ghe mau speetla foremiela organometallie compound B Mo esHs (u)C,0, exhibit peake at m/z an 282, as4, 226, 198, 133 & 98 amu. What is ik probable aMuctuu
Q: Draw and provide the Functional Group and R Group for the following: 1. Benzaldehyde (aldehyde) 2....
A:
Q: Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing the solubility constant...
A:
Q: ОН a) b)
A:
Q: The diagram shows the energy of a reaction as the reaction progresses. Label each of the following i...
A: Energy diagram is the graphical representation of a reaction In this diagram a graph is drawn be...
Q: What is the volume of the solute dissolved in 100mL of solution in a 22%V/V solution?
A: Given, the volume of the solute dissolved in 100mL of solution in a 22%V/V solution is :
Q: Consider the following system at equilibrium where Kc = 1.80×10-4 and AH° = 92.7 kJ/mol at 298 K. NH...
A: Answer: Given reaction is shown below: NH4HS(s)↔NH3(g)+H2S(g) 1 mole of NH4HS is decomposing to give...
Q: 3. A solution that may contain any of the group III cations. Treatment of the solution with CIO in a...
A: Group 3 cation involve Cr3+, Al3+ , Ni2+ and Fe3+ .
Q: Consider the following system at equilibrium where H° = -10.4 kJ, and Kc = 55.6, at 698 K. H2(g) + ...
A:
Q: which of the following statements regarding orbital hybridization is true? a. it results to a simil...
A: Mixing of atomic orbital of comparable energy is known as hybridization .
Q: Enumerate
A: Enumerate the 4 Guidelines of ICH.,
Q: MULTIPLE CHOICE PROBLEM SOLVING The boiling point of 350mL of substance A was determined to be 97.5o...
A:
Q: What is the IUPAC name of the substance shown in the following model? H ball & stick labels Submit A...
A: Answer of the question given below,
Q: A 14.92 g sample of bacon was pureed in a blender with 100.0 mL of water. The suspension was filtere...
A:
Q: 2.50 moles H,0 and 100. grams of C are placed in a 50.0 liters container. At equilibrium for the rea...
A:
Q: A student ran the following reaction in the laboratory at 695 K: N2(g) + 3H2(g) 2NH3(g) When she int...
A:
Q: 1.00 g of H2, 2.00 g of N2 and 2.00 g of CO2 at 25 °C and 1 atm were mixed isothermally, assuming th...
A:
Q: 28. This is the most ideal bond angle of hybrid orbitals in methane. A. 109.5° B. 120° C. 180° D. 90...
A: 28. This is the most ideal bond angle of hybrid orbitals in methane Answer- A. 109.5' methane is a ...
Q: Please do help me in this mathematics of measurement subjec please. I promise to give a good feedbac...
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: Identify the product, if any, that would form in each of the following reactions. 73) CH 3 - CH2-CH2...
A: Alcohol can be defined as the organic compound in which hydroxyl group is attached to carbon atoms.
Q: What is the meaning of half-life of an isotope. Explain with the help of an example
A: What is the meaning of half-life of an isotope. Explain with the help of an example
Q: 3. A solution that may contain any of the group III cations. Treatment of the solution with CIO in a...
A: Group 3 cation involve Cr3+ , Al3+ , Ni2+ and Fe3+.
Q: A dilute HCI solution is added to sodium carbonate in a closed flask (5.0 L). After the temperature ...
A: 3. HCl reacts with Na2CO3 as per the following chemical reaction: 2HCl(aq)+Na2CO3(aq)→2NaCl(aq)+CO...
Q: Given the reaction 2A (g) = B (g) + C(g) and the fact that Kc = 3.5 at a certain temperature. You pl...
A:
Q: What is molality of a solution prepared by dissolving 20g MgCl2 in 250 g water?
A: We have to calculate the molality of solution.
Q: Compare the density values of O2 at 480 K and 280 bar using the ideal gas and the van der Waals equa...
A: To determine relative error.,
Q: What is the major product of the Diels Alder reaction shown in the box? ? +
A:
Q: Identify the following substance as aromatic, anti-aromatic, or non-aromatic. Explain why.
A:
Q: Part A: For calculations involving gaseous systems, it is important to use the kelvin temperature sc...
A: Part A: Thermometer reading = 221 °C Part B: Temperature of the gas = 286 K
Q: Predict the products of this nucleophilic acyl substitution reaction (i.e. show the leaving group). ...
A:
Q: A chemist receive different mixtures for analysis with the statement that it contain NaOH, NaHCO3 , ...
A: NaOH and NaHCO3 can't be present together in a mixture since they react to form Na2CO3. Thus, poss...
Q: activation energy (E.) enthalpy of reaction (A,H) reverse activation energy (En.r) reactants product...
A: Energy profile diagram shows the Energy V/s reaction process. By which we can identify the speed of ...
Q: i) How many moles of hydrogen in FeSO«'6 H;O are present in one 500-mg tablet of Feosol? Iv) How man...
A:
Q: Consider the dissolution of CaCl2 : CaCl2 (s) → Ca2+ (ag) + 2 C1-(ag) AH = -81.5 kJ An 11.7-g sample...
A: ΔH = -81.5 kJ of 1 mol CaCl2 Molar mass of CaCl2 = 110.98 g /mol Moles of CaCl2 present in solution...
Q: Which of the following is a primary amine? CH,CHCH, OH CH3 CH CH3 H3C. CH,CHCH, 1. NH, H2
A: Organic chemistry is branch of chemistry in which we deal with compound related to carbon and hydrog...
Q: i) How many moles of hydrogen in FeS0«6 H;O are present in one 500-mg tablet of Feosol? iv) How many...
A: Since it is not given in the question about the composition distribution of the drug, assume that Fe...
Q: Prior to the discovery that freon-12 (CF2C12) was harmful to the Earth's ozone
A: Prior to the discovery that freon-12 (CF2Cl2 ) was harmful to the Earth’s ozone layer, it was freque...
Q: H2N' IZ
A: The objective of the question is to find the product when the given molecule treated with strong red...
Q: Sample Problem: A 12.1 Moles NaOH solution means there are 12.1 moles of NaOH in liter of solution. ...
A:
Q: 1. All of the following statements on solution equilibrium is not true, except: A. Heating allows mo...
A: Two questions based on equilibrium concept that is to be accomplished.
Q: Which compound will react the slowest upon nitration Br A D
A:
Q: 21. An orbital which does not exhibit symmetry along the internuclear axis: A. Sigma orbital B. Pi o...
A:
Q: 3.Allicin is a compound known to be the cause of the distinct smell of garlic. If an analysis of the...
A: Determination of empirical formula The percentage composition of elements present in given compoun...
Q: Give the IUPAC name of the following compounds: а. SH b. HS
A: Given compounds are : Give the IUPAC names of the given compounds = ?
Q: An elementary process has an activation energy of 88 kJ/mol. If the enthalpy change for the reaction...
A:
Q: If 98%= 18.1 M (molarity) Find the corresponding molarity of the given %HCI solution: a) 61% b) 52.4...
A:
Q: Water has a vapor pressure of 18.0 torr and a density of 0.997 g/mL. Acetone (CHCOCH:) has a vapor p...
A:
Q: Please solve for Ka
A: Weak acid is an acid that does not dissociates completely, rather there occurs equilibrium between w...
Q: ОН d) a) b)
A: Here we have to write resonance structures of the given species with curved arrow and the resonance ...
Q: ne rate c
A:
Q: Question 5 Why do we need to monitor water temperature when dealing with enzymes? O to awaken the sl...
A: Few questions based on general chemistry that is to be accomplished.
