Name: Total /18 Guacamole Assignment Amadeo Avogadro wants to make guacamole. He needs two avocados per serving of guacamole as well as: 1/2 teaspoon (tsp) of salt; 1.25 cloves of garlic; the juice of one lime; and cilantro to taste. Theoretical: how many avocados does Avogadro need and how many moles of guacamole can he make? Actual: Answer all questions on this sheet and record answers in the table. Include rough work on a separate sheet of paper. Final answers should be rounded to the correct number of significant figures and units should be included. A) Don't get salty [5 marks] 1. What is the chemical formula for table salt? 2. How many moles of salt will you be using in this recipe? 3. How many sodium atoms is that? B) Keep the vampires at bay [8 marks] Garlic's active ingredient is a chemical called alliin (C6H11NO3S). 1. What is the molar mass of alliin? 2. Assume alliin takes up 10.0% (by mass) of the garlic in the recipe. How many moles of alliin are you using? 3. How many atoms of Hydrogen is that?
Name: Total /18 Guacamole Assignment Amadeo Avogadro wants to make guacamole. He needs two avocados per serving of guacamole as well as: 1/2 teaspoon (tsp) of salt; 1.25 cloves of garlic; the juice of one lime; and cilantro to taste. Theoretical: how many avocados does Avogadro need and how many moles of guacamole can he make? Actual: Answer all questions on this sheet and record answers in the table. Include rough work on a separate sheet of paper. Final answers should be rounded to the correct number of significant figures and units should be included. A) Don't get salty [5 marks] 1. What is the chemical formula for table salt? 2. How many moles of salt will you be using in this recipe? 3. How many sodium atoms is that? B) Keep the vampires at bay [8 marks] Garlic's active ingredient is a chemical called alliin (C6H11NO3S). 1. What is the molar mass of alliin? 2. Assume alliin takes up 10.0% (by mass) of the garlic in the recipe. How many moles of alliin are you using? 3. How many atoms of Hydrogen is that?
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter8: Chemical Composition
Section: Chapter Questions
Problem 128CP: itamin B12 , cyancobalamin, is essential for human nutrition. Its molecular formula is...
Related questions
Question
I just need parts C and D. Thanks!
![Name:
Total
/18
Guacamole Assignment
Amadeo Avogadro wants to make guacamole. He needs two avocados per serving of guacamole as well as: 1/2
teaspoon (tsp) of salt; 1.25 cloves of garlic; the juice of one lime; and cilantro to taste.
Theoretical: how many avocados does Avogadro need and how many moles of guacamole can he make?
Actual: Answer all questions on this sheet and record answers in the table. Include rough work on a separate
sheet of paper. Final answers should be rounded to the correct number of significant figures and units should
be included.
A) Don't get salty [5 marks]
1. What is the chemical formula for table salt?
2. How many moles of salt will you be using in this recipe?
3. How many sodium atoms is that?
B) Keep the vampires at bay [8 marks]
Garlic's active ingredient is a chemical called alliin (C6H11NO3S).
1. What is the molar mass of alliin?
2. Assume alliin takes up 10.0% (by mass) of the garlic in the recipe. How many moles of alliin are you
using?
3. How many atoms of Hydrogen is that?
4. How much sulfur is that (in grams)?
C) This recipe is sublime [5 marks]
Citric acid (C6H8O7) is a chemical present in citrus fruits such as limes.
1. What is the molar mass of citric acid?
2. Assume citric acid makes up 7.96% (by volume) of lime juice. How many moles of citric acid are in the
recipe?
3. What is the concentration of citric acid in lime juice (in moles/L)?
D) Say it ain't cilantro [6 marks]
Vitamin K1 (phytomenadione) is a vitamin present in cilantro that helps with blood clotting.
1. Research to find the chemical formula for Vitamin K1
2. What is the molar mass of Vitamin K,?
3. Assume Vitamin K1 makes up 0.052% (by mass) of cilantro, based off your cilantro taste in the table
below how much Vitamin Ki is there in your guacamole? (indicate your choice in the answer table)
4. How many molecules of Vitamin K1 are there in your guacamole?
How much cilantro do you like?
1/8 tsp
1 tsp
1 tbsp
A wee bit
Not my first rodeo
Gimme dat 'tro
I will not rest until I have become cilantro in
body and soul
¼ cup](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F6348aaff-33c3-4518-864b-f2bb6ae8d72c%2F43849cc9-1601-46c6-8117-79d9c8554eb0%2Fypm9ilq_processed.jpeg&w=3840&q=75)
Transcribed Image Text:Name:
Total
/18
Guacamole Assignment
Amadeo Avogadro wants to make guacamole. He needs two avocados per serving of guacamole as well as: 1/2
teaspoon (tsp) of salt; 1.25 cloves of garlic; the juice of one lime; and cilantro to taste.
Theoretical: how many avocados does Avogadro need and how many moles of guacamole can he make?
Actual: Answer all questions on this sheet and record answers in the table. Include rough work on a separate
sheet of paper. Final answers should be rounded to the correct number of significant figures and units should
be included.
A) Don't get salty [5 marks]
1. What is the chemical formula for table salt?
2. How many moles of salt will you be using in this recipe?
3. How many sodium atoms is that?
B) Keep the vampires at bay [8 marks]
Garlic's active ingredient is a chemical called alliin (C6H11NO3S).
1. What is the molar mass of alliin?
2. Assume alliin takes up 10.0% (by mass) of the garlic in the recipe. How many moles of alliin are you
using?
