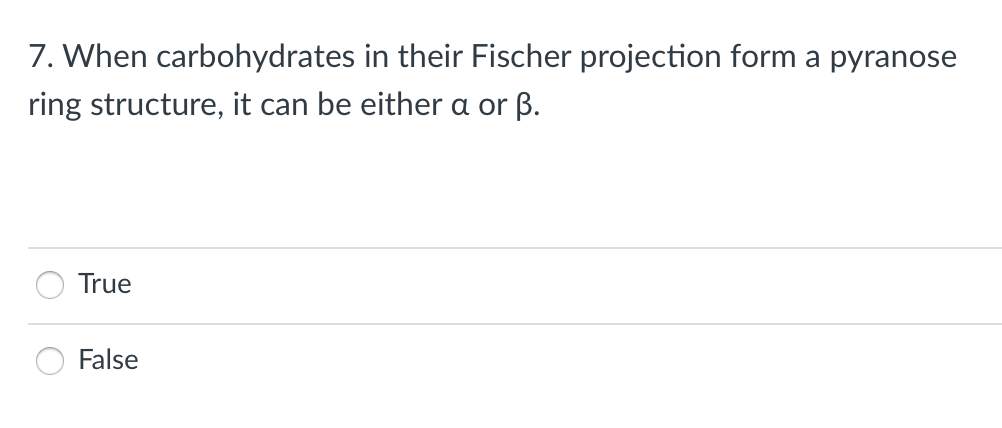
7. When carbohydrates in their Fischer projection form a pyranose ring structure, it can be either a or ß.
Q: 4. The following rules on aromaticity promotes a planar structure of nitrogenous bases EXCEPT: A.…
A: DNA and RNA are nucleic acids composed of a nitrogenous base, sugar molecule, and phosphate groups.…
Q: но choices for number 1: HO H NH HN H =0 *HN NH3 a b но OH NH *H3N H H *H;N d
A: Hi! Since you have posted multiple questions, we will answer the first one for you as we are bound…
Q: 3. On the DIPEPTIDE, label the following: Two central carbons, N-term, C-term, two Rgroups A…
A: Amino acids are the building blocks of proteins where several amino acids are linked together by…
Q: 6) What is the cognate a-keto acid of alanine? You may have to look at alanine.
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: 5. Draw the structures of the TWO glycoside products formed below. CH2OH OH CH3OH, H* но НО Он
A:
Q: 19. amylose it has B (1-4) glycoside bond
A: 19. Starch is made up of water soluble amylose and water insoluble amylopectin. Glycosidic bond is a…
Q: Part 2: Draw the enantiomer structure of the following given carbohydrates. Be sure to include…
A: In chemistry, an enantiomer is one of two stereoisomers that are mirror images of each other but…
Q: 3. Which of the following pair is an enantiomeric pair? *
A: Enantiomeric pairs are the isomers that are non-superimposable mirror images of each other. they…
Q: 4. Which of the following structure is a B-D-glucose? * CH2OH ОН ОН Н H. ОН Option 1 CH2OH H. ОН Н 1…
A: Glucose is a monosaccharide molecule and is the most abundant sugar of the carbohydrate family. The…
Q: 5. In a portion of polypeptide shown below, which bond is described by o O0 0? А. 1 В. 2 С.3 D. 4 E.…
A: A polypeptide chain is a linear chain of many amino acids.
Q: Which functional groups are present?
A: Compound is a chemical substance that is made up of more than two atoms which are joined by chemical…
Q: 2. Which of the following atoms can be found in every macromolecule (Select all that apply) (LS1-6)…
A: Each small organic molecule can form large molecule called macromolecule. There are four major…
Q: 4. Draw the -forms of the following sugars. CHO CHO H- OH но H- HO H- HO. OH OH CH,OH CH;OH 5. Draw…
A: A biomolecule is a molecule produced by living organisms or cells. Carbohydrates, proteins, lipids,…
Q: CO CH2 O CH2 O H3N-CH-C- NH-CH-C-OCH3
A: Amino Acids: These are the building blocks of protein. It contains an N-terminal which has an Amine…
Q: 22. All of the following are examples of disaccharide, EXCEPT: * A. maltose B. sucrose C. raffinose…
A: Introduction Disaccharide:- Disaccharides are molecules that are composed of two monosaccharides…
Q: 20. Which of the following is a polysaccharide with B-1,4 glycosidic linkages? C. glycogen E. None…
A: Polysaccharides are formed by bonding monosaccharides with each other. This bonding between two…
Q: . When digesting a complex carbohydrate, water is added and, a simple sugar is obtained through…
A: During digestion the large molecules are broken down into simple molecules like during protein…
Q: 5. Which of the following structure is the not stable structure of hexoses? 6 Он ОН4 HO HO. 5 HO- 2…
A: In the hexose structure or any structure when the position of OH group is in equatorial postion it…
Q: 8. Which of the following fatty acids is an essential omega-3-fatty acids? *
A: Omega 3 fatty acids play an important role in maintaining a healthy diet and healthy physiology.…
Q: 5. The main function of bile salts is:
A: Bile salts are made up of different kinds of elements called as free bile acids. Bile salts makes…
Q: 16. Which of the following choices correctly describes the molecular structure of a..ova A. There…
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen, but other elements are…
Q: 2. Draw the structure of the tripeptide Gly-Ala-Tyr H H. N-C-C H N-C-C + N-C-C + H он он CH3 H. 0-H…
A: Proteins have four levels of structural organization including Primary, secondary,…
Q: 1. What is the net charge of aspartic acid when the pH is 0.10? 2. Draw the pre-dominant form of…
A: A titration curve is formed when the pH of the sample solution changes with the addition of alkali…
Q: I. Apply your knowledge on the basic structure of amino acids by creating a polypeptide chain that…
A: The amino acids are the basic structural unit of proteins that contain an alpha carbon which contain…
Q: 1. Draw the condensed structural formulas for A and B. [0] [0] CH3 A B CH,-C I-CH,-C-OH
A: We are authorized to answer one question at a time since you have not mentioned which question you…
Q: 1. Label the "C" monosaccharide 2. Draw the open chain Fischer projection formula of the…
A: In the given molecule the different sugars are attached with the help of glycosidic bonds.
Q: 9. Identify the monosaccharides and their linkages in the following disaccharide. HOCH, но 0-CH он…
A: A carbohydrate is a biomolecule composed of carbon, hydrogen, and oxygen. Carbohydrates are…
Q: The C-C bond angles for the ring in M2 is: 60º 109.5º 120º 180º
A: Asked : C-C bond angles for the ring in M2
Q: 29) Starch is composed ofe A) brangha .
A: C) Branched amylopectin and unbranched amylose.
Q: Which of the following is the correct structure for the compound (S)-3-methylheptane. II II IV ON
A: 3-methylheptane is C8H18. So because of that option III and IV is clearly rejected
Q: 3. What ingredients are likely to contribute to the largest amount of saturated fat? b. How could…
A: The answer to the above question is given below.
Q: . The sugars glucose, fructose, and inositol all have the same formula i.e. C6H12O6 yet they have 3…
A: Glucose is the main type of sugar in the blood and is the major source of energy for the body's…
Q: 6. Convert each of the following chair conformations to an open-chain from and to a Fischer…
A: Carbohydrates are the polyhydroxy ketones or polyhydroxy aldehydes or their derivatives. In…
Q: 2. The structure of a vitamin is shown below. H HH H. H H C Č-H H. но нн Identify two potentially…
A: Elements are arranged according to the atomic numbers. Atomic masses, as well as numbers, increase…
Q: 1) Below you are given the structures of the disaccharides lactose and trehalose. но OH OH OH но но…
A: Lets first assume that all the carbohydrates given here are D isomers , cause that the general case…
Q: 5 Are the pairs of structures shown below constitutional isomers, enantiomers, diastereomers, or the…
A: Isomers: They are chemical compounds with the same molecular formula but different structural…
Q: 8. What kind of linkage is shown in the disaccha ОН ОН OH HO НО OH ÓH Α. α (1> 4) B. a (1 → 6) С.…
A: Saccharide is the basic structural unit of complex sugars or carbohydrates. These sugars or…
Q: 1. At pH 7, the net charge of lysine is А. -2 В. -1 С. 0 D. +1 Е. +2
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 4. Below is the structure of a disaccharide. ÇH2OH a. Give the systematic name of the disaccharide…
A: There are many biomolecules are found in the human body. Carbohydrates, lipids, protein, nucleic…
Q: 1. Draw the structure of the following triacylglycerols (TAGS) 1,2-dioleoyl-3-myristyl-sn-glycerol…
A: Triacylglycerol: Triacylglycerides are esters of fatty acids that are hydrolyzed to glycerol and…
Q: 1. provide three reasons why most of the research on carotenoids concentrates on b-carotene.
A: *NOTE: Kindly repost for other question Dear Student as per the guidelines we are supposed to answer…
Q: Different amino acids are distinguished by
A: Amino acids are small organic molecules which are the building blocks of proteins. These are…
Q: 4. This question is about the effect of geometric isomerism on the melting and boiling points of…
A: Cis-1,2- dichloroethene - Polar (i) Same groups(Cl) are present on the same side : So, net dipole…
Q: 18. Which of the following characteristics are found in the class of C20 carboxylic acids called…
A: Prostaglandins, thromboxane, as well as leukotrienes, are known to be derived from the essential…
Q: 6-An example of a hydrolysis reaction is * a.dipeptide + H20 → amino acid + amino acid O b.…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 7. Convert the following Haworth projections to a Fischer projection. Name the monosaccharides you…
A: Carbohydrates are the polyhydroxy ketones or polyhydroxy aldehydes or their derivatives. In…
Q: 5. Give the molecular formula of the functional group that is missing. H H,N-C-? R a. NH+ b. CH3 с.…
A: First, we will analyze the given structure and decide which can be the functional group. There is an…
Q: 9. Write the tripeptide structure for: val-ser-cys. 10. Write the reactions for: phe + ser
A: Amino acids are the building blocks of proteins. whereas each amino acid consists of an amino group,…
please answer question
Step by step
Solved in 3 steps

- _______ groups are the acid part of amino acids and fatty acids. a. Hydroxyl (OH) b. Carboxyl (COOH) c. Methyl (CH3) d. Phosphate (PO4)Lactose is a disaccharide formed by the formation of a bond between glucose and glycosidic; lactose glycosidic; galactose hydrogen; sucrose hydrogen; fructose1. Construct the two enantiomeric forms/structure of the following monosaccharides and designate the handedness of each using D, L system 1. Ribulose
- A monosaccharide that consists of 5 carbon atoms, one of which is in a ketonegroup, is classified as a(n) ________. Question 15 options: aldohexose ketopentose ketotetrose aldopentose aldotetrose2. The C-C bond angles for the ring in M2 is: 60º 109.5º 120º 180º1. What is the identity of the monosaccharide components of the tetrasaccharide: A. B. C. D. 2. Give the Haworth structure of the tetrasaccharide
- 22. All of the following are examples of disaccharide, EXCEPT: * A. maltose B. sucrose C. raffinose D. lactose2. a. What ingredient(s) necessary to white chocolate does this product have? b. Why is this product labeled as “white chocolate style” instead of being simply labeled as “White chocolate”?I. True or False. Write the word “reducing” if the statement is correct, otherwise write “non-reducing”. ____________1] Mutarotation affects the reducing property of carbohydrates. ____________2] Anthrone test can be use to test for the helical configuration of a polysaccharide. ____________3] Upon treatment with phenylhydrazine reagent & hydrolysis, maltose gives glucuronic acid and glucosazone. ____________4] Xylose gives a positive result with Molisch’s, Seliwanoff’s, & Bial’s-Orcinol tests. ____________5] Aniline-acid oxalate can be use as a mobile phase for the development of the chromatogram. ____________6] Ethanol (95%) & 10% TCA are used to precipitate glycogen from the solution. ____________7] Salivary amylase can be use to hydrolyze 1,4 glycosidic bonds of cellulose. ____________8] Lobry de Bruyn-van Ekenstein Transformation take place when a sugar is treated with a strongly basic reagent. ____________9] Ribulose yields 5-hydroxymethylfurfural upon treatment with…
- 1. Define and give there functions a. Carbohydrates b.polysaccharidesWhat is unusual about the structure of N-acetylmuramic acid (Figure 16.18) compared with the structures of other carbohydrates12. The sugars glucose, fructose, and inositol all have the same formula i.e. C6H12O6 yet they have 3 very similar but different structures. This is an example of: a) geometric isomers b) structural isomers c) stereo isomers d) none are correct



