Q: A 100 mL of 0.125 M CH₂COOH solution was shaken with 1.2 g charcoal for 5 min. After filtration, 10…
A: I have given a detailed solution for the problem as follows:
Q: Give hand written explanation Asap Thanks 1.Calculate the mass of water produced when 3.52 g of…
A: Given, Butane reacts with excess oxygen to produced carbon dioxide and water if 3.52 g of butane…
Q: The rate of a certain reaction is given by the following rate law: rate=k = K[N₂] [H₂]² Use this…
A:
Q: A 1.00 L flask initially contains 4.40 mol of HI at a certain temperature. The K at this temperature…
A:
Q: Question 20 of 20 Potassium permanganate solutions used in oxidation-reduction titrations are often…
A:
Q: I Ш NH3 П IV (CH3)2NH NH₂
A: Basicity depends on the availability of lone pair of electron on Nitrogen atom. Greater the…
Q: 1 10 O O TRS MKR SRH 4. Junghwan wanted to determine which of the following skin care product which…
A: Paper chromatography is used to separate different chemical compounds based on their different…
Q: a reaction has the experimental rate equation: Rate= k [A] [B]^2. how will the rate change if the…
A: Let's say, we have the following hypothetical reaction: A + B →P Here, A and B both are reactants…
Q: ncreasing the concentration of the salt in a colloidal system will: a) Increase the radius of sheer.…
A: Given in following question a concentration of the salt in a colloidal system will increase the .…
Q: Which of the four structures is consistent with the 1H-NMR spectrum shown below? t, 2H 5 (a) (c) 4…
A:
Q: The structure of the alkaloid tryptamine is shown below. Encircle the more basic nitrogen atom.…
A: Answer : -NH2 nitrogen is more basic in nature than indole nitrogen. Short explanation : basicity…
Q: A student is examining a bacterium under the microscope. The E. coli bacterial cell has a mass of m=…
A: Data given: The mass of bacterial cell, m = 1.10 fg = 1.10×10–15 g (1 fg = 10–15 g) m = 1.10×10–18…
Q: Complete the reactions below by providing the predicted products. Where a reaction is not feasible,…
A: The Grignard reagent reacts with a ketone to form tertiary alcohol. The reaction is proceeded by the…
Q: 3. Calculate the hydronium ion concentration of a solution with a pH of -1.07.
A:
Q: separation
A: According to the question, we need to determine how to purify the black copper for the wire using…
Q: The absorbance of 1.48 x 105 M methyl red solution, prepared in 0.1 M HCI, was measured using a 1.0…
A:
Q: Mechanism of separation is partitioning SEC OIEX HILIC ONPLC
A: Chromatography is the laboratory technique for the separation and isolation of a variety of samples…
Q: Is the reaction of zinc and sulfur a redox reaction? Write a complete, balanced chemical equation.If…
A: Redox reaction : A reactions in which one species undergoes oxidation whereas other gets reduced…
Q: Draw the Lewis structure (3 resonance structures) and determine the formal charges on each atom for…
A: Given that, CO32- Write formal charge on each atom and draw the resonance structure ?
Q: Table 1. Results of the solubility tests for C4H6O2. SOLUBILITY TEST distilled water Litmus paper…
A:
Q: 9. A suspension of milk of magnesia, Mg(OH)2, is analysed by a back-titration. A 1.0391 g sample is…
A: In an acid-base reaction salt and water are formed.
Q: Select all of the solutions from the first part of the experiment that you would expect to be acidic…
A:
Q: N Polymer H H HH H N Monomer 4. CH₂CHCOOCH, 7. Addition 5. CH₂CH=CH₂ 8. Condensation COM COM COH CO…
A: Polymer : The molecule made up of repeating units is called as polymer . This units is called as “…
Q: The PH Vale off0.025 ±0.002) and di -3 #cl solubionis -±--
A:
Q: What is the product to the reaction below? NO₂ He at NO₂ b. Select one: Oa. Structure a O b.…
A:
Q: An oxidizing agent undergoes a/an ________ in oxidation state
A: In this question, we will see that an oxidizing agent is undergoes reduction or oxidation in…
Q: 3. Predict the major and minor products expected for the following elimination reactions. OH HCI Хон…
A: All the reactions are shown below.
