// IN B R tion 8 of 25 2 NaCl(aq) + K, S(aq) → Na, S(aq) + 2 KCI(aq) (3)°0E + (s)1D – (s)*(*01) O acid-base neutralization O acid-base neutralization O precipitation O precipitation xopə1 O none of these reaction types хораг none of these reaction types KOH(aq) + AgNO, (aq) KNO, (aq)+A£OH(s) CaO(s) + CO,(g) → CACO3(s) → CaCO,(s) O acid-base neutralization O acid-base neutralizatin O precipitation O precipitation хорәл none of these reaction types xopə1 O O none of these reaction types Ba(OH), (aq) +2 HNO, (aq) Ba(NO,),(aq) + 2 H,O(I) O acid-base neutralization O precipitation MacBook Pro Q Search or enter website name | & %24 4 %23 7. 5. 6. { 3. E. K. C.
// IN B R tion 8 of 25 2 NaCl(aq) + K, S(aq) → Na, S(aq) + 2 KCI(aq) (3)°0E + (s)1D – (s)*(*01) O acid-base neutralization O acid-base neutralization O precipitation O precipitation xopə1 O none of these reaction types хораг none of these reaction types KOH(aq) + AgNO, (aq) KNO, (aq)+A£OH(s) CaO(s) + CO,(g) → CACO3(s) → CaCO,(s) O acid-base neutralization O acid-base neutralizatin O precipitation O precipitation хорәл none of these reaction types xopə1 O O none of these reaction types Ba(OH), (aq) +2 HNO, (aq) Ba(NO,),(aq) + 2 H,O(I) O acid-base neutralization O precipitation MacBook Pro Q Search or enter website name | & %24 4 %23 7. 5. 6. { 3. E. K. C.
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
ChapterA1: Evaluation Of Analytical Data
Section: Chapter Questions
Problem A1.22QAP
Related questions
Question
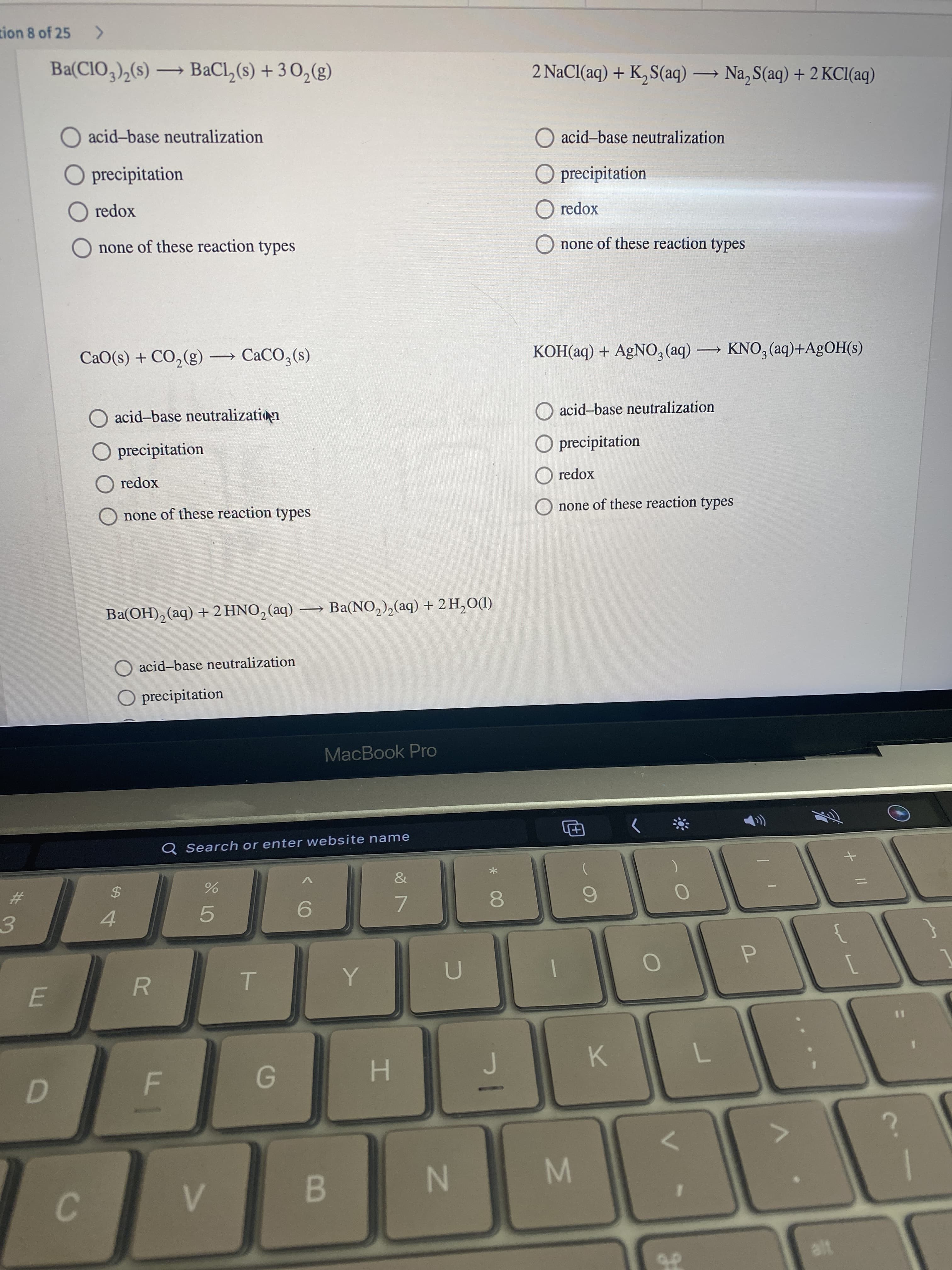
Transcribed Image Text://
IN
B
R
tion 8 of 25
2 NaCl(aq) + K, S(aq) → Na, S(aq) + 2 KCI(aq)
(3)°0E + (s)1D – (s)*(*01)
O acid-base neutralization
O acid-base neutralization
O precipitation
O precipitation
xopə1 O
none of these reaction types
хораг
none of these reaction types
KOH(aq) + AgNO, (aq) KNO, (aq)+A£OH(s)
CaO(s) + CO,(g) → CACO3(s)
→ CaCO,(s)
O acid-base neutralization
O acid-base neutralizatin
O precipitation
O precipitation
хорәл
none of these reaction types
xopə1 O
O none of these reaction types
Ba(OH), (aq) +2 HNO, (aq) Ba(NO,),(aq) + 2 H,O(I)
O acid-base neutralization
O precipitation
MacBook Pro
Q Search or enter website name
|
&
%24
4
%23
7.
5.
6.
{
3.
E.
K.
C.
Expert Solution
Step 1
Introduction :
Redox Reactions can be defined as the chemical reactions in which transfer of electrons occurs from the reductant to the oxidant.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
