Q: What is the IUPAC name for the following molecule? HO H3C A) 4-methoxy-5,5-hexadiol B)…
A:
Q: HO H H-C-C-CH₂OH NHCCHCI₂ Ö NO₂ chloramphenicol
A: R-S nomenclature: R and S system of nomenclature is a naming of absolute configuration to chiral…
Q: From the Eº calculate Ksp for: a- Ag2CrO4 if Ag2CrO4 +2e- → 2 Ag+ + CrO42- Eº= 0.446v and Ag+ e- →…
A:
Q: A solution of 2.50 g of so What is the molar mass deally (d of H₂O at 25°C g/mol
A:
Q: In the reaction 3 Ms-¹. i. What is the rate of reaction with respect to B? ii. What is the rate of…
A:
Q: A student ran the following reaction in the laboratory at 314 K: 2NOBr(g) =2NO(g) + Br₂(g) When he…
A: Given, 2NOBr(g) ⇌ 2NO(g) + Br2(g) Initial partial pressure of NOBr(g) = 0.430 atm At equilibrium…
Q: Write the coordination number and oxidation state of Platinum in the complex [ Pt ( en ) , C1₂ ]
A:
Q: A sample of pure CaCO3 weighing 0.2428g is dissolved in HCI acid and the solution diluted to 250.0ml…
A:
Q: Identify the structure of the important fragments. Show the arrow pushing mechanism to form the…
A: To find: The absorption, appearance, and compound for the IR spectra Statement: The stretching and…
Q: Identify the structure of the important fragments. Show the arrow pushing mechanism to form the…
A:
Q: (250+2) Sample cell calculations : Mmo01.3mL Fraze (need) (stock) а така им enzyme fatty acid 120 MM…
A:
Q: evise an acid/base extraction method capable of separating Eugenol from affeine. The pKa of eugenol…
A:
Q: Draw the products formed when 1-chloro-1-methylcyclopentane is treated with NaOMe in MeOH.
A: Here we have to determine the products formed in the following given reaction. Here the mechanism of…
Q: How many grams of solid sodium cyanide should be added to 1.00 L of 0.116 M hydrocyanic acid…
A: Given-> Volume of HCN = 1.00 L Molarity of HCN = 0.116 M pH = 9.990 Ka(HCN) = 4.00 × 10-10
Q: Examine the following pairs of compounds and determine which compound in each pair would be most…
A: 1. RO- is more basic than RS- 2. RO- is more basic than ROH 3. S- attached to saturated carbon is…
Q: What is the relation between the two molecules shown below? Darvon, a pain-relieving opioid A) The…
A:
Q: Please draw a more stable resonance form for each of the following, and explain why it is more…
A:
Q: Balance the following equation Cu + O₂ - Cu₂O
A:
Q: H H Br Br Br Acetona/agua Reflujo Acetona/agua Reflujo MeOH Reflujo + NHƠN + NHƠN + MeONa + CH₂SNa…
A: These reactions are based on elimination and Substitution Mechanism depending upon the reagent…
Q: Calculate the Ksp value for a solution of magnesium fluoride. The molar solubility of magnesium…
A:
Q: When a 28.6 mL sample of a 0.394 M aqueous hypochlorous acid solution is titrated with a 0.481 M…
A: The solution is given below -
Q: Suppose a 250. mL flask is filled with 2.0 mol of 1₂ and 1.0 mol of HI. The following reaction…
A:
Q: Make an inference about the effect of applied current and electrolysis time on the mass of deposited…
A: To make a conclusion on the effect of change in the amount of applied current and the time for which…
Q: (a) The discharge reaction for a zinc-manganese dioxide battery can be represented as: Zn(s) + 2MnO₂…
A: Given, Reaction : Zn(s) +2MnO2(s) →ZnO(s) +2Mn2O3
Q: What is the correct structure for 1-bromo-2-ethylcyclopentane? Br b) a) CH₂CH3 c) CH₂CH3 Br Br…
A:
Q: The rate law for the reaction below is rate = k[H2]x[O2]y 2 H2 + O2 → 2 H2O Which of the…
A: Rate law expression explains relationship between rate of reaction and concentration. Powers of…
Q: The OH radical reacts with itself in a disproportionation type reaction according to the reaction:…
A: we can predict the order of the reaction by seeing the unit of the rate constant of the reaction.…
Q: Using the equation C12H22O11+12O2-->12CO2+11H2O if 49.8 L of O2 are consumed, how many moles of…
A: Given, C12H22O11 + 12O2 --> 12CO2 + 11H2O Volume of O2 reacts = 49.8 L moles of sucrose…
Q: a group 2 metal hydride MH2 (2.000 g) reacts with water to give a basic solution and 0.1520 moles of…
A: Group 2 metal hydride MH2 and it is completely react with water and give H2 . MH2 + H2O…
Q: How many moles of sodium chloride can be produced if 1.2 mol Na reacts with .85 mol of Cl2?
A: Here, we have to find the number of moles of sodium chloride that can be produced if 1.2 mol Na…
Q: Question 8 Assign the R or S configuration for two stereocenters in the following molecules H H CI…
A:
Q: Which of the labeled carbons in 2-chloro-4-ethyl-1-methylenecyclopentane are chiral centers? CI (II)…
A: A chiral centre is the carbon having 4 different groups surrounding it on its 4 valency. Let us…
Q: The surface tension of water at 20°C is 72.7 mN m and its density is 0.998 g cm" Assuming a contact…
A: Given that, at 20oC The surface tension of water is γ = 72.7 mN/m = 0.0727 N/m = 0.0727 kg/s2. The…
Q: what would be the percent yeild for the reaction?
A:
Q: Rank the following from weakest (1) to strongest (4) base. A) B) C) H₂O D) 00
A:
Q: Consider the reaction PO + DONG → PODONG, determine the reaction order for each component and the…
A: Here we are required to find the time needed to form PODONG having concentration 0.324M
Q: In the Hall-Heroult process, a large electric current is passed through a solution of aluminum oxide…
A: Amount of pure aluminium formed can be calculated from the moles of charge passed.
Q: Module 2b: How much NaOH (in g) will * be needed to form a 1000L 100 ppm solution? 10 O 100 O 1000…
A: Given, Note: 1 ppm = 1 mg/L Concentration of the solution = 100 ppm = 100 mg/L Volume of the…
Q: 21- The mass-action effect is a shift in the position of an equilibrium caused by adding one of the…
A: Equilbrium is an condition in which products and Reactants are at state of dynamic Equilbrium.
Q: 226, 88 A radium source contains 50 mg of 238Ra in equilibrium with all its progeny. Assuming 1/2 is…
A: The given radium source contains 50mg of 226 Ra88 in a equilibrium. T1/2= 1602 years.
Q: Be sure to answer all parts. A solution of 2.01 g of anhydrous aluminum chloride (AlCl3) in 54.46 g…
A:
Q: Which of the following is/are meso? Br Br Br Br I A) I B) II C) III D) IV E) I and II F) I and III…
A: Meso compounds are those compounds that are optically inactive due to the presence of a plane of…
Q: NOTES ASK YOUR TEACHER The Shroud of Turin, which shows the negative image of the body of a man who…
A:
Q: determine the electron count of an octahedral Ti^3+ complex. Calculate the crystal field splitting…
A: Crystal field splitting energy is defined as energy difference between two set of orbital t2g and eg
Q: Which intermolecular force is generally considered the weakest? O Covalent bonding O London…
A: In this question, we will see that which force is a weakest force between the molecules or atoms.…
Q: Draw the lewis structure for Na2Cl₂??
A:
Q: Using Curved Arrow Formalism, draw the reaction of 1-methylcyclopentanol treated with aqueous…
A:
Q: 9. Nitric oxide, NO, reacts with hydrogen to give nitrous oxide, N,O, and water. 2NO+H₂> N₂O + H₂O…
A:
Q: What is the action of the following reaction LAVORO O A
A: Oxymercuration reaction: Hydration of alkene proceeds by an electrophilic addition reaction…
Q: If the density of Carbon tetra chloride is 1.59 g/mL, what is the volume in L of 2.59 kg of carbon…
A:
Step by step
Solved in 4 steps with 3 images

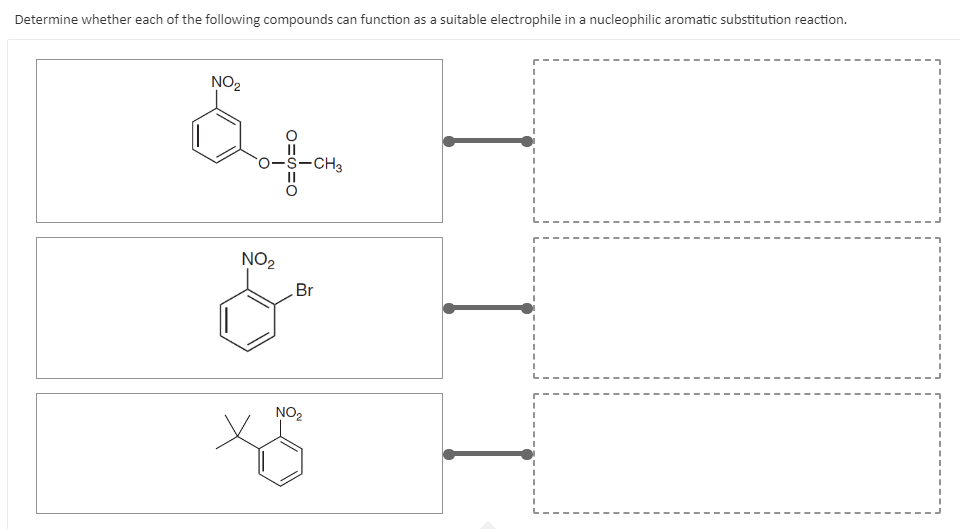
- Which of the following are most likely to behave as electrophiles, and which as nucleophiles? Explain.(a) NH4(b) CqN (c) Br (d) CH3NH2 (e) HOCqCOHWhich of the following can behave as an electrophile? A. HBr B. Cl- C. CH3O- D. CH3CH2CH2CH3Which of the following bases would be able to irreversibly deprotonate butanal? 1. NaOH 2. H2O 3. NaOCH3 4. More than one 5. None
- Which of the following compounds will NOT undergo nucleophilic substitution reaction? isopropanol Benzoyl chloride propylene oxide butanoneWhat direction does the nucleophile approach the substrate in an SN2 reaction?What effect and orientation does -OH group exhibit in electrophilic aromatic substitution reaction ?
- Are the following nucleophiles, electrophiles, they be both a nucleophile or electrophile, or neither? pyridine alkenes Hg+ (ex. Hg(OAc)2 CH3ONa PBr3Which of the following could act as an electrophile? A) CN^- B) NH2^- C) NH3 D) H3O^+Draw the products of each of the following reactions and indicate whether the reaction occurs by: a) Aromatic electrophilic substitution, b) Aromatic nucleophilic substitution: Addition elimination, c) Aromatic nucleophilic substitution: Mechanism of benzene:
- The –OH, -OR, -NH2 groups cannot be substituted directly by electrophilic substitution on the aromatic ring, but can be done by substitution of a diazonium group. Design the synthesis of p-nitro aniline compoundsName and Draw the structures of all possible chemical (Electrophilic aromatic substitution) reactions of the o-nitrobenzamide compound : Halogenation (Chlorination or Bromination) Nitration Sulphonation Friedal Craft Alkylation Friedal Craft AcylationWhich of the following compounds is most reactive towards nucleophilic addition reactions?

