of the substance at 50 oC and at pressure just slightly above 1 atm? 4.3 Give the estimate critical temperature ( oC) and pressure (atm) of the unknown substance. 4.4 What is the normal boiling point (oC) of this
of the substance at 50 oC and at pressure just slightly above 1 atm? 4.3 Give the estimate critical temperature ( oC) and pressure (atm) of the unknown substance. 4.4 What is the normal boiling point (oC) of this
Chapter11: The Air Around Us
Section: Chapter Questions
Problem 8E
Related questions
Question
4.1 How will the melting point of the substance
change if the pressure is increased above 1
atm?
4.2 What will be the phase (solid, liquid or gas)
of the substance at 50 oC and at pressure
just slightly above 1 atm?
4.3 Give the estimate critical temperature (
oC)
and pressure (atm) of the unknown
substance.
4.4 What is the normal boiling point (oC) of this
substance?
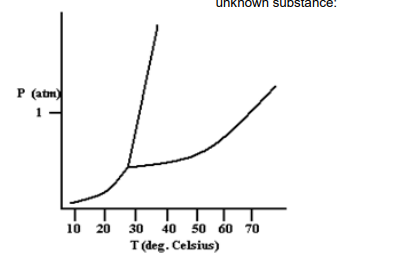
Transcribed Image Text:unknown substance:
P (atm)
1
10 20
30
40 50 60 70
T (deg. Celsius)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,