но choices for number 1: HO H NH HN H =0 *HN NH3 a b но OH NH *H3N H H *H;N d
Q: OH „HC D CH. E F B A
A: Cyclopentane ring: It is the cyclic structure of alkane with 5-carbon atoms in its structure.…
Q: OH HO HO A NH2 Но. HO R-O. HO. HO HO R-O, OH Но HO `OH OH Но R-O, D HO. но но HO Но `OH OH B но. `OH…
A: Glycosidic bond is a type of covalent bond formed after reaction between hydroxyl group of…
Q: 39. Arrange the following amino acid according to decreasing non-polarity (left to right). 1-…
A: Amino acids are biomolecules in which an alpha carbon is bonded to 4 groups. These groups are ; an…
Q: Below is the structure of D-Talose in Fischer projection. In Haworth projection draw the structure…
A: Introduction: Talose is an aldohexose sugar that is a C-2 epimer of galactose and a C-4 epimer of…
Q: 1. For the following trisaccharide: a. Provide the abbreviated name b. Draw the chemical structure…
A: Trisaccharide are the oligosaccharides composed of three monosaccharide units with two glycosidic…
Q: 2. Draw the structure of 1-palmitoleyl-2-linolenyl-3-stearyl triacylglyceride
A: 1-Palmitoleyl-2-linolenyl-3-stearyl triglyceride: This is a triglyceride compound containing…
Q: 1. Indicate whether each of the following molecules is an alpha amino acid or not and explain why.…
A: Proteins are defined as the polymers of amino acids. It is a macronutrient that plays a very…
Q: 3. Consider the following acids and their dissociation constants: H:SO:(aq) + H20(1) = H:O-(aq) +…
A: a) If Ka value is less this means that ionisation is less and if Ka value is more this means that…
Q: For each amino acid: [1] draw the L enantiomer in a Fischer projection; [2] classify the amino acid…
A: Introduction: Amino acids are organic compounds that contain amino and carboxyl functional groups,…
Q: explain and describe briefly the structure of MgO-NH2-Ru-Pd-Sn MgO o-Si-(CH,),NH, RuClx…
A: Asked : Explanation of given structure
Q: 11. Which of the following correctly illustrates a dipeptide and an amino acid in the optimal…
A: A COOH is facing a NH2. when these 2 compounds react a dehydration reaction (removal of water) will…
Q: Identify the chiral carbons in -D-fructose. Identify the appropriate atom by selecting an atom and…
A: A chiral center is defined as an atom in a molecule that is bonded to four unique atoms or groups…
Q: H. N-C-c H он -CH H,C ÇH2 CH, The monomer above is a(n) ( Select] . The changeable side g in this…
A: Introduction: Amino acid (isoleucine) Non-polar branched hydrocarbon Translation Polypeptide…
Q: 1. Is the disaccharide reducing? Yes or no and why. 2. Will the disachharide react positively with…
A: Reducing disaccharide sugars are those that consists of two monosacharides , in which one of the…
Q: 7. When carbohydrates in their Fischer projection form a pyranose ring structure, it can be either a…
A: Carbohydrates are polyhydroxy aldehydes and ketones which are associated with reducing properties.…
Q: uestion 13 /hich of the following is the correct structure of the tripeptide Gly-Phe-Ala? a. Ph H.…
A: A tripeptide is a polypeptide that was formed by joining an alpha carboxyl group of one amino acid…
Q: The structure given below is a tetrasaccharide. i) For each pair of sugars (1-2, 2-3, 3-4), identify…
A: Carbohydrates are defined as a group of carbonyl compounds that occur naturally. These are aldehydes…
Q: 64. How much of the R enantiomer is present in 10 g of a mixture which has an enantiomeric excess of…
A: 64. The enantiomers are stereoisomers that are non-superimposable mirror images of each other but…
Q: 16. Which of the following choices correctly describes the molecular structure of a..ova A. There…
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen, but other elements are…
Q: 2. Draw the structure of the tripeptide Gly-Ala-Tyr H H. N-C-C H N-C-C + N-C-C + H он он CH3 H. 0-H…
A: Proteins have four levels of structural organization including Primary, secondary,…
Q: 29) Polyethylene can be obtained in two different forms. High-density polyethylene (0.94 g/cm3 is a…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Lysine is similar to ornithine. .Why does lysine not form a lactam? a. Infrequency of lysine…
A: A lactam is a cyclic amide (lactone+amide). A lactam antibiotic is an antibiotic that contains a…
Q: 2. Calculate the overall charge (pH 7) on the following three polypeptides and answer the questions…
A: Proteins or peptides are composed of twenty standard amino acids that differ from each other on the…
Q: 1. Draw the condensed structural formulas for A and B. [0] [0] CH3 A B CH,-C I-CH,-C-OH
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: 9. Identify the monosaccharides and their linkages in the following disaccharide. HOCH, но 0-CH он…
A: A carbohydrate is a biomolecule composed of carbon, hydrogen, and oxygen. Carbohydrates are…
Q: A polypeptide with a net positive charge at physiologic pH (~7.4) most likely contains amino acids…
A: The pKa values of the side chains of amino acids determine the net charge on a protein at a…
Q: 3. Write the names of following sugars as Pyranose/Furanose, Pyranoside Furanoside, Alfa/beta,…
A: Sugars or carbohydrates are the most abundant biopolymers in living organisms. Carbohydrates are…
Q: The initial pH of a 0.2M arginine solution is 14. a. What is the predominant structure of arginine…
A: Arginine is a basic amino acid and it contains a guanidino group in its side-chain. Guanidino group…
Q: gal(a1–6)gal(a1→6)glc(a1→2B)fru. Which of the complete IUPAC of this tetrasaccharide? O…
A: The term "biomolecule" refers to a molecule created by living organisms or cells. The most common…
Q: Draw the Fischer, Haworth & chair representations of the given monosaccharides. Sugar Fischer…
A: Monosaccharides are polyhydroxy aldehydes or ketones which are associated with reducing properties.…
Q: 1) Below you are given the structures of the disaccharides lactose and trehalose. но OH OH OH но но…
A: Lets first assume that all the carbohydrates given here are D isomers , cause that the general case…
Q: CH-OH НО OH What is the name of this structure? OH OH alpha-D-gulopryranose O beta-L-gulofuransone O…
A: Pyranose : Has 6 membered ring with 5 carbon atoms and 1 O atom Furanose : Has 5 membered ring with…
Q: Using Haworth projections, draw the α and β anomers of the given monosaccharide (Fischer Projection…
A: Carbohydrates are polyhydroxy aldehydes or ketones that are associated with reducing properties.…
Q: 1. Draw the tetrapeptide Met-Ala-Thr-Thr at a ph of 7? 2. Draw the tetrapeptide Met-Ala-Thr-Thr at…
A: The pKa values of amino acid side chains determines the pH-dependent net charge of an amino acid in…
Q: 2. Calculate the overall charge (pH 7) on the following three polypeptides and answer the questions…
A: Hi! Thank you for the question. We are authorized to answer three subparts at a time, since you have…
Q: 1. Draw the structure of the following triacylglycerols (TAGS) 1,2-dioleoyl-3-myristyl-sn-glycerol…
A: Triacylglycerol: Triacylglycerides are esters of fatty acids that are hydrolyzed to glycerol and…
Q: For the complete name of the Hawort structure given, (use the structure given fischer projection) *…
A:
Q: 1. provide three reasons why most of the research on carotenoids concentrates on b-carotene.
A: *NOTE: Kindly repost for other question Dear Student as per the guidelines we are supposed to answer…
Q: 6. Treatment of cholesterol with mCPBA results in formation of a single epoxide A, with the…
A: mCPBA is a peroxycarboxylic acid. It is often preferred to other peroxy acids because of its…
Q: Shortly discuss the above antibodie structure? Please discuss at your own words . Discussion should…
A: The picture given is the basic structure of an antibody(immunoglobulin) which is a Y-shaped…
Q: choices for question 1: H LOH NH2 INH3* HO H 'N' `NH2 `NH2 a b H LOH LOH NH2 INH2 `NH2 `NH2 H A H A…
A: Hi! Since you have posted multiple questions, we will answer the first one for you because we are…
Q: 4. This question is about the effect of geometric isomerism on the melting and boiling points of…
A: Cis-1,2- dichloroethene - Polar (i) Same groups(Cl) are present on the same side : So, net dipole…
Q: 9. Titration of valine by a strong base, for example NaOH, reveals two pk's. The titration reaction…
A: Answer 9 - option D) -NH3+ + OH → ¬NH₂ + H₂0 Explanation: Valine has two acid-base groups: the…
Q: 1. (a) Arrange the following lipid molecules in the order of their relative melting points. Н.С…
A: Hi! Thanks for your question. As you have posted multiple questions and have not mentioned which one…
Q: 7. Convert the following Haworth projections to a Fischer projection. Name the monosaccharides you…
A: Carbohydrates are the polyhydroxy ketones or polyhydroxy aldehydes or their derivatives. In…
Q: (a) A decapeptide has the following amino acid composition: Ala2 , Arg, Cys, Glu, Gly, Leu, Lys,…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Convert the Fischer projection to the Haworth projection of the a-furanose form by moving the…
A: The Fischer projections are a standard method for depicting the three-dimensional arrangement of…
Q: 9. Write the tripeptide structure for: val-ser-cys. 10. Write the reactions for: phe + ser
A: Amino acids are the building blocks of proteins. whereas each amino acid consists of an amino group,…
Q: 2- Please consider the polysaccharide structure below CH2OH CH2OH A CHOH CHOH -CHe CHOH о он OH CH…
A: Carbohydrates are the biomolecules also known as sugars that have carbon, hydrogen, and oxygen. They…
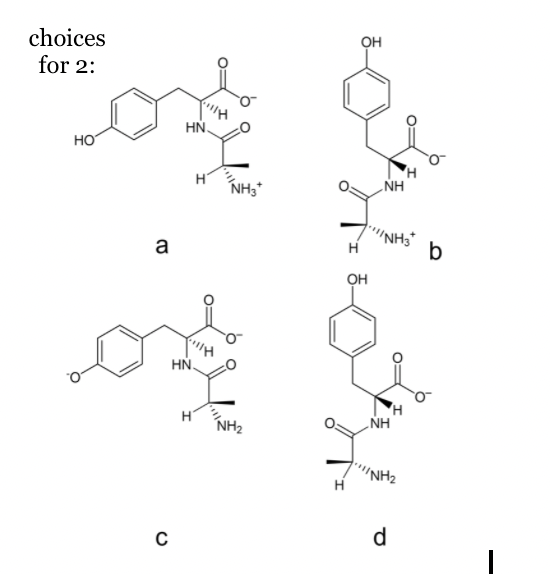
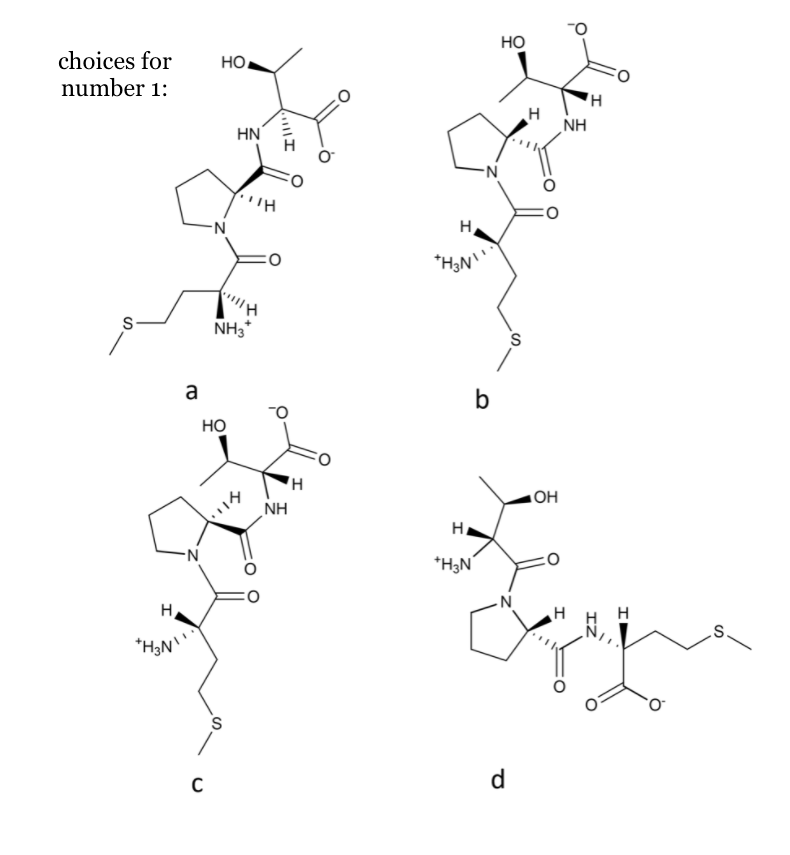
1. Choose the proper structure of the predominant form of L-M-L-P-D-T at pH 5. Provide a short explanation.
2. Choose the proper structure of the predominant form of L-A-L-Y at pH 11. Provide a short explanation.
Step by step
Solved in 5 steps with 4 images

- ASAP explain Correctly. Please explain .b and c please.... explain wellFifth Hospitalization At age 58, Vincent was rushed to the emergency room with severe vomiting of bright red blood. On examination, he had a blood pressure of 60 mmHg / 30 mmHg. The bleeding and vomiting started abruptly while Vincent was eating some hard, dry French bread. An endoscope (i.e. a flexible tube equipped with a camera) was placed down Vincent's esophagus, and a diagnosis of esophageal varices was quickly made. Why is bleeding particularly dangerous for Vincent?



