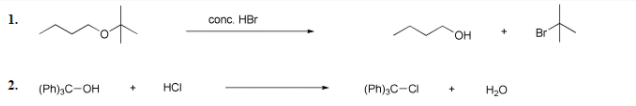
of 1. conc. HBr HO. Br 2. (Ph),C-OH HCI (Ph)3C-CI H20 f = Sy1 Nucleophilic substitution d = E1 Elimination e = E2 Elimination a = Proton transfer b= Lewis acid base c = Electrophilic addition g= SN2 Nucleophilic substitution The rections above involve synthesis or reactions of alcohols and ethers. Identify the mechanism by which they proceed from among the mechanisms listed. Use the letters a - g for your answers.
of 1. conc. HBr HO. Br 2. (Ph),C-OH HCI (Ph)3C-CI H20 f = Sy1 Nucleophilic substitution d = E1 Elimination e = E2 Elimination a = Proton transfer b= Lewis acid base c = Electrophilic addition g= SN2 Nucleophilic substitution The rections above involve synthesis or reactions of alcohols and ethers. Identify the mechanism by which they proceed from among the mechanisms listed. Use the letters a - g for your answers.
Chapter10: Organohalides
Section10.2: Preparing Alkyl Halides From Alkanes: Radical Halogenation
Problem 4P: Taking the relative reactivities of 1°, 2°, and 3° hydrogen atoms into account, what product(s)...
Related questions
Question
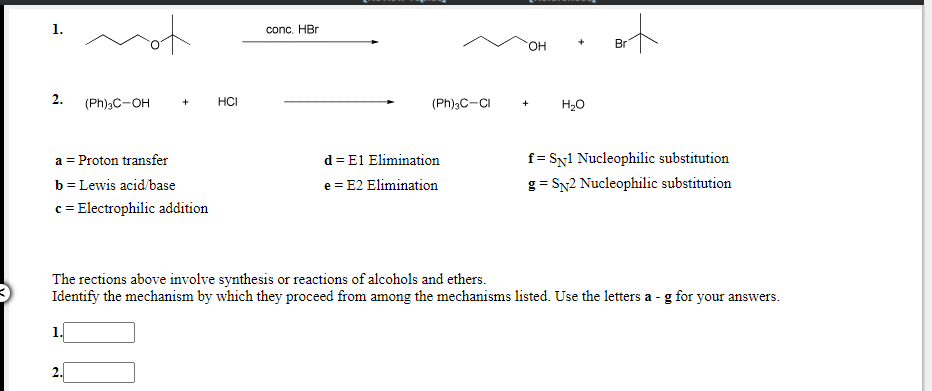
Transcribed Image Text:conc. HBr
HO.
Br
2.
(Ph)3C-OH
HCI
(Ph);C-CI
H20
d = E1 Elimination
e = E2 Elimination
a = Proton transfer
f = Sy1 Nucleophilic substitution
b = Lewis acid/base
c = Electrophilic addition
g = SN2 Nucleophilic substitution
The rections above involve synthesis or reactions of alcohols and ethers.
Identify the mechanism by which they proceed from among the mechanisms listed. Use the letters a - g for your answers.
2.
Expert Solution
Step 1
The two reactions given are,
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

