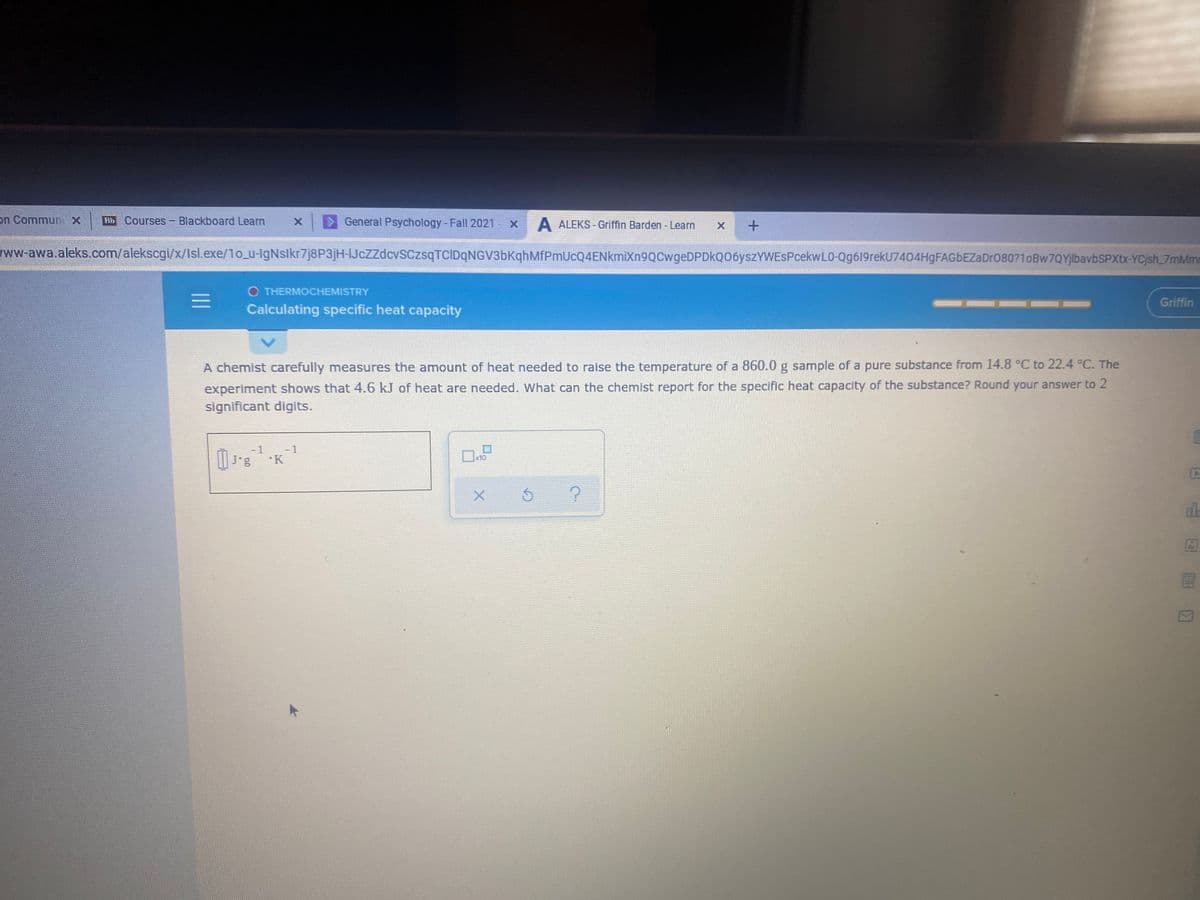
O THERMOCHEMISTRY Griffin Calculating specific heat capacity A chemist carefully measures the amount of heat needed to raise the temperature of a 860.0 g sample of a pure substance from 14.8 °C to 22.4 C. The experiment shows that 4.6 kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Round your answer to 2 significant digits. II
O THERMOCHEMISTRY Griffin Calculating specific heat capacity A chemist carefully measures the amount of heat needed to raise the temperature of a 860.0 g sample of a pure substance from 14.8 °C to 22.4 C. The experiment shows that 4.6 kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Round your answer to 2 significant digits. II
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.9QAP
Related questions
Question
Transcribed Image Text:on Commun x
Bb Courses- Blackboard Learn
> General Psychology-Fall 2021 - x
A ALEKS - Griffin Barden - Learn
ww-awa.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-IJcZZdcvSCzsqTCIDqNGV3bKqhMfPmUcQ4ENkmiXn9QCwgeDPDkQ06yszYWEsPcekwL0-Qg619rekU7404HgFAGbEZaDr080?1oBw7QYjlbavbSPXtx-YCjsh_7mMm
O THERMOCHEMISTRY
Griffin
Calculating specific heat capacity
A chemist carefully measures the amount of heat needed to raise the temperature of a 860.0 g sample of a pure substance from 14.8 °C to 22.4 °C. The
experiment shows that 4.6 kJ of heat are needed. What can the chemist report for the specific heat capacity of the substance? Round your answer to 2
significant digits.
-1
- 1
x10
J'g K
X.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