Q: In the flame test if two ions have similar colors what could help you distinguish one from the…
A: Flame test provide preliminary idea about the presence of metal ions. To further confirm this we…
Q: Can be applied for the separation of ionic (nonprotein) analytes
A: Ans: IEX
Q: Tip: Add curved arrows in this sketcher to represent the electron flows for the next mechanism stop…
A: The given reaction is SN1 reaction. In SN1 reaction in the first step a stable carbocation is…
Q: Carbon uses [orbitals] hybrid orbitals in the molecule below. :CI-C=N: 1—C=N:
A: The hybridization of carbon is given below
Q: Name the coordination compound KING (CN) 3 (OH)3]
A:
Q: Cu (s) + HNO3 (aq) right arrow Cu(NO3)2 (aq) + H2O (l) + NO2 (g) what is the…
A:
Q: ve been struggling with these questions for my homework although I put my answers on them I have to…
A:
Q: 20 ml 01 M HNO3 SA 2 mg NaOH SB 10mi 0.1 M H₂CO3 u 0.15m Na₂CO3 WB 5ml Find PH na₁ = 10-3 192= 107
A:
Q: Given Some acid, choose the weakest acid a (1) butyric acid (ka = 10 5x10-5) (ii) ascorbic acid (ka…
A:
Q: 1. 4Fe + 30₂ 2Fe2O3 Oxidation Reaction: Reduction Reaction: Reducing Agent: Oxidizing Agent: 2. Cl₂…
A: Oxidation reaction : Increase in oxidation number. Reduction reaction: Decrease in oxidation number.…
Q: Q7. What is the correct Lewis structure for the molecule in the box, including the formal charge(s),…
A:
Q: Propose a reaction scheme for the synthesis of the following ethers through the Williamson…
A: Here we are required to propose the reaction scheme for the synthesis of ether
Q: A. Use Bohr's equation to calculate how much energy (in J) is needed to promote an electron from the…
A:
Q: QUESTION 8 How much heat is needed for the following process for Ag, silver? The melting point of…
A: Here we have to determine the amount of heat required to convert 1 mole of Ag (s) at 25°C to Ag(l)…
Q: MISSED THIS? Read Section 146 (Pas 601 63 Watch KGV 14.6 A solution containing 27 25 ung of an…
A: The osmotic pressure of a compound is related to the concentration and temperature as: π=c×R×T…
Q: same compounds. and (a) 0-H O +0-H (c) and 0- CH₂-C-H (e) CH₂=C-H and CH₂-C-H
A:
Q: A block of iron (density = 7.87 g/cm³) has dimensions of 4.25 cm x 5.80 in x 1.48 ft. Calculate its…
A: Given, The density of a block of iron is 7.87 g/cm3, The mass of block of iron when it has…
Q: What is ∆Gο at 25° for the reaction: 2 CO (g) + 2 H2 (g) → CO2(g) + CH4(g), given that at 25° ∆Hο =…
A: Recall data, ∆H°=-247.3 KJ mol-1 ∆S°=-256.5 J mol-1K-1= -0.2565 KJ mol-1K-1 ∆G°=?
Q: Which species are present in aqueous solution of NH? 1. NH3 I. NHƯ III. H30*
A:
Q: (a) A sample of xenon gas at a pressure of 865 mm Hg and a temperature of 36 °C, occupies a volume…
A:
Q: tetraphenylboron
A: We know that, sodium tetraphenylboron Na(C6H5)4B, reacts with potassium ions to form a white…
Q: A solution is made by dissolving 23.4 g of potassium sulfate, K₂SO4, in enough water to make exactly…
A: Given, The mass of K2SO4 = 23.4 g. The volume of the solution = 500. mL The molarity of K2SO4 , K+…
Q: QUESTION 22 Identify the balanced reaction for the complete combustion of propanal (C,H,OH). Select…
A: Combustion reaction is the reaction of organic compound with oxygen to form carbon dioxide and…
How to draw product C? Please give detailed structure for the product C
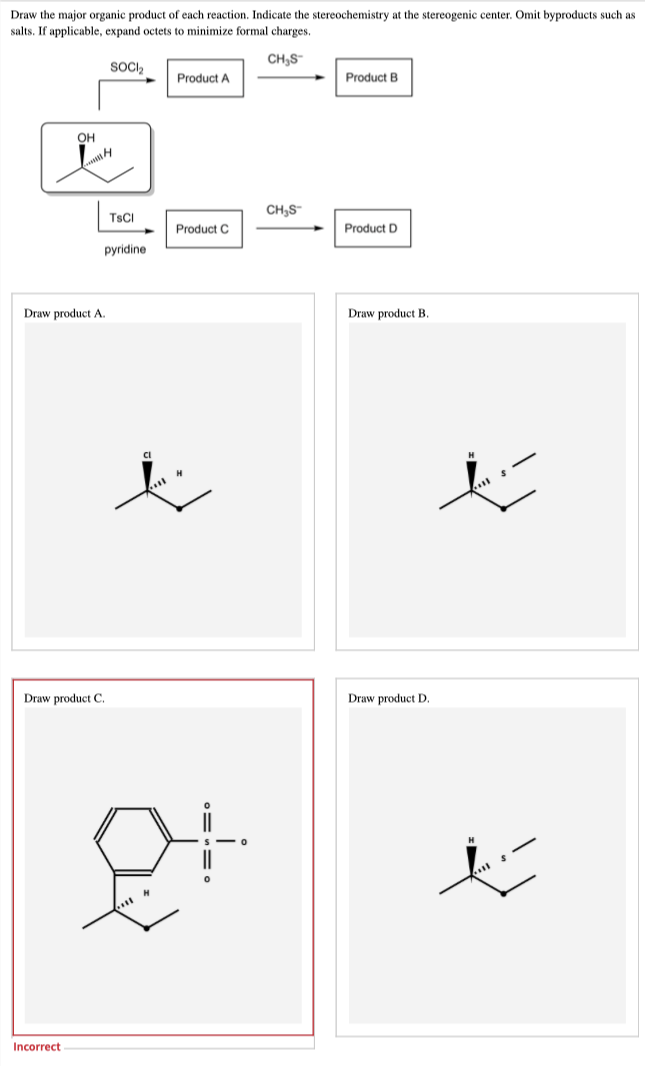
Step by step
Solved in 3 steps with 2 images

- The reactants, intermediates, final products, and all curved arrows showing bonds forming andbreaking are collectively referred to as the mechanism of a reaction. For the following reactants: a. Explain why the original statement of Markovnikov’s rule does not help in this case, but themodern restatement of Markovnikov’s rule tells you which carbon will get the X (Cl). b. Show the mechanism of the most likely addition reaction between the reactants.O chem - please break down each step as much as possible, draw all possible products and consider stereochemistryIf reacted with Cl2 how many distinct mini chlorinated products will be present? How many monocholtinated products will form at 1 and 2? Which Carbon will be attached to a Cl in the major product?
- Fill in the missing structure(s) for the reaction pathways shown below. Provideall starting material(s), reagent(s), or product(s). If more than one product can beformed, draw all major products. If more than one starting material or reagent ispossible, write all starting materials or reagents. Include stereochemistry in youranswer, if applicable, and draw all stereoisomers. Don't forget about resonance.o chem - please show stereochemistry and break down each step as much as possibleFill in the missing structure(s) for the reaction pathways shown below. Provideall starting material(s), reagent(s), or product(s). If more than one product can beformed, draw all major products. If more than one starting material or reagent ispossible, write all starting materials or reagents. Include stereochemistry in youranswer, if applicable, and draw all stereoisomers.
- For eaach image, predict the MAJOR product(s). Show stereochemistry where applicable and draw out ALL stereoisomers formed (as major products) in each reaction. State the mechanism(s) by which the major product(s) are formed (SN1, SN2, E1, and/or E2). Reagents are NOT present in excess.(need answer in step by step) Deaw major product of followinv reavtion, make sure to draw enantiomers if expectedHello, could someone help me with this practice problem? thx in advance (: For the major product of this reaction, draw the perspective diagram of the major products. The mechanism isn't necessary, just the products. If appropriate, add an enantiomer where required. Lastly, state the stereochemistry of the process (syn, anti or none).
- For each section, circle the mechanism from the two options given (SN1 or SN2) and draw the main organic product resulting from that mechanism. Indicate the stereochemistry and if two configurational isomers form, draw both.1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.

