Q: Write a formula for molecular compound. Dichlorine monoxide
A: A chemical compound consists of two or more different elements which are bonded with each other thro...
Q: Chrysanthemic acid occurs as a mixture of esters in flowers of the chrysanthemum (pyrethrum) family....
A:
Q: I need help on parts A through C.
A: Complete combustion reaction is a reaction in which the hydrocarbon reacts with oxygen to produce CO...
Q: The following concentrations were measured for an equilibrium mixture at 500 K: [N2] = 3.7 ×10-2 M; ...
A: The concentrations are given as, [N2] = 3.7 x 10-2 M [H2] = 3.2 x 10-2 M [NH3] = 1.9 x 10-2 M The re...
Q: draw the mechanism for this reaction
A: The mechanisms in organic synthesis of the products are the most defined route to explain how the re...
Q: D and H please
A: PBr3 is a source ob bromide ion, therefore it is called as a brominating agent. CrO3 is an Oxidizi...
Q: One of the radiographic devices used in a dentist's office emits an X-ray of wavelength 2.090 × 10−1...
A:
Q: From each pair of compounds, select the one that is more soluble in water.
A: Water is the polar molecule due to the presence of electronegative element oxygen which attract ele...
Q: Write an equation for the formation of each compound from its elements in their standard states, and...
A: Step 1: Hello. Since the question multiple sub-parts, the first three are solved. In case the soluti...
Q: Why is the reference electrode placed near the working electrode in a three electrode cell?
A: We know that the working electrode is the place where the reactants undergo reaction like oxidation...
Q: A chemist dissolves 1.406 g pure platinum (Pt) in an excess of a mixture of hydrochloric and nitric ...
A: Concept introduction: According to stoichiometry, the number of moles of each atom on either side of...
Q: A weather balloon calibrated at 0.00 °C to have a volume of 20.0 L has what volume in L at -14.8 °C ...
A:
Q: What is the Clausius–Clapeyron equation and why is it important?
A: The gas pressure is the pressure that is exerted by the gaseous particles on the wall of container. ...
Q: A weather forecaster predicts the temperature will reach 31 °C. What is this temperature (a) in K,
A: While converting the ˚C (Celcius) scale to K (Kelvin) scale, 273.15 is added. 0˚C is equal to 273.15...
Q: Calculate the molarity of an acetic acid solution if 37.5 mL of an HC2 H3 O2 solution is titrated wi...
A: The titration reaction given is HC2H3O2 (aq) + KOH (aq) → H2O (l) + KC2H3O2 (aq)
Q: Select the member of each pair that shows the greater rate of SN2 reaction with KN3 in acetone.
A: SN2 reaction rate depends upon the steric effect. Higher the steric hindrance, lower would be the ra...
Q: Chemistry Question
A:
Q: The solubility of copper(I) chloride is 3.91 mg per 100.0 mL of solution. Calculate Ksp for CuCl.
A: The solubility of copper (I) chloride is 3.91 mg per 100.0 mL of solution. Ksp for CuCl is to be de...
Q: e-please see attached
A:
Q: Explain why an equilibrium between Br2(l) and Br2(g) would not be established if the container were ...
A:
Q: What is the pH of a buffer solution containing 0.21 M HNO2 and 0.10 M NO, ?
A: Since HNO2 is a weak monoprotic acid with pKa = 3.15 And the solution is given buffer solution with...
Q: Write balanced equations for the dissolution reactions and the corresponding solubility product expr...
A: (a) The dissociation reaction of Ag2CO3
Q: 3-ethylpentane Draw the condensed structural formula
A: Given compound, 3-ethylpentane
Q: Complete sentences
A: According to VSEPR (valence shell electron pair repulsion) theory, when there are lone pair of elect...
Q: For each bond, show the direction of polarity by selecting the correct partial charges. v I-F I-CI v...
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: Which of the following statements about vitamin A, drawn below, is true? HO. vitamin A Multiple Choi...
A: The structure of Vitamin A given is
Q: Write the balanced NET IONIC equation for the reaction that occurs when hydrochloric acid and potass...
A: The balanced net ionic equation for the reaction that occurs when hydrochloride acid and potassium h...
Q: d) please see attached question concentration KOH=20.8325mL pH solution=2.9 beginning of the titrati...
A:
Q: 1-What color is CoCl42-? Group of answer choices blue pink purple No answer text pr...
A: 1-What color is CoCl42-? Blue is the correct answer.
Q: The gas in a 250. mL piston experiences a change in pressure from 1.00 atm to 2.75 atm. What is the ...
A: Expression for the Boyle’s law at constant moles and constant temperature is given below,
Q: 15.0 L of an ideal gas at 298 K and 3.36 atm are heated to 383 K with a new pressure of 5.35 atm. Wh...
A: Given: Initial volume = 15.0 L Initial temperature = 298 K Final temperature = 383 K Initial pre...
Q: 1-For the reaction, 4 Cl- + Co(H2O)62+ <--> CoCl42- + 6 H2O (color 1) ...
A: 1. Given:
Q: Calculate the osmotic pressure exerted by 68.2 g of aqueous sucrose (C12H22O11) in 3 L of solution a...
A: Since the osmotic pressure is given by Osmotic pressure = i X CRT where i = Van't Hoff factor = 1 ...
Q: Which ionic radius is largest of the ions listed? Na+ N3– F– Mg2+ How to the cations/anion...
A: The ionic radius of isoelectronic species i.e the species which have same number of electrons, depen...
Q: Determine the percent ionization of a 0.130 M HCN solution.
A: Given data : eo Initial concentration of [HCN] = 0.130 The reaction involve in the ionization of HC...
Q: What is the pOH of a 0.013 M Ca(OH)2 solution
A:
Q: 1-For the reaction, 4 Cl- + Co(H2O)62+ <--> CoCl42- + 6 H2O (color 1) ...
A: A reaction is said to be in equilibrium if the rate of the forward reaction is equal to the rate of ...
Q: Balance the two half reactions tor te reaction in an acid solutich. H,0,+ I→ I,t H, O
A: The reaction given is H2O2 + I- -------> I2 + H2O Since in the above reaction, the oxidation stat...
Q: How many mL of H20 must be added to 25.0 of NAcl (solute) to prepare a 15% (v/v) NAcl solution?
A: Since the units of NaCl solute is not given. Hence I'm assuming it to be 25 mL of NaCl solute.
Q: Calculate the percent ionization of hydrazoic acid (HN3) in solutions of each of the following conce...
A: HN3 is a weak acid with Ka = 1.9 X 10-5 Hence the partial dissociation reaction of HN3 is => HN3...
Q: What are the units for molar mass? Group of answer choices A) g B) g/L C) g/mol D) g/mL
A: The molar mass is defined as the mass of a sample of that compound which when divided by the amount...
Q: A 1.2 L weather balloon on the ground has a temperature of 25°C and is at atmospheric pressure (1.0 ...
A: According to ideal gas law, PV = nRT where P = pressure V = volume n = moles R = gas constant = 0.08...
Q: How much and in what direction will each of the following affect the rate of the reaction: ...
A:
Q: 2b-please see attached
A: Given : The solution is having 0.2 M NaI salt which is completely soluble. Hence the concentration o...
Q: On the elemental level, decomposition of buried organic matter occurs by reactions classified as oxi...
A: The breakdown of the complex organic material returning the important minerals back to the soil is c...
Q: The concentration of Ag+ in a solution saturated with Ag2C2O4(s) is 2.3 × 10-4 M. Calculate Ksp for ...
A:
Q: The pH of a 0.72 M solution of hydrofluoric acid (HF) is measured to be 1.66. Calculate the acid dis...
A: HF is a weak acid. Hence it will dissociate partially as per the reaction HF ------> H+ + F-
Q: How many grams of CuO would be required to produce 22.5 moles of nitrogen gas in the following chemi...
A: The reaction given is 2 NH3 (g) + 3 CuO (s) ------> 3 Cu (s) + N2 (g) + 3 H2O (g)
Q: Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol more rapidly
A: SN1 reactions are the unimolecular nucleophilic substitution reaction in which the rate of the react...
What is the major
pyridine? Explain why the major product is different in these reactions.
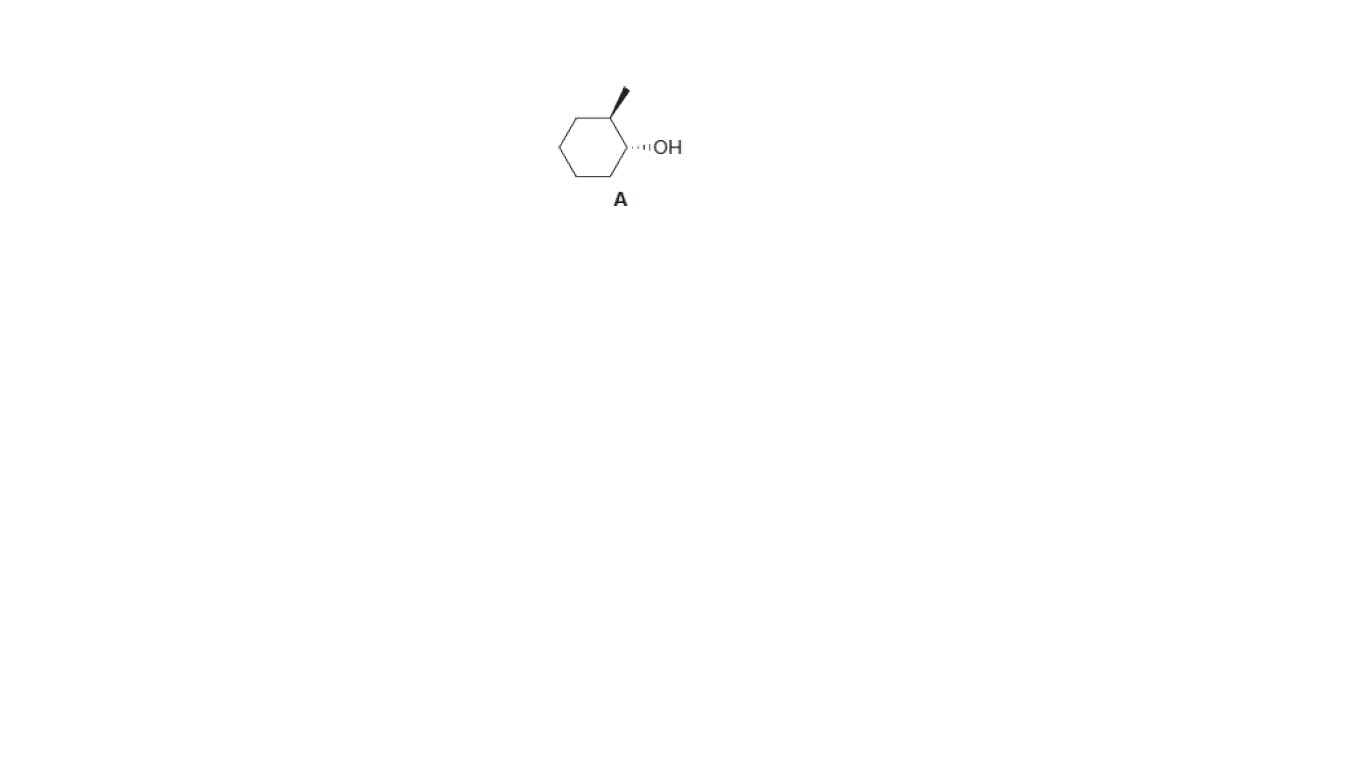
Step by step
Solved in 2 steps with 1 images


