the test tubes to boiling and add...
A:
Q: When a piece of aluminum metal is dropped into hydrochloric acid, HCl, hydrogen is released as a gas...
A: Given : Reaction of Aluminum with HCl To find : Balance reaction
Q: How to calculate theoretical mass percentage of water in a solution
A: Here we are aksed how to calculate theoretical mass percentage of water in a solution.
Q: 1e
A:
Q: You have the following solutions available in the lab: NH4Cl, NaOH, HCl, and CuSO4. 1. Predict w...
A: Solubility of a salt is aqueous solution depends on the its cationic and anionic composition. If lat...
Q: 3. Give the systematic names of the following compounds: Br -CI HO. HO
A: Few questions based on nomenclature, which is to be accomplished.
Q: please write the formal charge of nitrogen trioxide on each atom and the total charge,,,,,
A:
Q: (+) H9OAC
A: A) Electron pair
Q: What is the standard free energy change, A°G, for the following reaction at 298K? C;H,OH(1) + 30:(g)...
A: Consider the given reaction is as follows; C2H5OH l + 3 O2 g ⇄ 2 CO2 g + 3 H2O g The...
Q: What volume of a 0.282 M hydrobromic acid solution is required to neutralize 24.5 mL of a 0.102 M ca...
A:
Q: A student was given two metal cubes that looked similar. One was 1.05 cm on an edge and had a mass o...
A: We have to predict whether given cubes are same or not.
Q: When the following equation is balanced properly under basic conditions, what are the coefficients o...
A:
Q: Potassium hydrogen phthalate is a solid, monoprotic acid frequently used in the laboratory to standa...
A: The given data is as follows: The molarity of barium hydroxide = 0.370 M The mass of KHC8H4O4 = 4.24...
Q: Below is the graph for the melting point of the period 6 elements versus atomic number. Determine th...
A:
Q: How many oxygens are in (NO3)2? A. 1 B. 2 C. 3 D. 6
A: NOTE: Looks like the metal is missing. But from the given formula, we can say that the metal is a bi...
Q: What sugar units will be formed after the hydrolysis sucrose? A. 2 glucose units B. glucose and ga...
A: Sucrose is a disaccharide which on hydrolysis gives the monosaccharides from which it was made up of...
Q: For the reaction below, Kc = 1.10 x 10-8. What is the equilibrium concentration of OH if the reactio...
A: Given:
Q: Lab Question #13: Observed rotation varies when either concentration or path length is varied. What ...
A: A question based on polarimetry, which is to be accomplished.
Q: Draw the Newman projection that corresponds to the following species. CH3 CI Br SH
A: The given diagram is a line diagram (it contains total three types of bond that are shown with wedge...
Q: 1.0 mol of an ideal gas with CV,m=3R/2 are transformed from an initial state T= 685K and P= 1.40bar ...
A: "Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for y...
Q: Name the following Mo(CN)4 Mo3(PO4)4 Mo2(HPO4)3 Mo(H2PO4)3 a. molybdenum phosphate b. molybdenum (IV...
A:
Q: Consider an ice cube of 300 grams at an initial temperature of -40 °C that is placed in a thermos of...
A:
Q: mól or ideal gas 02 are transformed fom an initial state 685K and P= 1.40bar to a final state T=273K...
A: The change in internal energy, ∆U = nCv∆T = 1*(3/2)*8.314*(685-273) = 5138.051 Joules The change in ...
Q: Outline Rutherford’s Gold foil experiment, including the observations and conclusions from his exper...
A: The arrangement of electrons, protons and neutrons inside the atoms is explained by different atomic...
Q: Which one of the following statements is true? Group of answer choices The enthalpy change for a rea...
A:
Q: Consider an ice cube of 300 grams at an initial temperature of -40 °C that is placed in a thermos of...
A:
Q: How many grams of boric acid should be used in compounding the following prescription? Report o the ...
A:
Q: Provide a systematic name for the following compound, including stereochemistry where necessary (don...
A: Write IUPAC NAME of the given structure---
Q: 9. Glucose is a molecular compounc
A: We are given Statement and we have to tell whether they are true or false
Q: Which of the following compounds is a major product of the reaction sequence below? OH (1) Mg, ether...
A:
Follow the curved arrows and draw the products of the following reaction. Include all lone pairs and charges as appropriate. Draw
Step by step
Solved in 2 steps with 1 images