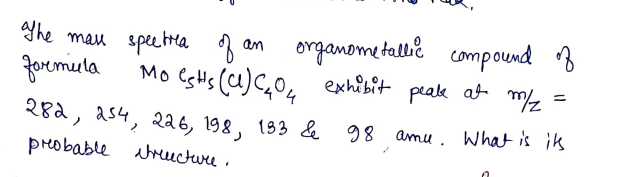
Step by step
Solved in 2 steps with 1 images

- Calculate the amount of phycocyanin in Sample 1 in mg where A620=0.211 and A650=0.086, taking into account the dilution factor as per question 6 (100ul), and the total volume of extract as per question 4 (140ml) . Note your answer to 2 decimal placesCalculate the amount of phycocyanin in Sample 1 in mg where A620 = 0.193 and A650 = 0.095, taking into account the dilution factor of 100 ul, and the total volume of extract 45ml. Note your answer to 2 decimal places.Elaborat ur answer pls thank u
- Hello, I would like help with problem 42 in the image attached.A sample of steel (0.506 g) was digested in acid and the solution made up to 100 cm3 with deionised water. Aliquots (25.0 cm3) of this solution were transferred into two volumetric flasks (100 cm3) labelled A and B. Flask A was made up to volume with deionised water. To flask B was added 10.0 cm3 of a 260 ppm standard solution of Mn2+ which was then also made up to volume with deionised water. The following absorbance results were obtained: Solution A = 0.356, Solution B = 0.578. Use these results to calculate the percentage (% w/w) of manganese in the steel sample.For Indirect Iodometric Analysis of Copper... ~0.0896g KIO3 necessary to consume 350mL of 0.1 M Na2S2O3, Na2S2O3 is stored in an amber glass bottle until ready for use. Primary Standard KIO3 has 2g of KI, 50mL of DI water, and 10 mL of 1.0M HCl is added then immediately titrated with Na2S2O3 until medium yellow or straw... then 5mL of starch indicator is added and titrated again until blue black color turns clear. Unknown CuO use 1.2G of Unknown, 20mL of HNO3 heated until sample dissolved, 25 mL of DI water added and boiled until clear light blue color, after cooling 1:1 NH3 added (~34.47 mL of NH4OH reagent) until permanent deep blue color amine complex, 2g of NH4HF2 added and swirled until dissolved, 3 g of KI is added then titrated immediately with Na2S2O3 until brown color of iodide is nearly gone (brown milk color), 2 g of KSCN and 3 mL of starch indicator is then added with titration continuing until disappearance of new blue black color. 1. Na2CO3 is often added to thiosulfate…
- For Indirect Iodometric Analysis of Copper... ~0.0896g KIO3 necessary to consume 350mL of 0.1 M Na2S2O3, Na2S2O3 is stored in an amber glass bottle until ready for use. Primary Standard KIO3 has 2g of KI, 50mL of DI water, and 10 mL of 1.0M HCl is added then immediately titrated with Na2S2O3 until medium yellow or straw... then 5mL of starch indicator is added and titrated again until blue black color turns clear. Unknown CuO use 1.2G of Unknown, 20mL of HNO3 heated until sample dissolved, 25 mL of DI water added and boiled until clear light blue color, after cooling 1:1 NH3 added (~34.47 mL of NH4OH reagent) until permanent deep blue color amine complex, 2g of NH4HF2 added and swirled until dissolved, 3 g of KI is added then titrated immediately with Na2S2O3 until brown color of iodide is nearly gone (brown milk color), 2 g of KSCN and 3 mL of starch indicator is then added with titration continuing until disappearance of new blue black color. 4. Why is the starch indicator solution…In the development o f an analytical method for the quantitative analysis o f iron using Atomic Absorption spectrophotometry (AAS), a linear calibration curve for standards o f 0.00, 5.00, 10.00, 15.00 and 20.00 ppm was prepared. An iron ore sample with an expected iron content o f 40-60% w/w is to be analysed by this method. An approximately 0.5-g sample is taken, dissolved in a minimum amount o f concentrated HC1 and diluted to 1 L in a volumetric flask using distilled water. A 5.00-mL aliquot is removed with a pipette. With explanation, to what volume (10, 25, 50, 100, 250 500 or 1000 mL) should the 5.00 mL aliquot be diluted, to minimize the uncertainty in the analysis?Solution for 0.105g of the K3[Fe(C2O4)3] (MW= 437.2 g/ mol) were dissolved in sulfuric acid and got rated with 9 mL of 0.028M KMn04. What is the experimental weight percent of oxalate in the sample?



