Q: What component of the buffer solution reacted with the addition of the strong acid, HCl? Watter komp...
A: Buffer is a solution that resists the change in pH when small amount of acid or base is added to it....
Q: Formulate a hypothesis that can be made concerning the isolation of casein from milk and testing for...
A: Casein is a protein present in milk.
Q: Consider an ice cube of 300 grams at an initial temperature of -40 °C that is placed in a thermos of...
A: Heat lost by water=Heat gained by ice -Mw ×Cw× (T2-T1)= Mice × Cice ×(T2-T1) T1 ice=-41°C=273-41=232...
Q: Write the balanced complete ionic equation for the reaction when aqueous Cs3PO4 and aqueous AgNO3are...
A: Given, Aqueous solution of Cs3PO4 and AgNO3 are mixed.
Q: Below is the graph for the melting point of the period 6 elements versus atomic number. Determine th...
A:
Q: Why does volume increase when pressure and temperature remain constant? What has happened to the gas...
A: Here we are aksed why does volume increase when pressure and temperature remain constant and what h...
Q: H H H H H H H H H H. H. I or H II H H H. H
A: Torsional strain is the repulsion that arises between atoms or group of atoms when a molecule is rot...
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of potassium hydroxid...
A:
Q: please write the formal charge of nitrogen trioxide on each atom and the total charge,,
A:
Q: mól or ideal gas 02 are transformed fom an initial state 685K and P= 1.40bar to a final state T=273K...
A: The change in internal energy, ∆U = nCv∆T = 1*(3/2)*8.314*(685-273) = 5138.051 Joules The change in ...
Q: For the following electron-transfer reaction: Cd(s) + Cu2+(aq) Cd2+(aq) + Cu(s) The oxidation hal...
A: Given Reaction Cd(s) + Cu2+(aq) → Cd2+(aq) + Cu(s) Oxidation half reaction : = ? Reductio...
Q: Give 6 ions that are isoelectronic with neon . Place these ions innorder of increase size
A:
Q: 1.0 mol of an ideal gas with CV,m=3R/ P= 1.40bar to a final state T=273K and A)Calculate AU for this...
A:
Q: tranegoom Jollawing (TEpine phatoridakan) The to 0.
A: Photo oxidation is a chain process incorporating a large number of chemical reaction. In this reacti...
Q: . The Law of Conservation of Mass proves that A. the number of atoms in a chemical reaction does no...
A: Given: Law of conservation of mass.
Q: 4. Which among the shells given below is closest to the nucleus? a. М с. L b. K d. O 5. The d subshe...
A: Answers of 4,5,6
Q: What gas forms when you add Cu(NO3)2(aq) and NaCO3(aq) together
A: Since both Cu(NO3)2(aq) and Na2CO3(aq) are salts, they undergo double displacement reaction to form ...
Q: B. Identify the following set of comp in provided blank, After which, draw the correct Lewis dot str...
A:
Q: Arrange the following molecules according to decreasing density. 1 CH3 2 CH3 3 CH3 H2N- H3C- HO CH3 ...
A: Given Structure = 2 Amino propane , 2 methyl propane , 2 Propanol Decreasing order of molecul...
Q: 1) Identify each of the following half-reactions as either an oxidation half-reaction or a reduction...
A:
Q: Include the 'H-NMR and 1³C-NMR data for the product in separate tables with a labeled diagram 'H: ch...
A: 1H-NMR and 13C-NMR are the spectroscopic techniques used in the laboratories for the identification ...
Q: Which conformation of cyclohexane has the greater steric strain?
A:
Q: 9. Glucose is a molecular compounc
A: We are given Statement and we have to tell whether they are true or false
Q: This is the electron configuration in which the octet rule is based on: A. np3 nd5 B. ns2 np3 nd3...
A: According to octet rule electron transfer or electron sharing proceeds until an atom has acquired an...
Q: Directions: Perform the following mole conversions. Show your detailed solutions 26 mol PCl5 (phosp...
A: The number of moles of a compound can be calculated by the following formula: Number of moles a comp...
Q: 2. __H2 + __O2 --> __H2O, What are the coefficients to be used to balance the equation? A. 2,2,2 B...
A:
Q: Consider the following cyclohexane derivative: *Figure is the screenshot* a. Draw a chair conforma...
A:
Q: Consider the following compound and decide if it is chiral or achiral. Show your work by determining...
A: Chiral compounds:- a compound that contains an asymmetric center, and mirror image is non-superimpos...
Q: of elementary step based on the mechanistic
A: It is Tertiary Alkyl halide. So, it follows Sn1 mechanism. And it is two step mechanism.
Q: please write the formal charge of nitrogen trioxide on each atom and the total charge,,,,,
A:
Q: How many grams of boric acid should be used in compounding the following prescription? Report to the...
A: Ans (0.5/100) ×60= 0.3 g of Proparacaine hydrochloride (2.5/100) ×60= 1.5 g of pilocarpine hydr...
Q: Consider an ice cube of 300 grams at an initial temperature of -40 °C that is placed in a thermos of...
A:
Q: student is asked to standardize a solution of barium hydroxide. He weighs out 0.952 g potassium hydr...
A: Given that: Mass of potassium hydrogen phthalate(KHP) = 0.952g Molar mass of KHP = 204.2 g/mol Mol...
Q: Which is true about the chemical equation for photosynthesis? A.carbon dioxide is produced B.oxyge...
A:
Q: 1.) Liquid water molecules escaping from the surface enter into the Blank. What is the blank?
A: Liquid water molecules escaping from the surface enter into the ......
Q: Gaseous ethane (CH;CH,) reacts with gaseous oxygen gas (O, to produce gaseous carbon dioxide (Co,) a...
A:
Q: It requires 35.7 mL of calcium hydroxide to reach the endpoint. A. What is the molarity of the calc...
A:
Q: Potassium perchlorate is used in the production of fireworks and explosives. It is made in a two-ste...
A: The number of moles of a substance is calculated by using the formula moles = mass / molar mass
Q: When a solution of ammonium chloride is added to a solution of lead(II) nitrate, Pb(NO3)2, a white p...
A:
Q: These phenomena can be strong enough to reshape the orbitals of the atoms due to repulsion between b...
A: C. Orbital Hybridisation
Q: For the neutralization reaction between methylamine and propanoic acid, draw curved arrows to indica...
A: In acid base reaction acid loose its proton and base accepts proton. Acid base reaction is also know...
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients ...
A: We have to balance given reaction and answer the questions based on reaction.
Q: Order these bonds in length and strength according to the periodic table trends (do not look up the ...
A: Higher the bond energy associated with a specific atom pair, stronger the bond is and smaller will b...
Q: NEED ASAPP with complete solution 1. Nitrogen at a temperature of 310°F occupies a volume of 10....
A:
Q: Given the data below, H°rxn for the reaction 2502 (g) + O2 (g) → 2SO3 (g) is kJ. AH°; (SO2 (g)) = -2...
A: Given Reaction 2SO2(g) + O2(g) → 2SO3(g) ∆H°f ( SO3(g) ) = - 396 KJ/mol ∆H°f ( SO2(g) ) ...
Q: Draw the following structure. 4-sec-Butyl-2-chlorononane cis-1-Bromo-2-ethylcyclopentane
A: Given Name of Compound = 4 - Sec- butyl 2 Chloro nonane Structure = ? Name of compound = Cis - 1 - ...
Q: trans-1,4-dimethylcyclohexane has two conformations with a difference in energy of 3.6 kcal/mol. Wha...
A:
Q: Find the Empirical Formula for the Following Compounds CH6 Ethane C5H12 Pentane CH4 Methane C2H4 Eth...
A: Find the empirical formula of the given compound ----
Q: Determine the % of acetic acid (MM = 60.05 g/mol) in a 2.00 mL vinegar sample that has a mass of 1.9...
A: Given: Volume of acetic acid solution = 2.00 mL> Molar mass of acetic acid = 60.05 g/mol. Mass of...
Q: Which of the species listed has a Lewis structure with a resonance structure? CO, H2S, SF6 , N2O A....
A: Which of the species listed has a Lewis structure with a resonance structure? CO, H2S, SF6 , N2O
2c.
Step by step
Solved in 2 steps with 2 images

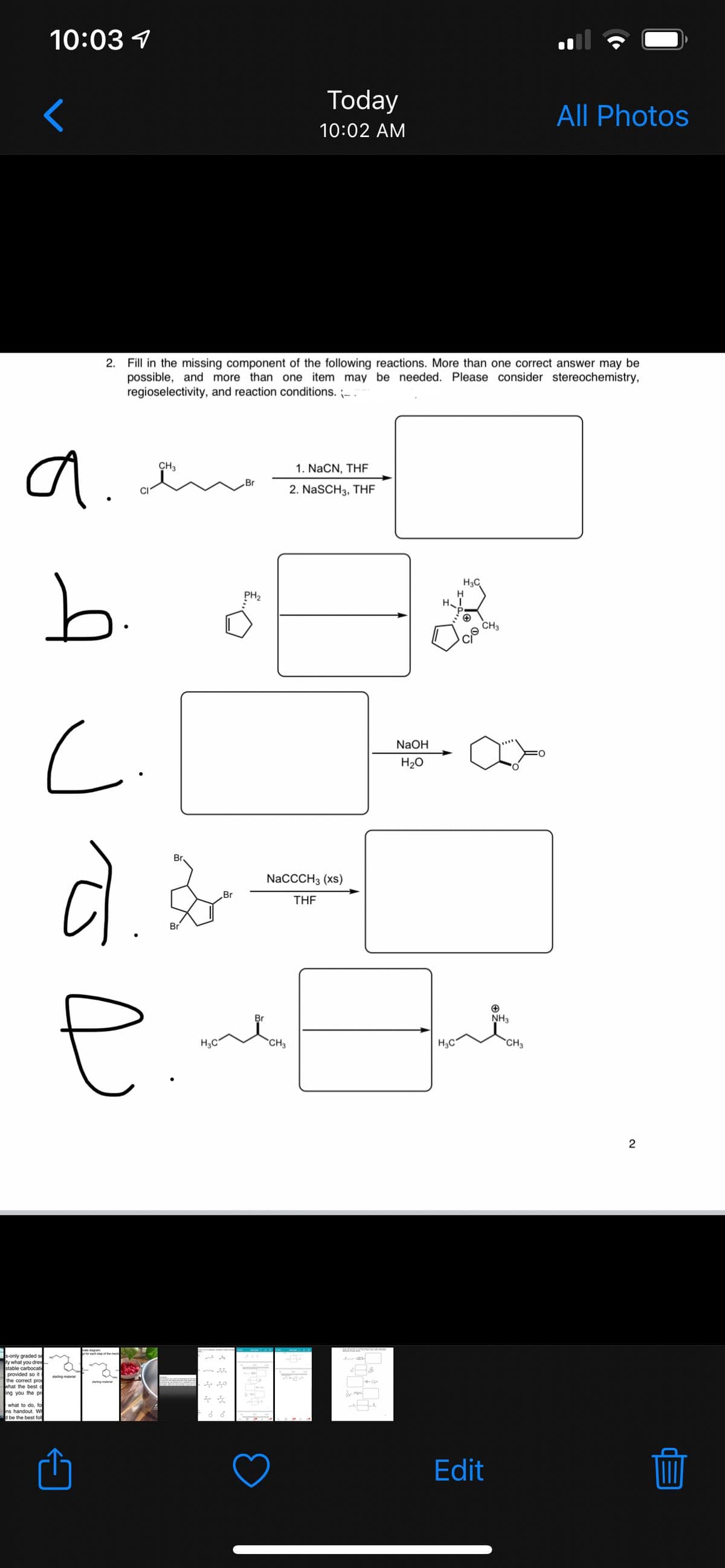
- Although the Diels–Alder reaction generally occurs between an electronrich diene and an electron-deficient dienophile, it is also possible to have inverse-demand Diels–Alder reactions between suitable electrondeficient conjugated double bonds and electron-rich alkenes. These reactions are particularly useful because they allow for the incorporation of heteroatoms into the new six-membered ring. Predict the products of each inverse-demand Diels–Alder reaction below. Be sure your products reflect the correct stereochemistry. If more than one regioisomer is possible, draw both.Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Fill in the blanks in the following reactions. Include stereochemistry when relevant.
- What would the final products look like? Pls specify stereochemistry if neededgive some real life application of stereochemistry1)Chemistry students are taking an experimental course in organic chemistry at a public university. During an experiment involving conjugated dienes, some doubts arose when discussing the results obtained so far: (a) A student obtained two products from the reaction of 1,3-cyclohexadiene with Br2. His lab partner was surprised to get only one product from the reaction of 1,3 - cyclohexadiene with HBr. Explain these distinct results. (b) One student, seeing the discussion of colleagues, commented that she obtained two distinct products when reacting 1,3,5-hexatriene with HBr, with different yields just by changing the reaction temperature. Explain the results she obtained using reaction mechanism and based on kinetic and thermodynamic control involving conjugated dienes.
- Chemistry Give the products of the reaction of 1 mole of 2-methy1-1,3-pentadiene with 1 mole of HBr. Whichproduct(s) will predominate if the reaction is under kinetic control? Which products) will predominateif the reaction is under thermodynamic control?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?please provide the machanisms of 1a, 1e, 1f
- What is the product (include the stereochemistry) of the 6 pi electron electrocyclic reaction below, use the appropriate Woodward-Hoffman rule.Uemura and coworkers studied a time dependent Diels-Alder reaction which first formed the endo product as the major organic product and with time produced the exo product (J. Org. Chem. 2018, 83, 9300−9304). Show the endo and exo product for the reaction below. Which is the thermodynamic product and which is the kinetic product? Explain your reasoning.Describe Suprafacial and Antarafacial Rearrangement:

