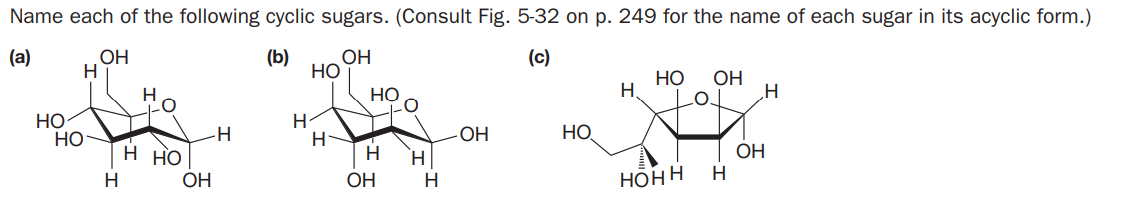
Name each of the following cyclic sugars. (Consult Fig. 5-32 on p. 249 for the name of each sugar in its acyclic form.) (a) OH H (b) OH (c) Но НО OH Но Но Но H HỌ HO- H. -H H ОН н но OH H OH H Нонн
Q: Provide the correct product(s) for the following reaction
A: This reaction is a cross aldol reaction.
Q: When the H dissociate from hydroxyl, the rest with different structures who can maintain the…
A: Alkyl groups have a tendency to donate electrons due to +I effect.
Q: Add curved arrows to draw the first part of the mechanism: tautomerization of a ketone to an enol.…
A: Keto-enol is a type of structural isomerism which involves a keto form (containing C=O) with alpha…
Q: For each organic compound in the table below, enter the locant of the highlighted side chain.
A: The parent chain of the molecule is selected in such a way that the number of carbon in it is…
Q: Name the PRODUCT compound and give it's molecular weight Q. Which is a better nucleophile methoxide…
A: Nucleophile takes a negative charge or lone pair of electrons on itself and attacks on a positively…
Q: If possible please answer all questions. Provide a complete, detailed curved-arrow mechanism for the…
A: Applying concept of Aldol Condensation reaction .
Q: Rank the following species from best nucleophile to poorest nucleophile in an aqueous solution (in…
A: Given that : We have to rank the following species form the best to the poorest nucleophile in…
Q: Draw the mechanism for the conversion of propanedial (see Problem 7.43) in its keto form to its enol…
A: The given compound is propanedial, and it is in keto form as shown below:
Q: (a) Indicate the following alkenes in order of increasing stability. Justify your answers.
A:
Q: KOCCH A Br в KOC(CH,), NaNH, CHI D (2 equiv) DMSO
A:
Q: Using curved arrow formalism, give the mechanism for the following reaction. Be sure to include…
A: Given,
Q: 2. Above is S-adenosylmethionine (SAM). Note that R'-S-R is a very good leaving group. You do not…
A:
Q: (c) Nitrobenzene will undergo electrophilic aromatic substitution at meta position, but not at the…
A:
Q: A в ctrophilic carbon in the structure below a
A:
Q: . Predict the products of the following acid-base reactions, and draw a curved arrow mechanism…
A: # Bronsted Acid- The species that can donate H+ ion. # Bronsted…
Q: Please be certain to use good curved arrow notation for your mechanism. HYH2O HO,
A:
Q: A. Arrange the following radicals in order of decreasing rate of bromination. Justify your answer.
A:
Q: Draw out the complete mechanism (curved arrow formalism) for the preparation of benz-alacetophene
A: Benzalacetophenone is also referred to as benzylideneacetophenone , Dibenzalacetone or Chalcone. It…
Q: N. H. c) Carry out the following conversion and provide mechanism. NO2 NO2 i) N. COCH3 .COOH ii) N.…
A:
Q: mechanism for the of hydrolysis (1-bromo of Propose a reaction methylpropyl)cyclohexane. Including…
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in three…
Q: (d) Carry out the following conversions and provide the mechanism : CH3 HO H3C CH3 -CH3 from H3C H3C…
A: alpha keto carbanions and p-benzoquinoes react to give the product
Q: In organic chemistry, an elimination process involves the removal of the hydrogen and a halide (i.e.…
A:
Q: H3O* single enantiomer
A: In an acidic medium, the epoxide is protonated and undergo a nucleophilic substitution reaction.…
Q: Provide efficient syntheses for the following transfo a) b) c)
A: Note- The given question is a multipart question, hence I solved first three questions according to…
Q: Compound E PBr3 Compound A H₂SO4 Heat Compound B (Major) KMnO₂/ H Heat 2-butanone OH + HCN OH…
A: Given reaction cycle is : Give the missing products and regents and conditions = ?
Q: ек ного сап ше benzene но synthesize this molecule from N
A: Given is organic compound.
Q: dho
A: The reactivity of alkene depends on the stability of alkene. Less stable alkenes will be more…
Q: Which of the following statement(s) is (are) TRUE? a. Addition of halogens to alkenes produces a…
A: The addition of halogen in presence of water gives rise to the formation of halohydrin. In this…
Q: Draw all constitutional isomers formed in attached E2 reaction, and predictthe major product using…
A: The E2 elimination reaction involves the removal of halide and neighboring β hydrogen atom in the…
Q: 1. Explain the the following,giving examples and illustrations to support your answer Electrophilic…
A:
Q: Show the complete and detailed reaction mechanism (cycloaddition) of heterocyclic thioamides with…
A:
Q: 1) Which one more reactive to word nucleophilic substitution P- nitro toluene or O- ethyl toluene ?…
A: P-nitro toulene will be more reactive towards nucleophilic substitution than O-ethyl toulene because…
Q: Et3N N. NH2 ZI O: O:
A:
Q: Complete the mechanism of hydration of alkene below. Please follow curved arrows to predict the…
A: Answer:- The hydration of alkene gives the corresponding alcohol according to Markovnikov rule which…
Q: Write a plausible mechanism for this transformation. Only two sequential pericyclic reactions are…
A: In organic chemistry, pericyclic reactions are those in which the shifting of double bonds or…
Q: Which would you think would be a strongerinteraction and why: an interaction between a sodium ion…
A: Given information: an interaction between a sodium ion and the partial negative charge on the oxygen…
Q: Outline all steps in a synthesis of propyne from each of the following: a) CH,COCH; b) CH;CH,CHB",…
A:
Q: which is the most and least nucleophilicc, Arrange in the order of increasing nucleophlicity and…
A: The compounds given are,
Q: Predict the major, organic product for the following reaction sequence. Assume cis conformation of…
A:
Q: what is the plausible synthesis?
A:
Q: n base-catalyzed halogenation of acetone, the second (and third) on the same carbon How can we…
A:
Q: Question attached
A: Given reactions:
Q: 1) NaH 2) 3) H*, heat
A: NaH (sodium hydride) is a strong base. It abstracts a proton from more acidic hydrogen. The…
Q: 1) NaOH H 2) H*, heat
A:
Q: Q2 Which compound liberate the lowest heat upon hydrogenation with excess H,? Why? C В A products)
A: Alkenes can be defined as the compound containing double bonds between carbon atoms in that…
Q: The image below shows a permitted cycloaddition reaction.?
A: For the permitted cycloaddition there should be supra-supra overlap between the terminal lobes of…
Q: Which of compounds A-C would you expect to have the greatest heat of combustion? The smallest?…
A: Heat of combustion of the substance can be defined as the energy value or calorific value is the…
Q: Study the curved arrows in the reaction and provide the line structures of all the intermediates and…
A:
Step by step
Solved in 4 steps with 6 images

- Please provide a complete, detailed curved-arrow mechanism for the following reaction.Include ALL lone pairs and formal charges. Using the mechanism and a few words,explain the why the NaH deprotonates at the selected a-position and not the other a-position. Also explain why the indicated alkene is formed in the 3rd step rather than the other possible alkene product.Identify the pericyclic reactions in the following reaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.Complete the following reactions, clearly indicating regio-and stereochemistry where applicable. In cases, where ortho-and para-products are formed, draw both.
- Please provide a complete, detailed curved-arrow mechanism for the following reaction. Include ALL lone pairs and formal charges. Using the mechanism and a few words, explain why the NaOH deprotonates at the a-position and not the b-position or the aldehyde hydrogen. Also, explain why the indicated alkene is formed in the 2nd second rather than the other possible alkene product.Provide a curved-arrow mechanism for the following reaction. In a few words, explain the regiochemistry of the OH in the product (is this conjugate or direct addition, and what causes the reaction to proceed via that addition mechanism).(Please give clear handwritten answer) Provide an arrow pushing mechanism for the following transformation. Classify all pericyclic reactions.
- Give only typing answer with explanation and conclusion What are the correct reaction conditions for the following transformation?Identify the pericyclic reactions in the followingreaction schemes. Give the complete reaction name and indicate the course of the reaction with the aid of the arrow notation.which is the most and least nucleophilicc, Arrange in the order of increasing nucleophlicity and provide reasons why
- Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE. The stereochemistry of the following alkenes are?Provide the complete arrow pushing mechanism and provide the product for the reactions. Be sure to consider stereochemistry in your product(s). Give a short concise statement about the step that determines the stereoselectivity, or lack of, for each reaction. Also give a statementProvide illustration/s and explanation of the difference between the Markovnikov and Anti-Markovnikov additions of HBr to alkene.

