nur ersion The following question is on ecuilibria What is the meaning of 7) a) equilibrium state? ii) position of equlibrium? mich s) Give one factor which can alter the magnitude of an equilbrium constant for a particular reaction. b) In order to determine the equilibrium constant for esterification, a mixture of 01 mole ethanol and 0.1 mole ethanoic acid were allowed to reach equilibnum. At equilibrium samples woro taken, frozen and analysed, and the amount of acio left unreacted was found to be 0.033 mole. entaly c) CH,CO;H(I) + CH.CH,OH() CH,CO,CH,CH )+H0() i) suggest qa ethod that could be used for analyzing the sample mixture. why was each sampte frozen before analyzing? what will be the effect on the posilion of equilibrium of adding a title ethaniuc acid? Explain why you do not need to know the volume of tne mixture to calculate the equilibrium constant, K. iv) ulation v) what ia the compoisiton of the oquilibrium mivture? lowing vi) Calcuiate the value of the equilibrium conslont K for this reaction, Phenolohthalein indicator which can be represented as HPh is a weak acid with a dissociation constant ka = 10, and whose corjugate base is coloured pink write the equalion of the dissociation of pheniolphthalein in water d) ks) what is a weak acid?. identify the conjugate base of phenolphthalein in) colo iv) calculate the ratio of the number of pink particles to colouriess particles of this indicator in a solution whose plH is 9. Underine the species acting as an acid in the left hand side of oach of the follewing reactions. nicn e) ks) HSO, (aq) + HO'(aq) H;SO.(egi H,O) ) NH, + BCl, -HNBC, Cid (Total = 16 marks) ks) CGCEB JUN 2004 O. 6
nur ersion The following question is on ecuilibria What is the meaning of 7) a) equilibrium state? ii) position of equlibrium? mich s) Give one factor which can alter the magnitude of an equilbrium constant for a particular reaction. b) In order to determine the equilibrium constant for esterification, a mixture of 01 mole ethanol and 0.1 mole ethanoic acid were allowed to reach equilibnum. At equilibrium samples woro taken, frozen and analysed, and the amount of acio left unreacted was found to be 0.033 mole. entaly c) CH,CO;H(I) + CH.CH,OH() CH,CO,CH,CH )+H0() i) suggest qa ethod that could be used for analyzing the sample mixture. why was each sampte frozen before analyzing? what will be the effect on the posilion of equilibrium of adding a title ethaniuc acid? Explain why you do not need to know the volume of tne mixture to calculate the equilibrium constant, K. iv) ulation v) what ia the compoisiton of the oquilibrium mivture? lowing vi) Calcuiate the value of the equilibrium conslont K for this reaction, Phenolohthalein indicator which can be represented as HPh is a weak acid with a dissociation constant ka = 10, and whose corjugate