OH 1. Na OEt CH3OH Br Br2 HBr ÓCH, d = Radical chain addition e = Electrophilic addition a = Proton transfer g= E2 Elimination h = SN1 Nucleophilic substitution i = SN2 Nucleophilic substitution b= Lewis acid/base c = Radical chain substitution f= El Elimination Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a - i for your answers. 1. 2. 2.
OH 1. Na OEt CH3OH Br Br2 HBr ÓCH, d = Radical chain addition e = Electrophilic addition a = Proton transfer g= E2 Elimination h = SN1 Nucleophilic substitution i = SN2 Nucleophilic substitution b= Lewis acid/base c = Radical chain substitution f= El Elimination Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a - i for your answers. 1. 2. 2.
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter20: Acidity And Pka Of Phenols
Section: Chapter Questions
Problem 11E
Related questions
Question
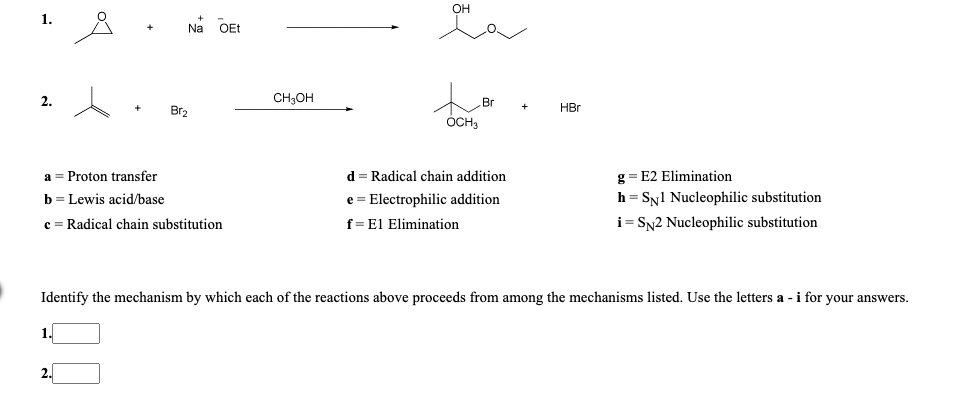
Transcribed Image Text:1.
Na
OEt
2.
CH;OH
Br
Br2
HBr
ÓCH
a = Proton transfer
d = Radical chain addition
g = E2 Elimination
h = SN1 Nucleophilic substitution
i= Sy2 Nucleophilic substitution
b = Lewis acid/base
e = Electrophilic addition
c = Radical chain substitution
f=El Elimination
Identify the mechanism by which each of the reactions above proceeds from among the mechanisms listed. Use the letters a -i for your answers.
1.
2.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning