1. Draw the structure of the following triacylglycerols (TAGS) 1,2-dioleoyl-3-myristyl-sn-glycerol b. sn-16:0-12:0-16:0 а. c. LLnO
Q: What sugar units make up the structure shown? ÇH,OSO,- Но Lo- H. H. ÇOO- NHCCH, OH H H он
A: Carbohydrate consists carbon, hydrogen and oxygen. It is of different types like monosaccharides, di...
Q: Which of the alternatives below indicates techniques used both to purify and to concentrate proteins...
A: Proteins are polymers of amino acids linked by peptide bond and it is one of the important biomolecu...
Q: Question 18 The tRNA having a cloverleaf structure suggests the following features EXCEPT some segme...
A: tRNA : It folds to form the cloverleaf 2° structure which is held in shape via base pairing in at le...
Q: Answer the problem correctly. Trust me. I'll rate you helpful. Cross my heart.
A: Given Values: The volume of the acetate buffer = 250 ml Concentration of the acetate buffer = 0.5 M ...
Q: The following sattements are true for glycogen. EXCEPT:
A: The answer of the following question is given below
Q: Which of the following statements concerning sugar polymers is false? Sugar polymers may vary ...
A: Carbohydrates, often known as sugar molecules, are sugars. Carbohydrates are one of three primary nu...
Q: Sucrose also known as invert sugar can be enzymatically hydrolyzed by the enzyme amylase to give the...
A: Sucrose is a di-saccharide made up of one glucose and one fructose joined together with α-D-Glucose-...
Q: B.1.351 is a variant of SARS-CoV-2. It has the amino acid substitution N501Y in the spike protein on...
A: The aromatic amino acids like Tyrosine, tryptophan and phenylalanine absorb ultraviolet light. The a...
Q: Which of the following is the correct systematic name of the disaccharide shown? OH OH но но но- он ...
A: Carbohydrates are composed of carbon, hydrogen, and oxygen in the ratio of 1:2:1. Carbohydrates act ...
Q: A poultry farmer is curious about the effectiveness of two types of exogenous enzymes, namely papain...
A: Proteolytic enzymes are useful as a supplement to thr diet of ducks and turkeys as well as chickens....
Q: What is the major tissue source of Alanine Aminotransferase? 2. What is the clinical significance o...
A: Alanine Aminotransferase(ALT) Enzyme which catalyzes amino group tranfer to alpha-ketoglutarate from...
Q: Indicator Low pH color • Transition pH range • High pH color • blue-violet green Gentian violet (Met...
A: Methyl orange = yellow; from reference table, color Red…<3.1-4.4<Yellow Methyl red = yellow;...
Q: What type of regulation would be expected when anaerobic metabolism is occurring in the muscle? Sele...
A: Depending on the need of the body, the fate of intermediates of Glycolysis, gluconeogenesis, Citric ...
Q: . Potassium: Insulin Overdose, Magnesium: Antacids a. both decreased b. both increased c. firs...
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any sp...
Q: A protein that is normally found in an aqueous solution has these amino acids in its primary structu...
A: Proteins are composed of the linear chain of amino acid sequences attached via peptide bonds. Protei...
Q: Exercises: Directions: Complete the following lines for frame shift mutations. Write each codon on t...
A: Insertion mutation involves addition of genetic material in the form of nitrogen base to the reading...
Q: With the aid of a simple generic diagram: i) IDENTIFY and EXPLAIN how the type(s) of chemical bondi...
A: Dear Students, I have dowanloaded the PDB ID 3GRS structure from https://www.rcsb.org/ This is the s...
Q: Which of the following are important biological functions of amino or acid derivatives of sugars? ...
A: Monosaccharides, which are single units of carbohydrates, can go through a variety of processes to p...
Q: Kindly answer 16, 17, 18, 19, and 20. Provide the letter of its answer
A: 16.The correct option is (c) Phosphatidylcholine 17. The correct option is (a) Membrane lipids
Q: 4. What is the major biochemical function for each of the following proteins? a. a Keratin b. Collag...
A: Fibrous proteins are proteins that have cellular or extracellular roles in behaving as a robust stru...
Q: Can u help me answer this 1. WHAT IS IMRAD FORMAT? 2. DRAW A DIAGRAM SHOWING THE LIFE OF THE SCIENTI...
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any sp...
Q: Draw out the peptide DPGS as it would appear in a Type II β-turn and clearly label/identify all the ...
A: Beta turns are a prominent secondary structure seen in proteins. The 2 most common types of beta tur...
Q: (In 10 sentences) Can disaccharide be completely assimilated and used by human cells as a direct sou...
A: All disaccharides are dimeric product of monosaccharide linked together with glycosidic bond. Sucros...
Q: how does the molecular basis of antigenic variation used by the trypanosome parasite makes the produ...
A: Antigenic variation is a powerful survival strategy adapted by certain species of parasitic protozoa...
Q: Which test can be used to differentiate ribose and glucose? Benedict’s Test Iodine Test ...
A: A molecule synthesized by living organisms or cells is referred to as a "biomolecule." Carbohydrates...
Q: DISCUSS THE IMPORTANCE OF THE CARBOHYDRATES IN PREPARATION OF CULINARY CUISINE
A: Any substance that can be consumed by living organisms in order to obtain energy and survive is refe...
Q: The graph below shows the change in the frequency of one allele over 500 generations in a large popu...
A: Environmental factors include temperature, food, pollutants, population density, sound, light and pa...
Q: For the following aspartate reaction in the presence of inhibitor, Km = 0.00065 M. Determine Vmax in...
A: Enzymes are usually protein molecules which catalyzes biochemical reaction by decreasing the activat...
Q: What is the difference in ATP yield per glucose molecule between the malate-aspartate shuttle and th...
A: The inner mitochondrial membrane is impermeable for NADH (electron carrier) during glycolysis. Hence...
Q: Please provide an explanation for how to work through this practice problem: Using table 12.1, cal...
A: Given, cAMP breakdown: -50.4 kJ/mol ATP formation: -30.5 kJ/mol (Also for ADP formation from AMP, as...
Q: 2) oxaloacetate (OAA) occurs as an important intermediate in 2 metabolic processes a) indicate these...
A: "Since you have posted multiple questions we will answer the first question for you. If you want the...
Q: QUESTION 6 Enzyme chymotrypsin cleaves polypeptides after aa. O Positively charged O Large, non-pola...
A: Chymotrypsin is a digestive enzyme. This enzyme belongs to the family of enzymes called serine prote...
Q: Serine is an uncharged, polar amino acid (below and left). Threonine (below and right) is another un...
A: Amino acids are the building blocks of proteins which are composed of amino group (NH3+), carboxyl g...
Q: Identify the chiral carbons in -D-fructose. Identify the appropriate atom by selecting an atom and a...
A: A chiral center is defined as an atom in a molecule that is bonded to four unique atoms or groups at...
Q: Complete the crossword puzzle below 2 3 4 EF Created using the Crossword Maker on TheTeachersCorner....
A: The Hormones that are produced by adrenal cortex are important for regulation of metabolism and immu...
Q: Lactose is made up of D-galactose and one unit of D-glucose joined by a alpa-1,4-glycosidic bond and...
A: Lactose is one of the disaccharides that is mainly present in milk. It is one of the most important ...
Q: True or False: Haworth and Fischer projections allows an individual to see reactive sites in glucos...
A: The structure of monosaccharides can be represented in the Fischer projection or Haworth projection....
Q: Tricarboxylic acid cycle (a.k.a.krebs cycle)?
A: Krebs cycle is one of the important cycle take place in organisms, it is also called as citric acid ...
Q: . TRUE OR FALSE a) Higher Rf value will be obtained from dextrin than that of glucose in a reverse-...
A: A Physical method for the separation of compounds is called chromatography. In two differen...
Q: Haworth Projection Formulas Practice Exercise Student Name:. Has 2 carbon Pentose? Ketose? Has pyran...
A: Carbohydrates contain aldehyde or ketone compounds with multiple hydroxyl groups present in them. Th...
Q: ou have given two proteins with a pI of 4.5 and pI of 7.7? Using an anion-exchange column, how can t...
A: Ion-exchange chromatography: The method involves the separation of ions and polar molecules based o...
Q: What is glycemic index (GI)? Which food in the Philippine market has significant GI and list them wi...
A: GI is also known as Glycemic Index
Q: Choose from the dropdown menu, the description that best matches each of the components shown. A se ...
A: The plasma membrane, also known as the cell membrane, is the membrane that divides the inside of the...
Q: d. Which types of amino acids could you expect to find on the exterior of transmembrane proteins tha...
A: The environment in the interior of the lipid bilayer of the membrane is hydrophobic in contrast to t...
Q: Balance the chemical equation for the oxidation of arachidonic acid to ATP, CO2, and water. (Use the...
A: In the question given, Arachidonic acid is oxidized to ATP CO2 and water. Fatty acid oxidation proce...
Q: Choose any/all that apply to carbohydrates. Monosaccharides are oxidizing agents. Monosaccharide...
A: Carbohydrates are the biomolecules that is used in our body to produce or store energy.
Q: What is the reaction involved in the conversion of a sugar's carbonyl to an alcohol?
A: The answer of the following question is given below
Q: In a table, differentiate the three blood cells. Take note of their properties and functions, as wel...
A: Blood is the most important tissue of the body. Blood performs variety of different functions among ...
Q: Use the Stokes-Einstein equation to estimate the diffusion coefficient in m2/s and in um?/s for the ...
A: The Stoke-Einstein equation is used for calculating the diffusion coefficient of the spherical molec...
Q: ATP is an ALLOSTERIC INHIBITOR of the phosphofructokinase enzyme, which is a key catalyst for one of...
A: Phosphofructokinase is a regulatory enzyme of glycolysis. Phosphofructokinase is negatively inhibit...
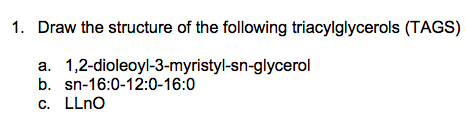
Step by step
Solved in 2 steps with 2 images

- 1. What is the possible identity of the AA? a. Aspartic acid b. Lysine c. Alanine d. Proline 2. What is the isoelectric point of AA? 3. What is the pKa responding to the dissociation of the alpha-carboxylic group?Amylopectin has which of the following structural features? a. only α(1,4) glycosidic linkages b. only β(1,4) glycosidic linkages c. both α(1,4) and α(1,6) glycosidic linkages d. both α(1,4) and β(1,6) glycosidic linkages1. Which one is NOT a property of glycerol? a. "backbone" in the simple lipid structure b. undergoes condensation with three molecules of fatty acids c. its OH groups reacts with the fatty functional groups of fatty acids d. it is a water-soluble ester 2. Which one has to bends in the structure? a. palmitic acid b. oleic acid c. linoleic acid d. myristic acid
- 3. One of the triacylglycerols found in corn oil contains palmitic acid, linoleic acid, and linolenic acid. During the production of spreadable margarine, all of the double bonds in this triacylglycerol are converted from cis to trans isomers. Linoleic acid: CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOHLinolenic acid: CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH Palmitic acid: CH3(CH2)14COOH a. Identify the unsaturated fatty acid(s) listed above and provide the associated omega designation. b. Draw an accurate representation of the structure of the triacylglycerol present in spreadable margarine. Circle all ester bonds in the structure you have drawn.1.A glucogenic amino acid is an amino acid that can be converted into glucose through glycogenesis. An example of glucogenic amino acid is _____________. * a.Histidine b.Lysine c.Leucine d.All of the given 2.The force between methylamine and acetate will be greater in methanol than in water because ___________. * a.Methanol has a lower dielectric constant than water b.Methanol, but not water, is structurally similar to methylamine c.Acetate and methylamine are more soluble in methanol than in water d.Dipole-dipole interactions between acetate and methylamine are induced in methanol, but not in water 3.Within the aqueous environment of an animal cell, sugars are stored as polymers rather than monomers. If the sugars were stored as monomers instead of polymers, which of the following properties would be least affected? * a.pH b.Viscosity c.Freezing point d.Osmotic pressure1. Which of the following is a structural component of a triacylglycerol? a. fatty acids b. glycerol c. both a and b d. neither a nor b 2. Which is TRUE regarding fatty acids? a. It contains a polar hydrocarbon chain. b. It is a component of oils and fats. c. both a and b d. neither a nor b 3. What happens in the stomach during lipid digestion? a. formation of micelles b. hydrolysis of lipids by salivary lipase c. both a and b d. neither a nor b 4. Which process does NOT happen in the small intestine during lipid digestion? a. secretion of bile salts b. formation of micelles c. absorption of long-chain fatty acids by the enterocytes d. a, b, and c are the correct answer. 5. Which of the following describes the enzymatic activity of pancreatic lipase? a. It hydrolyzes the bond between the fatty acids and glycerol. b. It emulsifies the lipid droplet into smaller globules. c. both a and b d. neither a nor b 6. What is the correct sequence of events during lipid digestion? I.…
- 7. The following compounds have high phosphoryl group-transfer potential except— phosphoenolpyruvate. phosphocreatine. glycerol-3-phosphate. adenosine triphosphate. 1,3-bisphosphoglycerate.1. a. Explain why the melting point of palmitic acid (16 carbons, no double bonds) is slightly lower thanthat of stearic acid (18 carbons, no double bonds). Explain why the melting point of oleic acid (18carbons, one double bond) is lower than that of stearic acid b. A mixture of lipids containing phosphatidic acid, cholesterol, testosterone, phosphatidylserine, andphosphatidylethanolamine was applied to a hydrophobic interaction chromatography column. Thecolumn was washed with a high salt buffer, and the lipids were then eluted with decreasing saltconcentrations. In what order would the lipids be eluted from the column? Explain your answer.A. A solution of amino acid having carboxylic side chain was titrated against NaOH. If the initial pH of the solution was 2.8. Describe the state of the functional groups of the amino acid at the initial pH and at 10.5 B. Briefly explain how you will separate amino acid mixture using the change difference between amino acids. C.A given glycine solution was titrated against NaOH and pH of the final solution was 4.8. Given that pKa1 and pKa2 of the glycine are 2.34 and 9.60 respectivel. Calculate the concentration of the dissociated amino acid in terms of the undissociated amino acid. If [x] and [y] are the concentration of dissociated and undissociated amino acid respectively.
- Which one of the following statements about the molecular structure of phosphatidylcholine is TRUE? a. It contains an isoprene chain. b. It contains a polar hydrophobic head group. c. It contains at least five alkene double bonds. d. It contains a phosphodiester bond. e. It contains a glycosidic linkage.A monosaccharide designated as an aldehyde sugar contains (a) a terminal carboxyl group (b) an internal carboxyl group (c) a terminal carbonyl group (d) an internal carbonyl group (e) a terminal carboxyl group and an internal carbonyl group_______ groups are the acid part of amino acids and fatty acids. a. Hydroxyl (OH) b. Carboxyl (COOH) c. Methyl (CH3) d. Phosphate (PO4)







