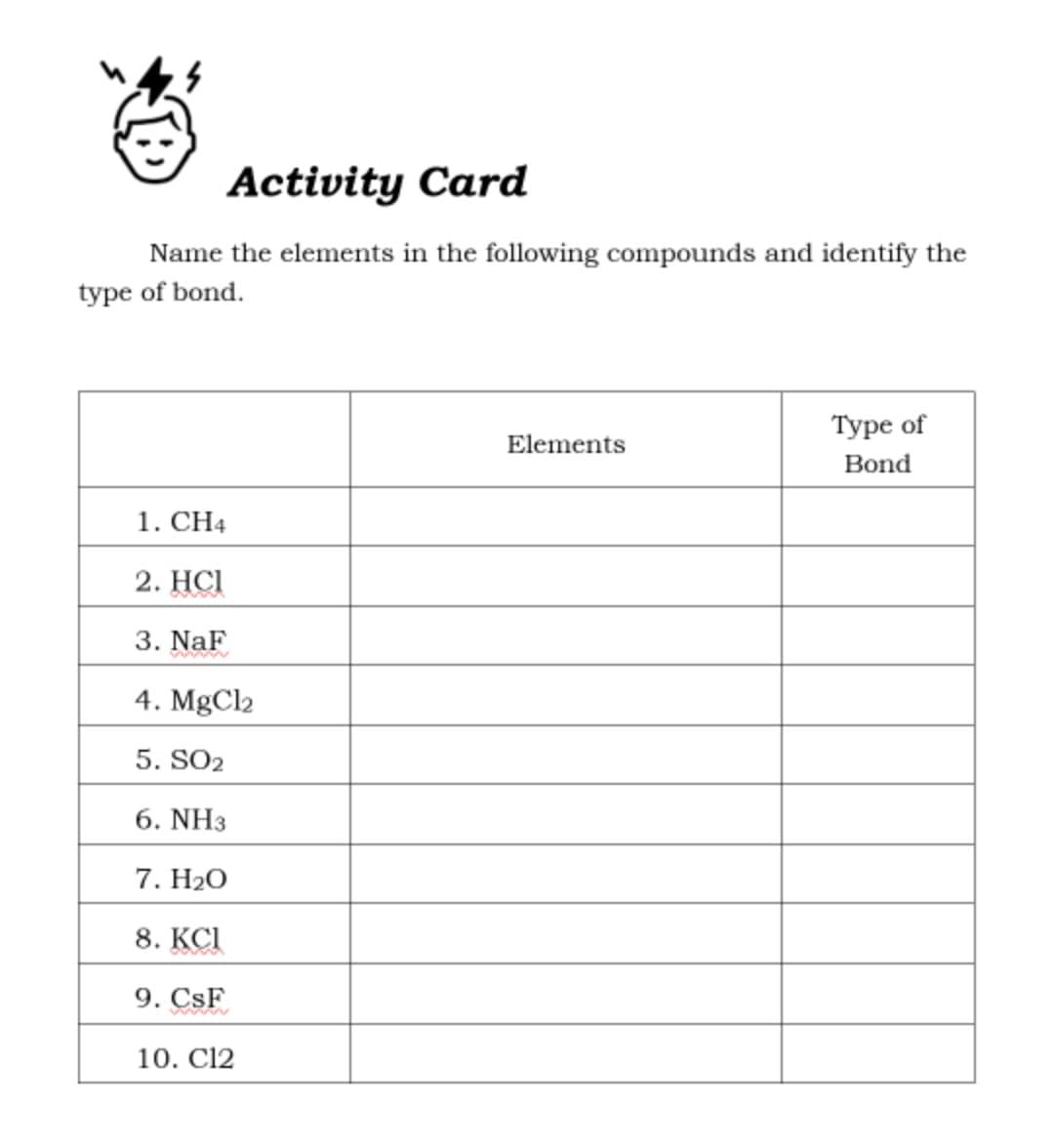
Name the elements in the following compounds and identify the type of bond. Туре of Elements Bond 1. CH4 2. HСI 3. NaF 4. MgCl2 5. SO2 6. NH3 7. Н2О 8. KCI 9. CsF 10. C12
Q: The element sulphur has atomic number 16. Sulphur forms a molecular compound with chloride with a…
A: Since you have asked questions with multiple sub parts, as per our guidelines,we are supposed to…
Q: The Lewis structure for which of the following molecules would have two non-bonded pairs of…
A:
Q: How many resonance structures can be drawn for the molecule 03? Select one: a. 3 b. C. d. 4 e. LO 2.
A: We will see number of how many possible resonating structure of O3. : The number of valence…
Q: 8. Which of the following is a nonpolar covalent molecule? C. HCI D. CC14 A. NH3 В. Н20
A: Ionic compounds are formed by the transfer of electrons from cations to anions. In the ionic…
Q: Which of the following is technically considered a nonpolar covalent bond? A. C – H B. O – N C. P –…
A: we can decide whether the bond is polar or nonpolar on the basis of their electronegativity. If the…
Q: Which of the following has the bonds correctly arranged in order of increasing polarity? Be—F,…
A: Polarity: The polarity of a bond is depends upon its electronegativity difference between the bonded…
Q: molecules. Use the step-by-step guidelines. a.) CH3Cl b.) CH3CH2Br c.) H2O d.) SO2
A:
Q: When a molecule can form two different structures, the structure with the stronger bonds is usually…
A: The bond energies are 1) N-H bond energy = 391 KJ/mol 2) N-O bond energy = 201 KJ/mol 3) O-H bond…
Q: Draw the Lewis structure(s) for the following substances. Determine the molecular shape and polarity…
A: Using the VSEPR theory and lewis structure theory shape and polarity of given molecule is…
Q: Look at the image below. Ne C ΟΝ D N Based on the distribution of electrons, which would you expect…
A:
Q: Which of the following pairs of elements is/are likely to form an ionic bond? Choose one or more: A.…
A: ->Ionic bond formed between metal and non-metal .When metal donate electron to the non metal then…
Q: Which of the following is most generally likely to happen when two atoms form a chemical bond?…
A:
Q: Determine the number and type of chemical bonds in silicon tetrachloride (SiCl4) and beryllium…
A: One formula unit of silicon tetra chloride has polar covalent bond. One formula unit of Beryllium…
Q: The Lewis structure for which of the following molecules would have only one non-bonded pair of…
A:
Q: H H2 Hydrogen atom Hydrogen atom Hydrogen molecule Figure 2.5 A hydrogen molecule forms when two…
A: Hydrogen is a colorless, odorless, tasteless, flammable gaseous substance that is the simplest…
Q: For the carbon atom Lewis dot symbol structure, how many valance electrons are unpaired?
A:
Q: Draw the Lewis structure of each molecule or ion and place the following in order of increasing…
A: BrCl3 Total valence electron = 7+7(3) =28 Central atom = Br Central atom Bromine has 3 bond pairs…
Q: what is the bond distance that gives the minimum energy ? What is the bond angle that gives the…
A: Hello. There are multiple subparts in the question. The first three subparts have been solved. To…
Q: Determine the type of bonds in the following: ionic, nonpolar, polar: a) NaCl b) Bel2 c) HF d)…
A:
Q: Calculate and determine if the compounds listed below or non-polar, polar or ionic compounds: 1.…
A: Ans
Q: H2CCH2O(1).jpg Consider the following molecule: How many sigma bonds in this molecule? See attached…
A: Sigma bond- Generally, single bonds between atoms are always sigma bonds. A single bond such as…
Q: Molecule BCl3 has --- number of lone pairs of electrons on the central atom. Question 3 options:…
A:
Q: What does a double bond consist of? none of the other choices is correct. b. Two sigma bonds c. Two…
A: Answer are as follows:
Q: Which of the following molecules contain polar covalent bonds ONLY? I. CH4 II. KNO3 III. H2O a.…
A: POLAR BONDS The polar bonds are the bonds between the two atoms in which the atoms bonded to have an…
Q: CHEMICAL BONDING A. Write the ionic-bond structure of the following compounds. Bal2 Ca3P2 SrSe B.…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which element will have 5 electrons in its Lewis dot symbol? a. Argon b. Boron c. Carbon d.…
A: NOTE-As the question contains multiple parts, we will solve the first three subparts for you. To get…
Q: Which pair of atoms forms the most polar bond?a) N and O b) C and O c) C and F d) N and F
A: A bond is polar because of the high electronegativity difference between the atoms. If the…
Q: Which element will have 5 electrons in its Lewis dot symbol? a. Argon b. Boron c. Carbon d.…
A: Lewis dot symbol:- It represents the number and arrangement of electrons for bond formation between…
Q: What noble gas has the same electron configuration as each of the ions in the following compounds?…
A: Periodic table represents the arrangement of the elements in increasing order of their atomic…
Q: 2JoM 9mulo Formula: BCI3 Name: Bunum Trichlornde Lewis Dot(Symbol of B atom Lewis Dot Symbol of…
A: Given table is : Complete the table = ?
Q: Show how the molecules satisfy the octet rule ff.the given example.1.CO2 2.PCI3 3.CCI4 4.NF3 5.SIH4…
A: The octet rule states that the atoms in a compound bonded through a covalent bond must complete…
Q: which contain only polar covalent bonds?
A:
Q: Which of the following contains the most ionic bond? BrI Cl2 CsF NO S8
A:
Q: What is the electron-pair geometry for Al in AlCl3? _______ There are ______ lone pair(s) around…
A: We have to tell the electron pair geometry of given compounds
Q: Consider selenium oxychloride (SEOCI2): What is the elemental symbol of its central atom? How many…
A: Given molecule is : SeOCl2 a). What is the elemental symbol of its central atom = ? b). How many…
Q: many valence electrons does the molecule have? How many lone pairs of electrons are on the central…
A: Atomic number of sulphur is 16 . Configuration is 2,8,6 It belongs to 16 th group.
Q: For which of the following we CAN NOT draw a stable Lewis structure ? NCI5 All of the above have…
A:
Q: Which of these substances contain both covalent and ionic bond? H2S CH4 NH4CI Н202
A: Covalent bond is formed by sharing of electrons where ionic bonds are formed by transfer of…
Q: Write the electron dot structure for ethylene, C2H4.
A: Carbon have 4 valence electrons.
Q: For eac V pound in the table below, decide whether there would be any hydrogen-bonding force between…
A: Hydrogen bonding is formed when highly electronegative atoms like N, O, and F pull the electron…
Q: On a piece of paper, complete the following table: Lewis structure Molecular Geometry Is the…
A: Lewis structure is the representation of electrons (bond and lone pair) arranged around the…
Q: For the bonds in these molecules: LiCl H2 K2O CO2 BCl3 Which has the most…
A:
Q: Below is the Lewis structure of the hydrazine (N,H,) molecule. H:N :: N : H Count the number of…
A: Lewis structures, also regarded as Lewis dot formulas, Lewis dot structures, electron dot…
Q: 14b
A: Given shape is see-saw structure.
Q: Question attached
A: A covalent bond is formed by sharing of electron between two atom. However, the ionic bond is formed…
Q: Valence electrons are the electrons in the (1) occupied electron shell, which participate in the…
A: Valence electrons are defined as the electrons, that resides in the outermost shell of the atom. An…
Q: What is the total number of valence electrons in the following strcuture? H C-H H О 16 О 32 О 2
A:
Q: Give the Lewis Dot Structures, then draw the structures with the correct molecular geometry and give…
A: In this question, we have to find out the correct answer of given problem by the help of Lewis dot…
Step by step
Solved in 2 steps with 2 images

- I need help ASAP Website for first simulation: https://www.learner.org/wp-content/interactive/periodic/bondin The second/last simulation: Open the following website: https://javalab.org/en/ion_model_en/ As seen in the table, compounds containing an integer ratio of elements depend on how many cations combine with how many anions to form a stable compound. For example, in table 1, to form a NaCl compound, first Na ionizes from the Na+ cation which is having +1 positive charge, which will lose 1 electron, it is having +1 charge, this electron then goes to Cl and it will change to Cl- anion by gaining the electron, so here 1:1 ratio charge which means 1 Na+ combines with 1 Cl-1 to form NaCl. so here integer ratio is 1:1 for this sodium chloride compound. As listed in table 2, Mg(OH)2, the integer ratio is 1:2 which means, Mg is a neutral atom that loses 2 electrons and forms an Mg+2 cation, which combines with OH which gains 1 electron to form OH- anion, So here Mg2+ can combine with 2…Using Lewis symbols and Lewis structures, diagram the formationof SiCl4 from Si and Cl atoms, showing valence-shellelectrons. (a) How many valence electrons does Si have initially?(b) How many valence electrons does each Cl haveinitially? (c) How many valence electrons surround the Siin the SiCl4 molecule? (d) How many valence electrons surroundeach Cl in the SiCl4 molecule? (e) How many bondingpairs of electrons are in the SiCl4 molecule?Use Lewis symbols and Lewis structures to diagram the formationof PF3 from P and F atoms, showing valence-shellelectrons. (a) How many valence electrons does P have initially?(b) How many valence electrons does each F haveinitially? (c) How many valence electrons surround the P inthe PF3 molecule? (d) How many valence electrons surroundeach F in the PF3 molecule? (e) How many bonding pairs ofelectrons are in the PF3 molecule?
- Draw Lewis structures for BrO4-, SiH4, PCl4, CH2Cl2 and BF4-.(a) Which species is/are likely to be paramagnetic?(b) What reaction(s) would occur for the paramagnetic species? (c) By VSEPR, suggest the molecular geometry of each species.Use the molar bond enthalpy data in the table to estimate the value of Δ?∘rxnΔHrxn° for the equation C2H4 (g) + HBr (g) --> C2H5Br (g) Average molar bond enthalpies. (?bond)(Hbond) Bond ??⋅???−1kJ⋅mol−1 Bond ??⋅???−1kJ⋅mol−1 O−HO−H 464464 C≡NC≡N 890890 O−OO−O 142142 N−HN−H 390390 C−OC−O 351351 N−NN−N 159159 O=OO=O 502502 N=NN=N 418418 C=OC=O 730730 N≡NN≡N 945945 C−CC−C 347347 F−FF−F 155155 C=CC=C 615615 Cl−ClCl−Cl 243243 C≡CC≡C 811811 Br−BrBr−Br 192192 C−HC−H 414414 H−HH−H 435435 C−FC−F 439439 H−FH−F 565565 C−ClC−Cl 331331 H−ClH−Cl 431431 C−BrC−Br 276276 H−BrH−Br 368368 C−NC−N 293293 H−SH−S 364364 C=NC=N 615615 S−SS−S 2252257.97 Consider the structure shown below for as well as any other important resonance structures. (a) What is the expected O—N—O bond angle in this structure? (b) The molecule contains N—O bonds of two different lengths. How many sborter N—O bonds would be present?
- 9.77 When a reaction is exothermic, is the sum of bond energies of products or of reactants greater?7.13 Figure 7-2 depicts the interactions of an ion with its first nearest neighbors, second nearest neighbors, and third nearest neighbors in a lattice, (a) Would the interactions with the fourth nearest neighbors be attractive or repulsive? (b) Based on Coulomb's law, how would the relative sizes of the terms compare if the potential energy were expressed as V=V1st+V2nd+V3rd+V4th ?Q3: (A) Answer the following questions: 1. Give an example of allotropy materials and explain it. 2.State three types of smart materials. 3. What is bonding of NaCl ? List its properties and sketch this bond. 4. Explain Pauli principle.
- Sulfur tetrafluoride (SF4) reacts slowly with O2 to form sulfurtetrafluoride monoxide (OSF4) according to the followingunbalanced reaction: SF4(g) + O2(g)---->OSF4(g)The O atom and the four F atoms in OSF4 are bonded to acentral S atom. (a) Balance the equation. (b) Write a Lewisstructure of OSF4 in which the formal charges of all atomsare zero. (c) Use average bond enthalpies (Table 8.3) to estimatethe enthalpy of the reaction. Is it endothermic or exothermic?(d) Determine the electron-domain geometry ofOSF4, and write two possible molecular geometries for themolecule based on this electron-domain geometry. (e) Foreach of the molecules you drew in part (d), state how manyfluorines are equatorial and how many are axial.Use average bond enthalpies (Table 8.3) to estimate ΔH forthe atomization of benzene, C6H6:C6H6(g)--->6 C(g) + 6 H(g)Compare the value to that obtained by using ΔHf° datagiven in Appendix C and Hess’s law. To what do you attributethe large discrepancy in the two values?Consider the following reaction:H2 (g) + O2 (g) --> H2O2 (g) (Delta)H = -153 kJGiven that the H2 bond energy is 432 kJ/mol, the O2 bond energy is 495 kJ/mol, and the OH bond energy is 467 kJ/mol, estimate the bond energy for the oxygen-oxygen single bond (in kJ/mol).



