Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 113AE: Some of the physical properties of H2O and D2O are as follows: Property H2O D2O Density at 20C...
Related questions
Question
Transcribed Image Text:30% C4
Tue 4:02 PM
File
View
History
Bookmarks Profiles
Tab
Window Help
Chrome
Edit
ALEKS
ALEKS - Davi
O Gloss Purple
O S550 Mustang X
(1 unread) - d x
WPAL 101 23
Purple/Black
$550 mustang
D Paused
Update
i www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-lvdWKW_BBZZI6tTytly4Fcfu6zOtOf80MM9smv0Tza6Ag2eojQ0KaHKVTXzDxvgb5pl..
David V
O CHEMICAL REACTIONS
Using a chemical equation to find moles of product from moles ..
OO OO D O/5
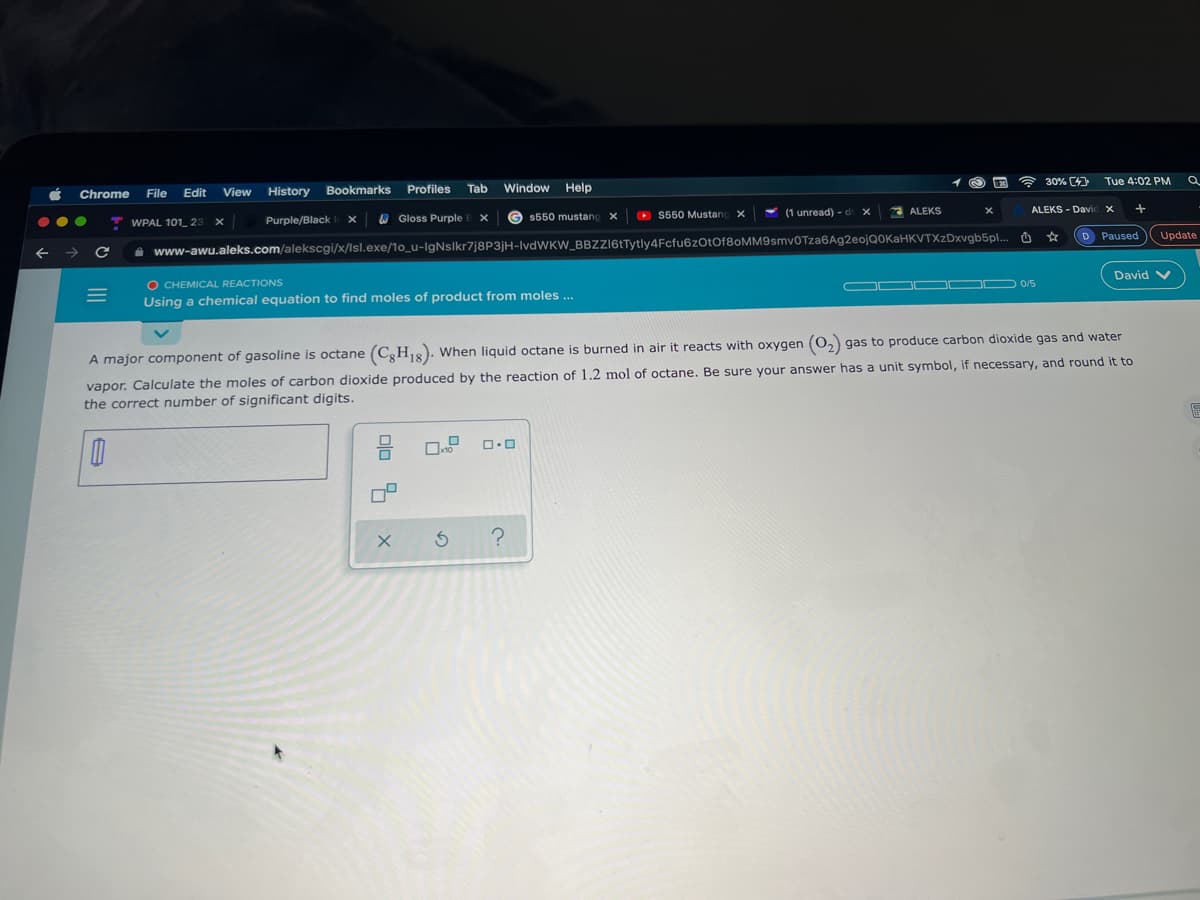
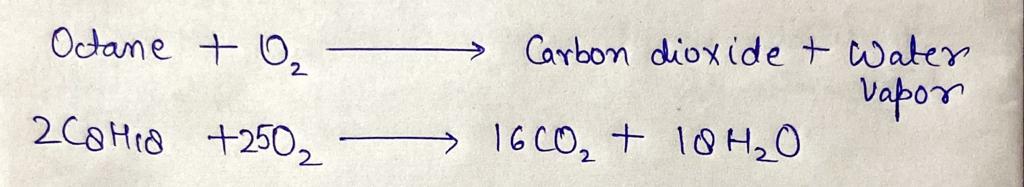
A major component of gasoline is octane (C,H,8). When liquid octane is burned in air it reacts with oxygen (0, gas to produce carbon dioxide gas and water
vapor. Calculate the moles of carbon dioxide produced by the reaction of 1.2 mol of octane. Be sure your answer has a unit symbol, if necessary, and round it to
the correct number of significant digits.
Expert Solution
Step 1
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
