Q: B. Write the complete and abbreviated electron configurations for the following elements. 4. Ag 5.…
A: Since, Electronic configuration represent the electron present in orbital in which way. Arrangement…
Q: What is the molarity of ions in a 0.648 M solution of Ca(OH)₂ assuming the compound dissociates…
A: Given, Molarity of Ca(OH)2 solution = [Ca(OH)2] 0.648 M Ca(OH)2 completed dissociate in the…
Q: dict the products of the following acid-base reaction. е осна HC С: + H-POCH3 H —OCH3 HC art 2: Ө…
A: Solution - If keq of reaction is less than 1 than less reactant will react to form product and…
Q: When the temperature is lowered, the speed at which molecules move is slowed. O False True
A: We have been given a statement that when temperature is lowered, the speed at which particles move…
Q: Draw the Lewis Structure of sulfate (SO4^2-) that minimizes charge. Include all electrons and formal…
A: We have been given an ion ,SO42-.We have been asked to draw the lewis structure that minimizes…
Q: Given that K₂ for HIO is 3.2 x 10-¹1 at 25 °C, what is the value of Kh for IO at 25 °C? Kb = Given…
A:
Q: In addition to more highly fluorinated products, fluorination of 2-methylbutane yields a mixture of…
A: Fluorination of alkane to give alkylflorides . The structure of 2-methylbutane is as follows:
Q: Vmax A represents Y (C) 0 B +1 Slope (C) KM Vmax -1 Slope = KM Vmax X Q13) Which mode of inhibition…
A: Michaelis-Menten kinetics are characterised by the assumption that the enzyme and substrate…
Q: Carborundum (silicon carbide), SiC, is a very hard material used as an abrasive on sandpaper and in…
A:
Q: The hydronium ion concentration in a sample of springwater is found to be 5.2 ✕ 10−6 M at 15°C. What…
A:
Q: me of the compound below. Br
A: We have been given an organic compound and we have been asked to name given organic compound.The…
Q: A 1.595 g samples of a new organic material is combusted in a bomb calorimeter. The temperature of…
A:
Q: the mass of sulphur is 14.4 g calculate the mass of iron required to set up a reaction between these…
A: The equation for the reaction between sulphur and iron that produces iron sulfide is Fe + S…
Q: Give the IUPAC name for the following compound. 3 di ethyl sa 2,7-dimethyl 5-tert methyl decane
A: 1) Select longest parent alkane chain having 10 carbon so decane 2) numbering always start from that…
Q: please draw lewis structure and give answer. + Click to draw a new structure S Draw the Lewis…
A: Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It…
Q: nces. ure.
A: 7-a true 7-b. True 7-c. True 8/a. Compound 8/b. Elements 8/c. Soap is not a compound, it…
Q: C. Calculate the number of atoms or molecules in each of the following samples. 19) 0.357 moles of…
A: Since you have asked multipart type questions, we will solve only first three questions for you. If…
Q: In naming alkyl halides, the -ide ending becomes -ic. If the halide is in an acidic form, the prefix…
A:
Q: What volume (in L) of a 0.779 M hydrochloric acid (HCl) solution can be prepared from the reaction…
A:
Q: Question 2 please
A: Conjugation : When p orbital of one atoms overlaps the p orbital of the neighbouring atom or sigma…
Q: Strong basic solution will turn a phenolphthalein indicator pink. Q47: Competitive inhibitors can…
A: Michaelis-Menten kinetics is the the steady-state approximation, the free ligand approximation and…
Q: please im in rush. I will give helpful rating promise. no need for solution or explanation
A: We have to match the given type of corrosion in the circumstances.
Q: What is the difference of analytical and physical chemistry?
A:
Q: A chemist weighed out 7.2g of mercury. Calculate the number of moles of mercury she weighed out. Be…
A: Given: mass of mercury (Hg) = 7.2g Molar mass of mercury (Hg)= 200.59g/mol
Q: The following sequence of reactions was employed during synthetic studies on reidispongiolide A, a…
A: In the given reaction step 1 involves reaction of epoxide with Grignard reagent.
Q: Calculate the concentration of Ag+ for the following cell's emf to be zero at 25°C. Given that the…
A: A standard cell potential (E°) is a measure of the potential difference between two half-cells at…
Q: Using the smallest possible coefficient number, can you balance these equations? Cr(s) + Se(s)…
A: Given Unbalanced chemical reaction 1. Cr (s) + S8 (s) → Cr2S3 (s) 2. NaHCO3 (s) → Na2CO3 (s) + CO2…
Q: A paramecium weighs about 5 picogram consumes about 10 bacteria (weighing 1 picogram each) per hour.…
A: To determine the paramecium's metabolic rate, we first need to calculate the number of moles of…
Q: Using the thermodynamic information in the ALEKS Data tab, calculate the standard reaction free…
A:
Q: Calculate the wavelength of each frequency of electromagnetic radiation. Show work: a. 100.2 MHz…
A:
Q: Explain major depressive disorder. State the factors which predispose an individual to depression
A: Depression is a common mental health disorder that affects millions of people worldwide. It is…
Q: During a lab, a student wrote the following on their data sheet: "A reaction occurred." How would…
A: We have been given that during lab, a student wrote A reaction occured on their data sheet.We have…
Q: Which one of the following electron configurations depicts an oxygen atom? Answers: 1s 22s 22p 4…
A: Electronic configuration: Electronic configuration can be define as the arrangement of electrons…
Q: Draw all significant resonance structures for the following compound:
A:
Q: (3) What will be following the Commutator. [*, Px] value where, the xax Px=-ind
A:
Q: How do you make a 10x stock solution for 10mM NaH2PO4 (monobasics). Show what should be the volume…
A: Molarity (M) is a concentration term for a solution. The molarity of a solution is defined as the…
Q: balance chemical equations with interfering coefficients
A: A chemical reaction follows the law of conservation of mass. According to the law of conservation of…
Q: The specific gravity of iron is 7.87 in the density of water at 4.00°C is 1.00 g/cm^3. You can use…
A: Specific gravity=(density of substance) ÷ (density of water @4° C) Given : density of water @ 4°C is…
Q: What mass of SrCl₂ (d = 3.05 g/ma must you measure in air to obtain a true mass 3.4351 g ? Assume.…
A: •Given:- Density of SrCl2 (ρx) = 3.05 gm/ml True mass(Mx) = 3.4352 gm Density of air (ρn) = 0.0012…
Q: Q44: Among the following, the least sensitive assay for measuring protein concentration is: A) Lowry…
A: The Biuret method was the least sensitive assay for measuring protein concentrations.
Q: The chemical formula for lithium hydride is LiH. A chemist measured the amount of lithium hydride…
A: Moles = Mass / Molar mass Moles of A = Mass of A / Molar mass of A
Q: A solution of Ibuprofen contains 150 grams in 800 mL. What is the percent strength of this solution?…
A: • Here we have to calculate the percent strength of the given Ibuprofen solution. • The formula to…
Q: mass (in g) of silver oxide, Ag2O, is required to produce 28.7 g of silver sulfadiazine,…
A: Given reaction is 2 C10H10N4SO2 + Ag2O → 2 AgC10H9N4SO2 + H2O Given mass of silver sulfadiazine,…
Q: Draw the most stable AND most unstable conformations of the following molecules. Briefly explain…
A: we know that gauche form is most stable than eclipsed form due to steric hinderance so a is more…
Q: Please correctly name this structure using IUPAC nomenclature N CO₂Et IZ CO₂Et
A:
Q: What is the pH of an aqueous solution of 8.57E-2 M barium hydroxide? pH =
A:
Q: Draw a Lewis structure and determine the point group for each of the following molecules. Indicate…
A: Xenon formal charge = 8-4-8/2 =0
Q: (1) Identify each of the following half-reactions as either an oxidation half-reaction or a…
A:
Q: Be sure to answer all parts Part 1 out of 3 Predict the products of the following acid-base…
A:
Q: C. Answer the following questions related to quantum numbers. 1. Indicate if the following sets of…
A: Since you asked specific parts of the question so as per Q&A guidelines of portal I solved…
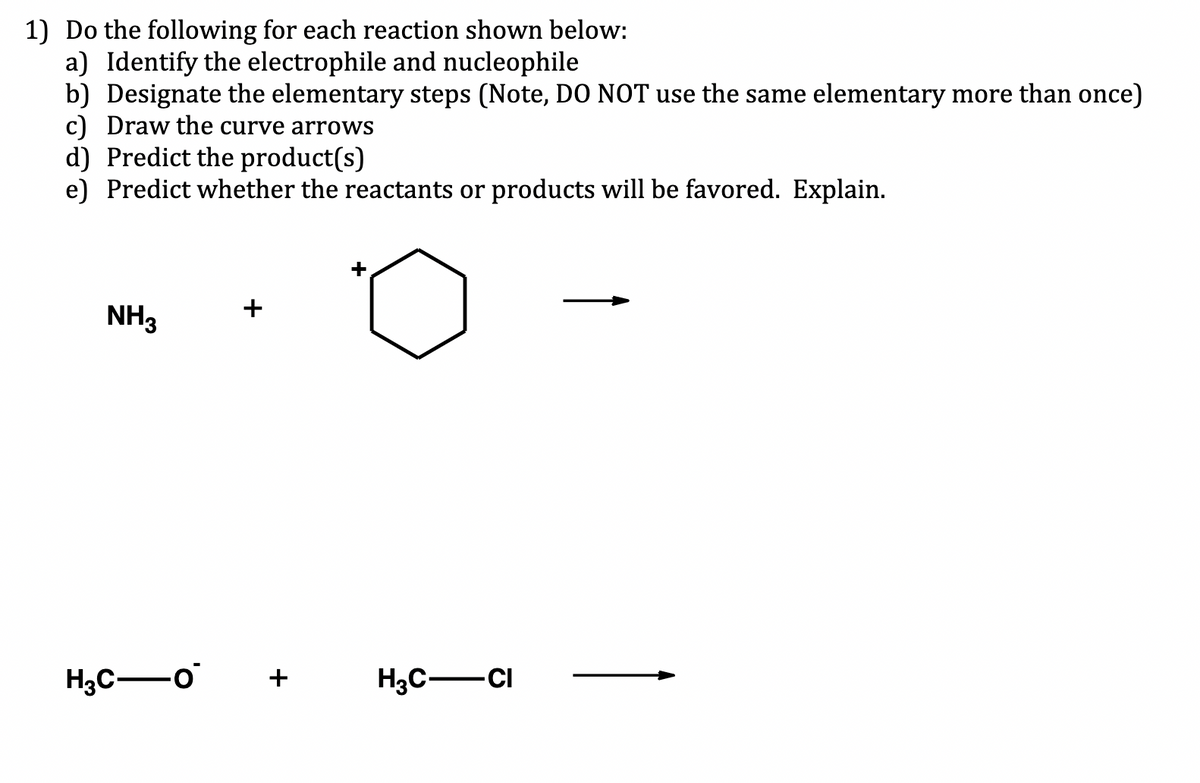
Step by step
Solved in 2 steps with 1 images

- (1) Identify nucleophile and electrophile (2) Draw mechanistic arrows (3) Identify which elementary step is occurring1 example for organic rearrangement reaction show: a. the overall reaction (reactants --> products) b. the reaction mechanism (indicate intermediate product) c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?) d. overall description of the reaction eg., radical substitution or SRDraw the products for the addition reactions below in the boxes. Then, draw the FULL electron-pushing mechanism for the reactions, including all intermediates (with formal charges and lone pairs of electrons) and all electron pushing arrows. Label the electrophile and nucleophile in each step.
- Match the reagents to the reaction (place letter on reaction arrow) Only letters C. & D. Are left to use. Which one is the best reagent for the reaction?Add curved arrow(s) to draw step 1 of the mechanism. Modify the given drawing of the product as needed to show the intermediate that is formed in this step.What is the major product of the reaction? The correct answer is C, please show the mechanism.
- 1 example for organic elimination reaction show: a. the overall reaction (reactants --> products) b. the reaction mechanism (indicate intermediate product) c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?) d. overall description of the reaction eg., radical substitution or SRfor part b, besides drawing the reaciton coordiate diagram, can you give. a quite detailed explanation about how do we know which curve corrresponds to A or B. i.e. how do we know A or B energy is higher ; whether theres energy differnce between reactant and products. thanks help with part b please1. Of the two unimolecular reactions shown, decide which you think would be faster, i.e., which would have a higher relative rate constant (krel)? 2. Based on your answer, draw two lines on one reaction coordinate diagram that show the relative progress of each reaction along its path to product indicated? 3. what is Bond dissociation energy of the C-LG bond? 4. what is the Nature of the leaving group (LG)? 5. what is the relative size of activation energy (Ea) for each reaction? 6. what is the Hammond's postulate? 7. what are the Relative thermodynamic stability of the reactive intermediates? 8. what is Influence of the solvent (if given) on the reactions and intermediates?
- What are the major products of the following reaction? The correct answer is B but please show the mechanism.1 example for organic substitution reaction show: a. the overall reaction (reactants --> products) b. the reaction mechanism (indicate intermediate product) c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?) d. overall description of the reaction eg., radical substitution or SRDraw the complete mechanism of each pair of reactants including any favorable rearrangements and all important resonance structures of all intermediates. a. Which reaction has a lower PE carbocation intermediate? b. Draw an energy diagram showing the reaction profiles of both reactions in the previous question. Use a dotted line for the first pair of reactants and a solid line for the second pair of reactants. (Assume the energy of the starting materials and products are the same for both pairs and the reactions are neither uphill nor downhill on net. c. Mark points on the energy diagram corresponding to each carbocation in your mechanisms.

