OH H OH H OH H H OHN HH JE OH HI H OH F CH₂OH Consider the structure of the dissaccharide cellobiose, which is produced in the enzymatic hydrolysis of cellulose. Identify the structural components of cellobiose. MCH₂OH 水 H K H Identify every acetal carbon. A B 0 D E L Identify every hemiacetal carbon. A B H H K K M M Identify every anomeric carbon. B E F H K M
OH H OH H OH H H OHN HH JE OH HI H OH F CH₂OH Consider the structure of the dissaccharide cellobiose, which is produced in the enzymatic hydrolysis of cellulose. Identify the structural components of cellobiose. MCH₂OH 水 H K H Identify every acetal carbon. A B 0 D E L Identify every hemiacetal carbon. A B H H K K M M Identify every anomeric carbon. B E F H K M
Chapter25: Biomolecules: Carbohydrates
Section25.SE: Something Extra
Problem 28VC
Related questions
Question
Transcribed Image Text:OH
H
OH
H
OH
H
H
OHN
HH JE
OH
HI
H
OH
F CH₂OH
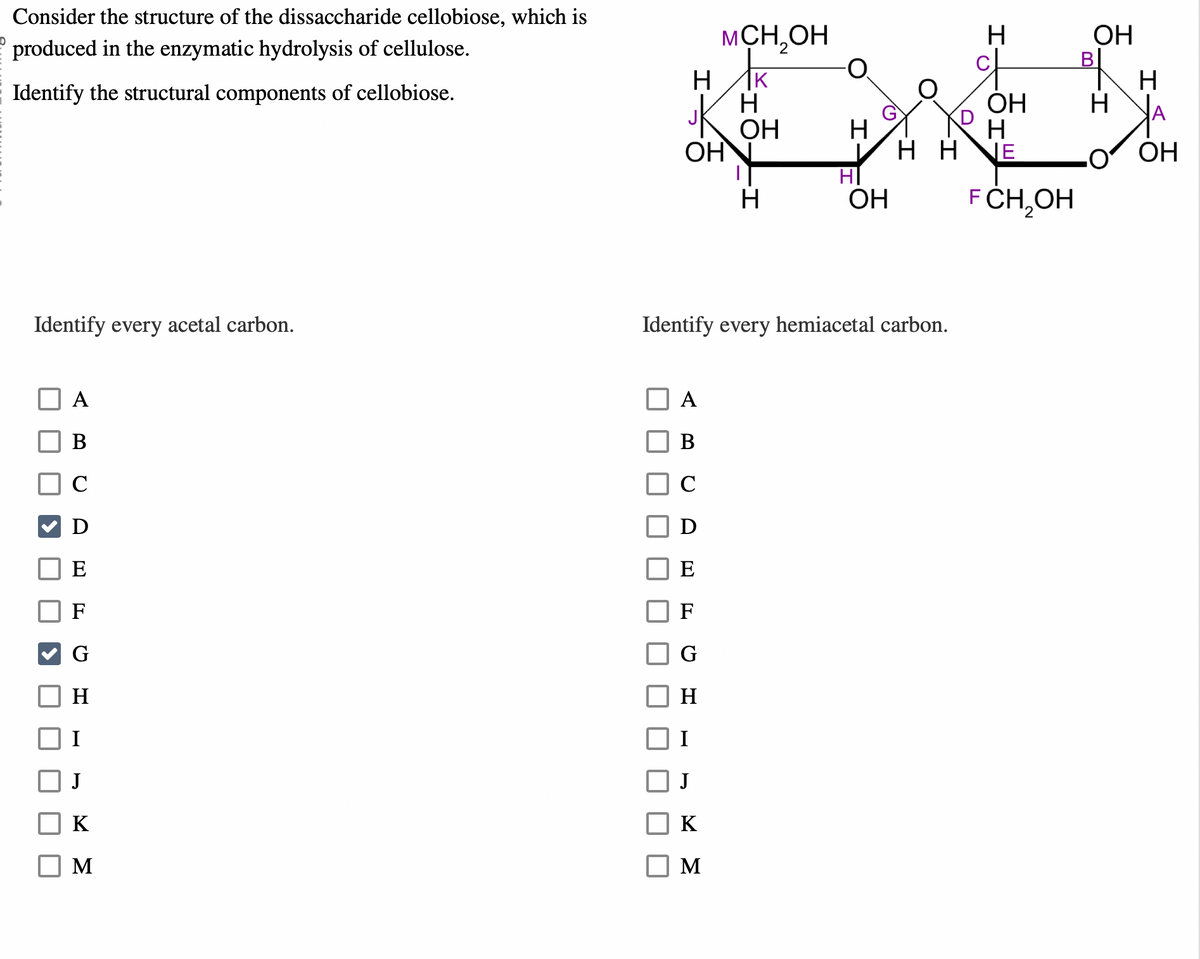
Consider the structure of the dissaccharide cellobiose, which is
produced in the enzymatic hydrolysis of cellulose.
Identify the structural components of cellobiose.
MCH₂OH
水
H K
H
Identify every acetal carbon.
A
B
0
D
E
L
Identify every hemiacetal carbon.
A
B
H
H
K
K
M
M

Transcribed Image Text:Identify every anomeric carbon.
B
E
F
H
K
M
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning