Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter20: Environmental Chemistry-earth's Environment, Energy, And Sustainability
Section: Chapter Questions
Problem 41PS
Related questions
Question
N2(g)+3h2(g)=2nh3(g) what type of reaction is this?
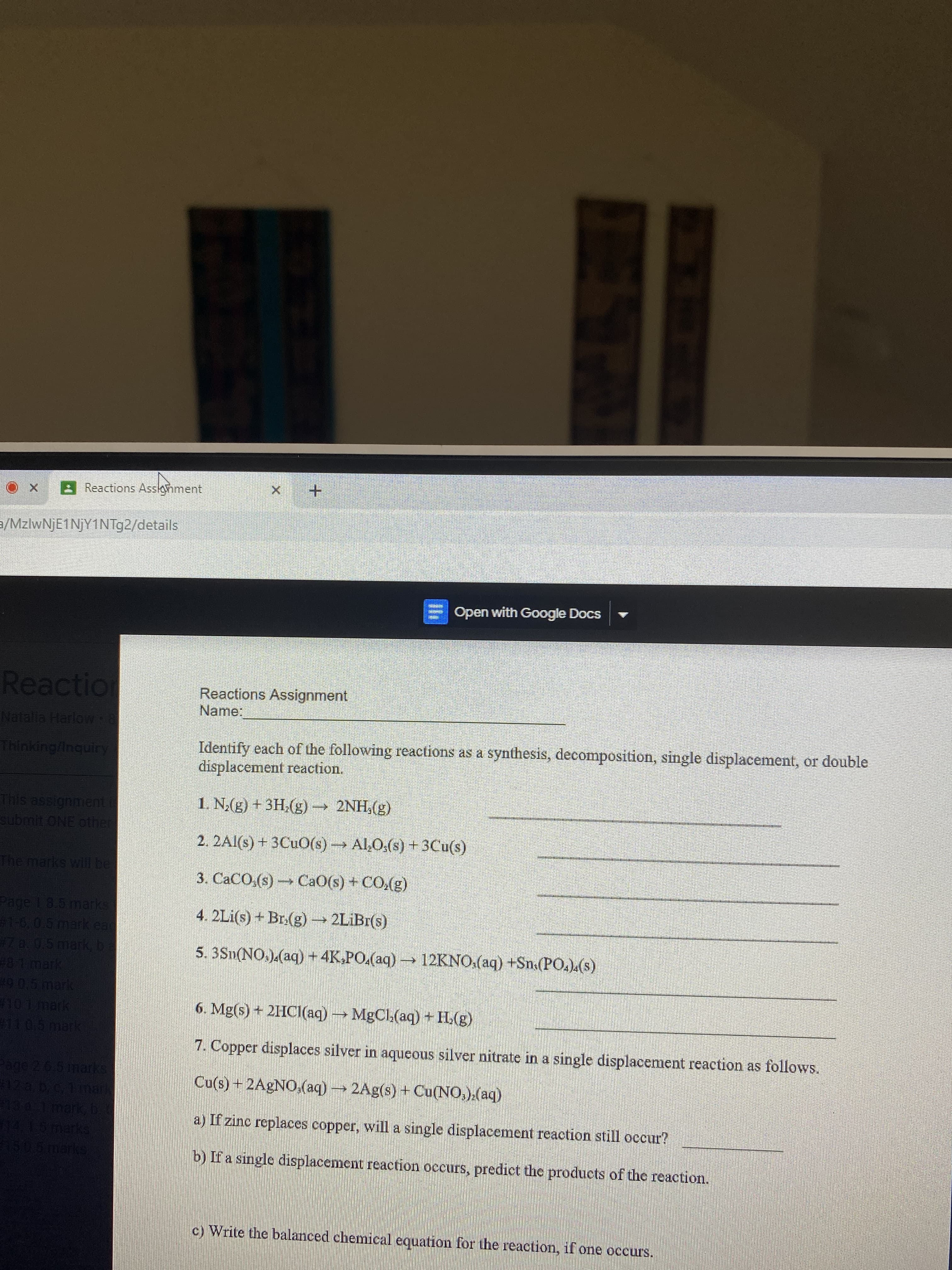
Transcribed Image Text:Reactions Assonment
+ x
a/MzlwNjE1NjY1NTg2/details
E Open with Google Docs
Reaction
Reactions Assignment
Name:
Natalia HarloW 8
Identify each of the following reactions as a synthesis, decomposition, single displacement, or double
displacement reaction.
Thinking/inquirry
(3)'HN7
2. 2Al(s) + 3CUO(s)→ Al,O:(s) + 3Cu(s)
This assignment i
1. N;(g) + 3H,(g) –→ 2NH,(g)
submit ONE other
The marks will be
(3)'0) + (s)OB) (s)'0
Page 1 8.5 marks:
4. 2Li(s) + Br.(g) → 2LİBI(s)
#1-6,0.5mark ead
47a.0.5mark, ba
481mark
5. 3Sn(NO.).(aq) + 4K.PO.(aq) → 12KNO.(aq) +Sn.(PO.).(s)
29.0.5mark
101 mark
6. Mg(s) + 2HCI(aq)MgCl.(aq) + H.(g)
(be) 1D3W+
7. Copper displaces silver in aqueous silver nitrate in a single displacement reaction as follows.
age 2 6.5 marks
12 a b.c, 1ar
134 1mark b
415marks
L506marks
Cu(s) + 2AGNO,(aq) 2Ag(s) + Cu(NO,).(aq)
a) If zinc replaces copper, will a single displacement reaction still occur?
b) If a single displacement reaction occurs, predict the products of the reaction.
c) Write the balanced chemical equation for the reaction, if one occurs.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
