Q: Zn(s)|Zn2+(aq) || Fe2+(aq, ) | Fe(s) What would you change to increase the Ecell decrease Zn2+con...
A:
Q: 1. If 16.33 mL of 0.6664 M KOH was needed to titrate a sample of HC;H;O2to its equivalence point, wh...
A:
Q: The colligative properties of solutions depend on the total concentration of solute particles, regar...
A:
Q: Consider the insoluble compound zine hydroxide , Zn(OH), . The zinc ion also forms a complex with am...
A: Dear Student, According to Bartleby guidelines we have to answer first question only (in Multiple q...
Q: The colligative properties of solutions depend on the total concentration of solute particles, regar...
A:
Q: A small part of a crystal lattice is sketched below. The unit cell is outlined in red. How many atom...
A: Given : Structure of Unit cell To find : Number of blue , black and red atom in unit cell = ?
Q: 717.7 grams of isomaltitol, C12H24O11, was combusted in a furnace completely in an excess oxygen. Th...
A:
Q: Of the atoms/ions listed, select those with the electron configuration [Ne]3s23p6 and no unpaired el...
A:
Q: What kind of intermolecular forces act between a hydrogen peroxide molecule and a water molecule
A: The strongest intermolecular force between a water molecule and hydrogen peroxide molecule is hydrog...
Q: A glucose solution is 3.25% glucose by mass and has a density of 1.03 g/ml. What mass of glucose is ...
A:
Q: What amount of heat (in kJ) is required to convert 14.2 g of an unknown liquid (MM = 83.21 g/mol) at...
A: We have to calculate the amount of heat required.
Q: Consider the ring structure of β-D-glucose. It will give a positive test as a reducing sugar. Descri...
A:
Q: Find the partial pressure of carbon dioxide
A: Partial pressure of CO2 gas is- 10.2 kPa.
Q: A 85.0-mg supply of iodine-131, used in hospitals in the treatment of hyperthyroidism, was stored fo...
A:
Q: Use the Nernst equation to calculate the cell potential for the following cell in Volts: Cr|Cr3+, 0...
A: The cell given is, => Cr (s) | Cr3+ (aq, 0.0010 M) || Cu2+ (aq, 1.0 M) | Cu (s)
Q: Using acetylene as your only source of carbon atoms, identify a synthetic route for the production o...
A: Part 1: The route for the synthesis of a given reaction is: The reagents are: BDEI
Q: Calculate the volume of an FCC unit cell in terms of the atomic radius R
A: Calculate volume of an FCC in terms of R--
Q: Balance the following reaction MnO-4(aq) + Br-(aq) → MnO2(s) + BrO-3(aq) in basic solution
A:
Q: Na CH3I DMSO Draw SN2 product
A:
Q: 6) Draw the structure of the products 23 to 27. HO H2SO4 23 1) NABH4 2) H-О PBR3 24 25 H20, H2SO4 OM...
A: These reactions involve protecting Alcohol group so that ketone group becomes activated for Reac...
Q: Describe Le Chatelier's principle and describe how it would slow down a endothermic reaction in a co...
A:
Q: Part A-Using the Ideal-Gas Equation The Goodyear blimp contains 5.74 x 10° L of helium at 25°C and 1...
A:
Q: What nitrogen base is shown here? NH2 ALureeil ZI
A:
Q: Calculate the solubility of Mg (OH) 2 in 0.50M NH4Cl. (Kps = 1.8 x 10-11 Kb NH3 = 1.8 x 10-5 ).
A: Given: Concentration of NH4Cl = 0.50 M Since NH4Cl is a completely soluble salt. Hence it will disso...
Q: A phase change graph is shown below. What is the boiling point of this substance? A) 60°C B) 80°C ...
A: Option (D)is the correct answer that is 120°c. In phase change diagram, when temperature remains con...
Q: Which mechanism is going to predominate under these conditions? A SN1 B SN2 HO HCI or E1 D Е2
A:
Q: Imagine that two bottles are placed at opposite ends of the room at equal distance from them. One bo...
A: We will use grahams law of diffusion to answer this question.
Q: SAIC Ex 8.4 Calculate the wavelength associated with a neutron moving at 2.20 km s-. Is this wavelen...
A: By using debroglie's wavelength formula we will find it.
Q: Calculate pH of a solution that has hydrogen ion (H+) concentration of 0.044 M
A:
Q: Lipid Metabolism 4. Please propose the biosynthesis pathway of linoleic acid. 12 18 HO, 1 linoleic a...
A: Since you have asked multiple questions we will answer the first one for you. To get the remaining q...
Q: 3. When compound X (C15H17N) is treated with bezenesulfonyl chloride and aqueous potassium hydroxide...
A:
Q: :0: エ
A: Naming of organic compounds can be done by using IUPAC rule .
Q: Primary bonds are from molecule-to-molecule attractive forces. Question 8 options: True False
A: Primary bonds involve sharing or donating electrons between atoms to form a more stable electron con...
Q: A sample of krypton gas at a pressure of 1.04 atm and a temperature of 25.1 °C, occupies a volume of...
A: We will answer this question using Boyle's law.
Q: A student collected the following data in the kinetics of the formation of carbon monoxide at 3000K:...
A: The kinetics data given is, Time, s PO2, bar 0 1.00E+00 2.907 6.03E-02 5.514 3.64E-03 8.6...
Q: Consider a buffer solution that consists of two separate components: the weak acid, HF(aq), and its ...
A: Given: A buffer solution of HF and F-.
Q: Question 53 How many moles of water (H,O) are in 35.0 grams of water? A 18.0 moles 1.94 moles 35.0 m...
A: We have to calculate the moles of water
Q: Part I: Calibration of the Plastic Pipette Number of Drops Mass of 10 Drops (9) Volume of 10 Total M...
A: Part I : To calculate mass of 10 drops , we would use the given data values. Then to calculate volu...
Q: Alisha went to the doctor and got her blood calcium levels measured. If the doctor found that a 1.7 ...
A:
Q: a) From this flow chart, which acid is extracted when sodium bicarbonate is used as an extraction so...
A: a. Benzoic acid strong acid and reacts with sodium bicarbonate (weak base). Phenol cannot react with...
Q: Draw the major product of the substitution reaction shown below. Ignore any inorganic byproducts. Br...
A:
Q: 03.jpg What is the electronic geometry of this molecule? (what is geometry of electron pairs?) Pleas...
A: In a given question, we have to find out the electronic geometry of given compound which is ozone O3...
Q: Drag the appropriate items to their respective bins. Reset Help XeF4 CI, NH3 GaH3 CH,F TeF, Nonpolar...
A: A polar molecule is the molecule in which one end of the molecule is slightly positive ,while other ...
Q: An unknown gas effuses at a rate 0.500 times the rate of CH₄. What is the molar mass of the unknown ...
A: The process of escaping of gas from containers through a hole is known as effusion
Q: What is the Normality of a 25ml solution of Hydrochloric acid, that neutralizes 20ml of a 0.50N sodi...
A:
Q: 8) I dentify the function al groups present in each: b) CH3- CH- cH, - CH,- CH- ċ - cH3 CH3 Br d) qN...
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only....
Q: Imagine that two bottles are placed at opposite ends of the room at equal distance from them. One bo...
A: Which has an odor like that of a rotten egg. Which odor will they sense first?
Q: A very long pipe is capped at one end with a semipermeable membrane. How deep (in meters) must the p...
A:
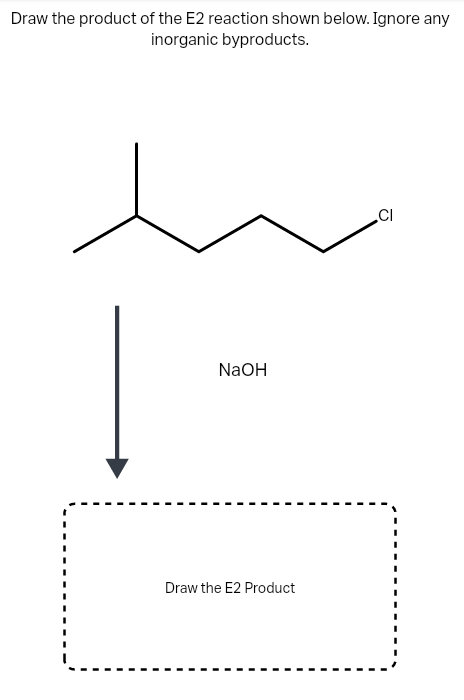
Draw the product of the E2 reaction shown below. Ignore any inorganic byproducts.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the major product of this SN1 reaction. Ignore any inorganic byproducts.Draw all elimination products that could be formed in an E1 reaction. Ignore the possibility of rearrangements. Ignore any inorganic byproducts.After E2 elimination, will this product contain deuterium (D)? Justify your answer.