base is coloured pink write the equalion of the dissociation of pheniolphthalein in water d) ks) what is a weak acid?. identify the conjugate base of phenolphthalein in) colo iv) calculate the ratio of the number of pink particles to colouriess particles of this indicator in a solution whose plH is 9. Underine the species acting as an acid in the left hand side of oach of the follewing reactions. nicn e) ks) HSO, (aq) + HO'(aq) H;SO.(egi H,O) ) NH, + BCl, -HNBC, Cid (Total = 16 marks) ks) CGCEB JUN 2004 O. 6
Chapter87: Steam Distillation
Section: Chapter Questions
Problem 4P
Related questions
Question
Question 7 (c) (i), (ii), (iii), and c(iv)
https://www.dropbox.com/s/hz1ic5jjr56ces1/Q7CH_rotated_page-0001.jpg?dl=0
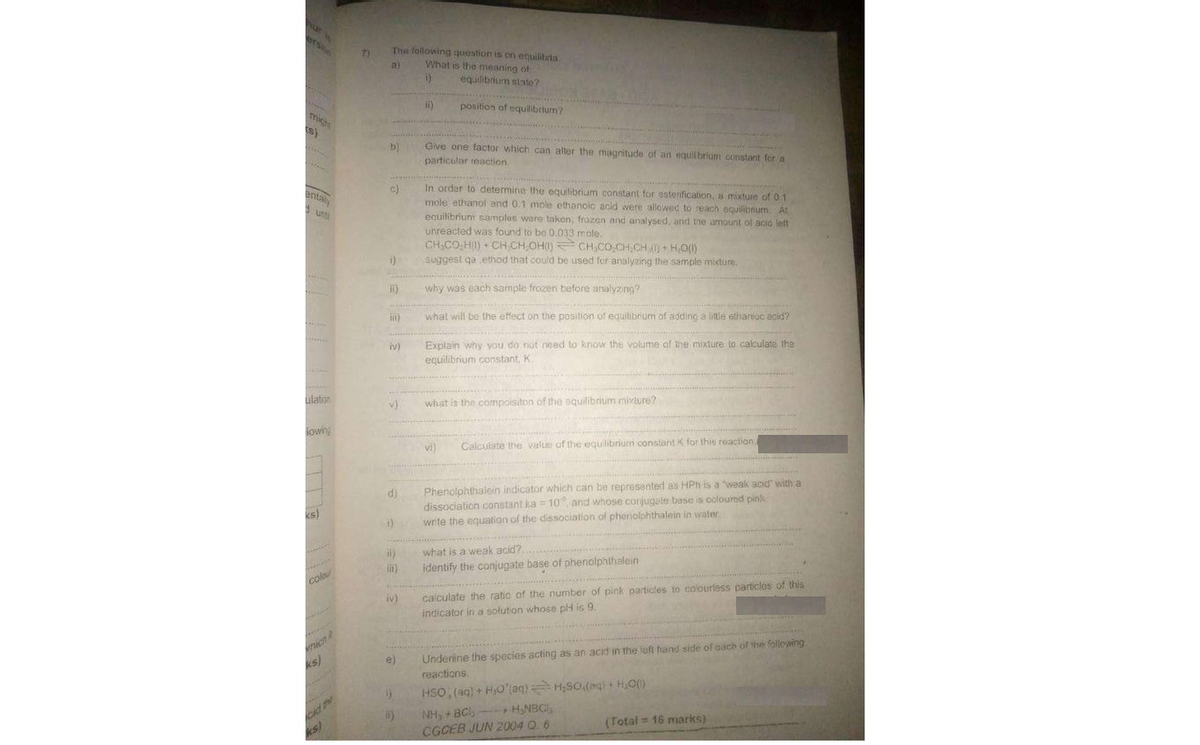
Transcribed Image Text:nur
ersion
The following question is on ecuilibria
What is the meaning of
7)
a)
equilibrium state?
ii)
position of equlibrium?
mich
s)
Give one factor which can alter the magnitude of an equilbrium constant for a
particular reaction.
b)
In order to determine the equilibrium constant for esterification, a mixture of 01
mole ethanol and 0.1 mole ethanoic acid were allowed to reach equilibnum. At
equilibrium samples woro taken, frozen and analysed, and the amount of acio left
unreacted was found to be 0.033 mole.
entaly
c)
CH,CO;H(I) + CH.CH,OH() CH,CO,CH,CH )+H0()
i)
suggest qa ethod that could be used for analyzing the sample mixture.
why was each sampte frozen before analyzing?
what will be the effect on the posilion of equilibrium of adding a title ethaniuc acid?
Explain why you do not need to know the volume of tne mixture to calculate the
equilibrium constant, K.
iv)
ulation
v)
what ia the compoisiton of the oquilibrium mivture?
lowing
vi)
Calcuiate the value of the equilibrium conslont K for this reaction,
Phenolohthalein indicator which can be represented as HPh is a weak acid with a
dissociation constant ka = 10, and whose corjugate base is coloured pink
write the equalion of the dissociation of pheniolphthalein in water
d)
ks)
what is a weak acid?.
identify the conjugate base of phenolphthalein
in)
colo
iv)
calculate the ratio of the number of pink particles to colouriess particles of this
indicator in a solution whose plH is 9.
Underine the species acting as an acid in the left hand side of oach of the follewing
reactions.
nicn
e)
ks)
HSO, (aq) + HO'(aq) H;SO.(egi H,O)
)
NH, + BCl, -HNBC,
Cid
(Total = 16 marks)
ks)
CGCEB JUN 2004 O. 6
Expert Solution
introduction
The kinetics of a reaction can be studied experimentally.
The equlibrium concentration of a reaction can be calculated from these analytical techiques, such that order of reaction can also be determined.
For these experiments we have to know the accurate concentration of the reactants.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole