Q: Predict the major organic product of the following reaction ^ Discuss key changes in the spectra…
A: Kindly see the attachmentExplanation:Approach to solving the question: Detailed explanation:…
Q: The three mechanisms for aromatic substitution differ in the intermediate, the leaving group, and…
A: The objective of the question is to verify the truthfulness of the statement that the three…
Q: 3. An alkene can be converted into an ether in the three mechanistic steps shown below. Complete…
A: Ethers are organic compounds that can be represented using a general formula, R-O-R', where, R and…
Q: What is the COD and ThOD of a 2.5 mM solution of C40H81O21N8P2S ?
A: The objective of the question is to calculate the-Theoretical Oxygen DemandChemical Oxygen Demand
Q: What is the stereochemical relationship of the 2 following compounds? OH HO HO HO HO. OH HO HO…
A: Stereochemical relation of following molecules are as follows.
Q: Rank the contributions of the resonance forms for the following compounds by selecting the…
A: Allylic carbocations:The positive charge is equally distributed between the three atoms, This means…
Q: 2) For each pair of molecules, CIRCLE the one with the lower pKa value for losing the indicated…
A: The acidity of a compound is related to its tendency to donate H atoms as H+. The higher tendency to…
Q: Name the following
A: The objective of this question is to predict the name of the given organic compounds.The process of…
Q: Identify the alkyne. O a. CH3CH2CH2CH3 O b. CH2CH2 CH2CH2 O c. CH3CHCHCH3 Od. CH3CCCH3 Clear my…
A:
Q: Indicate the number of carbon environments present in the molecule shown. Record your answer as a…
A:
Q: Draw the Lewis structure for the molecule CH3CH2CCH. How many sigma and pi bonds does it contain? A.…
A: The correct answer is: (E) 9 sigma, 2 piExplanation:The Lewis structure of a molecule is used to…
Q: This energy diagram shows the allowed energy levels of an electron in a certain atom. (Note: the SI…
A: Final answer = 3.755Explanation:
Q: Rahulchemistry
A: pH = 9.14 Explanation:Step 1:.
Q: Do not give handwriting solution.
A: The Detailed steps attached belowExplanation:
Q: 4. For each of the following, determine if either a (a) hydride shift or (b) methyl shift has…
A: “Since you have posted a question with multiple sub parts, we will provide the solution only to the…
Q: Example 6 An imine is a molecule that contains a carbon – nitrogen double bond. Describe the bonding…
A: See the answer in the explanation fieldExplanation:
Q: Complete the following for the below reaction. i. draw the complete Molecular Orbital diagram with…
A: The objective of this question is to predict the outcome of a cyclization reaction by applying…
Q: DETERMINE molecular structure of PTFE
A: The objective of the question is to determine the molecular structure of PTFE…
Q: 16 What is the product of the following reaction? MeOH acid heat MeO OMe O OMe OMe MeO. 11 Multiple…
A:
Q: A solution of HCl with a volume of 25.00 mL is titrated to the endpoint, with 0.250 M NaOH. If it…
A: Given:HCl (aq) + NaOH (aq) H2O (l) + NaCl (aq)Concentration (M1) of NaOH = 0.250 MVolume (V1) of…
Q: 1. Complete the following reactions by providing any missing components. Provide all products formed…
A: Given are organic reactions. Reaction a is oxidation reaction. Reaction b is nitration reaction…
Q: Give the IUPAC name for each compound. Part 1 of 2 Part 2 of 2 ︽﹀ » CH, CH, H,C=C=C=C—C—CH, CH; CH;
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: Please provide IUPAC names of parts - d , f, g, h, and i in details
A: Information about the question
Q: can this be written on paper please
A: ∆Hrxn=−1276 kJ/molThe reaction is exothermic.Explanation:Since is negative so the reaction is…
Q: Why is a mixture of four stereoisomers formed in this reaction? Draw the four products that form in…
A: We have to predict the products.
Q: A 1.00 mL aliquot of a hydrogen peroxide (H2O2) solution was added to 2.00 mL of acidified Ti(IV)…
A: The concentration (C) of the substance can be calculated based on the absorbance (A) value using the…
Q: 27 The addition of a catalyst to the reaction shown in the figure would change the value of For the…
A: The given diagram is that of a potential energy vs time for a reaction. A represents the energy of…
Q: Draw the product of the reaction shown below. Use wedge and dash bonds to indicate stereochemistry…
A: In the presence of light or heat, a conjugated alkene can undergo rearrangement to form a ring. Such…
Q: Which planet is known as the "Red Planet"? A) Mars B) Venus C) Jupiter D) Saturn Don't.
A: The objective of the question is to identify which planet is commonly referred to as the 'Red…
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Zn(s) + O2(g) → 2ZnO(s)…
A: Enthalpy change for a reaction is the amount that is evolved or absorbed by a reaction at different…
Q: Which of the following reagents can be used to convert the compound below into a carboxylic acid?…
A: The conversion of an alkyne to a carboxylic acid is a significant transformation in organic…
Q: 3. is? a. E,R b. E,S C. S,Z d. R,Z e. not a.-d.
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Propose synthetic routes to synthesize the target molecule OH Propose possible products formed in…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: HO H2SO4
A: The reactant is an alcohol. It is an organic compound. The reagent is sulfuric acid . It is used to…
Q: Identify the products formed in this Brønsted-Lowry reaction. HSO+HF acid + base acid: base:
A: Find out acid and base of the reaction.
Q: H₂C=CH2 ? 4 H3C Select reagents from the table to show how you would carry out this synthesis. CH3
A: Given is multistep organic synthesis reaction.The starting compound is alkene.
Q: Which one of the following statements describes partially compensated metabolic acidosis?…
A: The objective of the question is to identify the correct statement that describes the condition of…
Q: Give the systematic (IUPAC) names for these molecules. IUPAC name: 요 -OCCH2CH3 CH3OCCH2CH2CHCH3…
A: Given :Compound (I) :Compound (II) : Objective : Determine the IUPAC anme of the compound given.
Q: 1: For the HCI (2E,4R,5R)-4,5-dimethylhept-2-ene (3R,4R,5R)-3-chloro-4,5-dimethylheptane Question…
A:
Q: Part 2: A student carried out a synthesis as shown in the scheme below. A…
A: The amount in moles of reactants that completely react with each other to form a required number of…
Q: Predict the expected product for each reaction and provide IUPAC name for the correct starting…
A: The final answer attached belowExplanation:
Q: Combustion of glucose (C6H12O6) is the main source of energy for animal cells: CHO) +60, (e) 600,…
A: (0.00855501 g of glucose)Rounded to two significant figures0.0086 g of glucoseExplanation:Given the…
Q: provide mechanism
A: We have to predict the mechanism.
Q: HO OD 2 CH3CH2OH HO POH -P- OH + 2Н20 OH OH Incorrect
A: We will write the general reaction first of one ethanol. When we take one mol of ethanol the…
Q: H Η H H OH H NaOH, EtOH, reflux H2SO4
A: Given are organic reactions. The station compounds for reaction 1 are aldehydes and reagent is…
Q: When the following skeletal equation is balanced under basic conditions, what are the coefficients…
A: A redox reaction is balanced by balancing the mass and charge. Hence the number of atoms of all the…
Q: Draw the two major products obtained in the reaction shown. Br2 (one equivalent) CH2Cl2
A: Given compound is brominated in the presence of CH2Cl2
Q: 1.) Predict the major organic product of the following reaction. (Answer specifically and discuss…
A: 1. This reaction is the base hydrolysis of ester.2. in this reaction ester gives corresponding acid…
Q: Can the alkyl halide in the drawing area be made in one step from an alcohol, without losing…
A: The objective of this question is to draw the starting material and reagents used for the product…
Q: Thin layer chromatography experiment. Show calculations for the Rf of each spot you observed.
A: see the explanation partExplanation:
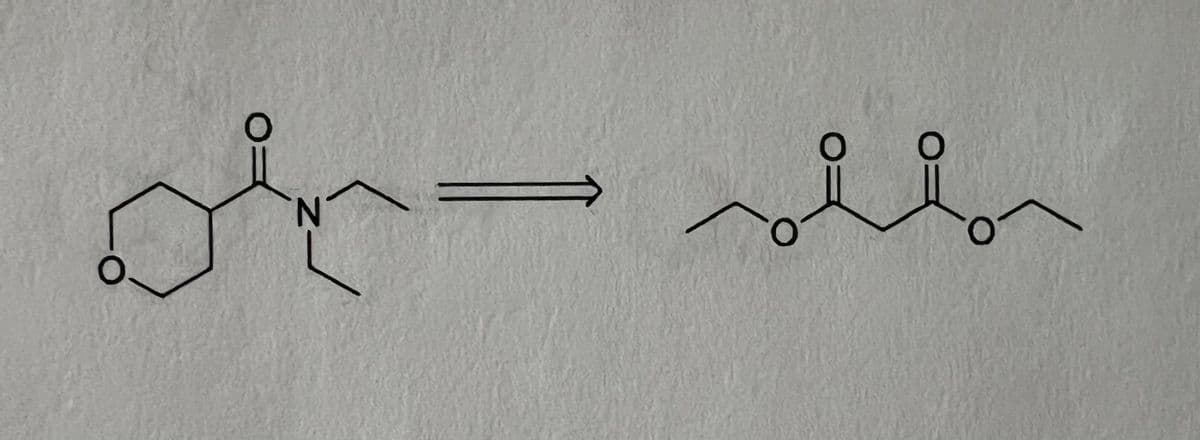
Step by step
Solved in 3 steps with 2 images