3. How many atoms of Hydrogen is that?
4. How much sulfur is that (in grams)?
C) This recipe is sublime [5 marks]
Citric acid (C6H8O7) is a chemical present in citrus fruits such as limes.
1. What is the molar mass of citric acid?
2. Assume citric acid makes up 7.96% (by volume) of lime juice. How many moles of citric acid are in the
recipe?
3. What is the concentration of citric acid in lime juice (in moles/L)?
D) Say it ain't cilantro [6 marks]
Vitamin K1 (phytomenadione) is a vitamin present in cilantro that helps with blood clotting.
1. Research to find the chemical formula for Vitamin K1
2. What is the molar mass of Vitamin K,?
3. Assume Vitamin K1 makes up 0.052% (by mass) of cilantro, based off your cilantro taste in the table
below how much Vitamin Ki is there in your guacamole? (indicate your choice in the answer table)
4. How many molecules of Vitamin K1 are there in your guacamole?
How much cilantro do you like?
1/8 tsp
1 tsp
1 tbsp
A wee bit
Not my first rodeo
Gimme dat 'tro
I will not rest until I have become cilantro in
body and soul
¼ cup
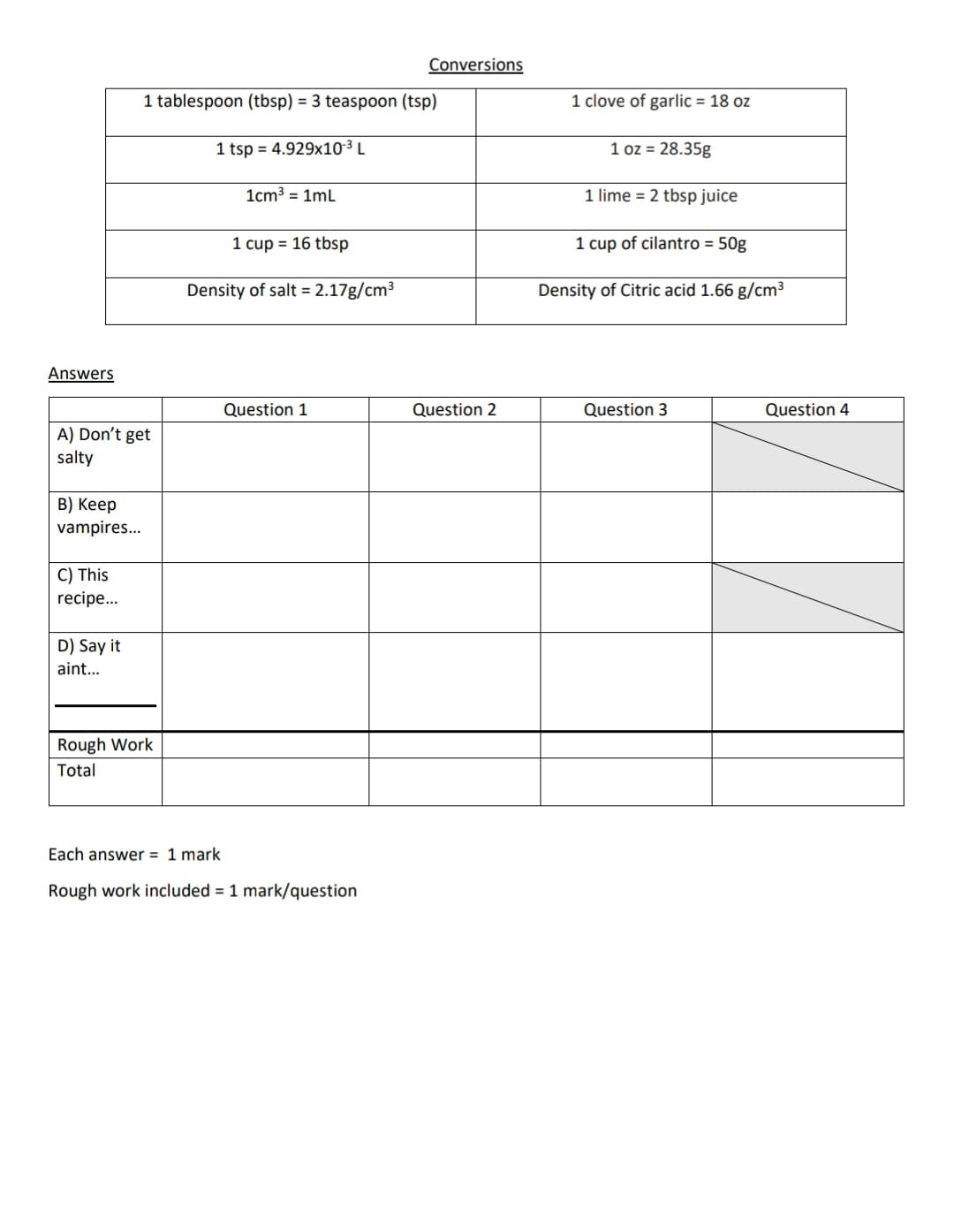
Transcribed Image Text:Conversions
1 tablespoon (tbsp) = 3 teaspoon (tsp)
1 clove of garlic = 18 oz
1 tsp = 4.929x10³ L
1 oz = 28.35g
1cm3 = 1mL
1 lime = 2 tbsp juice
1 cup = 16 tbsp
1 cup of cilantro = 50g
Density of salt = 2.17g/cm3
Density of Citric acid 1.66 g/cm3
Answers
Question
Question 2
Question 3
Question 4
A) Don't get
salty
В) Кeep
vampires..
C) This
recipe...
D) Say it
aint...
Rough Work
Total
Each answer = 1 mark
Rough work included = 1 mark/question
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning