Q: What volume of O2 at 988 mmHg and 39 ∘C is required to synthesize 12.0 mol of NO ? Express your…
A: Ideal gas law equation is equation which obeys gas laws under all condition of temperature and…
Q: Instruction For each reaction below, write the following information: 1. The type of the reaction…
A: Given that, a reaction is shown below We have to answer about the reaction type. N.B.: According…
Q: Is this an SN2 reaction?
A: SN2 reaction is a substitution reaction at the sp3 hybridized carbon where the approach of…
Q: The slope of an Arrhenius plot was found to be -4570 K, and the intercept was found to be 3.3 with…
A: Arrhenius equation explain the relation between rate constant and temperature.
Q: 4. Write balanced molecular and ionic equations for following reactions: a. CaCO3(s) and HCl(aq) b.…
A: According to law of conservation of mass, the chemical reaction must be balanced and the number of…
Q: How would a student make 60.0 mL of a 0.05000 M solution of iron(III) using a stock solution that is…
A: Answer:- This question is answered by using the simple concept of dilution in which a concentrated…
Q: Which of the highlighted chemical bonds in the molecules below is longest? Shortest? In between?…
A: The bond length is defined as the average distance between nuclei of two bonded atoms in a molecule.…
Q: What is an advantage of Gas Chromatography and Mass Spectrometry (GS/MS). a. It is time…
A: Both a and b are disadvantages of Gas Chromatography and Mass Spectrometry (GS/MS) Option d is also…
Q: 16.38 a. b. How does an enzyme speed up the reaction of a substrate? After the products have formed,…
A: Proteins called enzymes to aid in accelerating chemical reactions in our bodies. Many processes of…
Q: A chemist prepares a solution of calcium bromide (CaBr.) by measuring out 140. g of calcium bromide…
A:
Q: Ph S H3CC-Br CH3CH2 H₂O
A: Given reaction is an example of nucleophilic substitution reaction of second order, in which a…
Q: How many signals do you expect in the 13C-NMR spectrum of the following compound? HO O Four O Eight…
A: Similar to proton NMR, carbon-13 NMR detects the different types of carbon atoms in a molecule and…
Q: sac ve inhihi. following graph shows the activity versus pH curves for pepsin, sucrase, and trypsin.…
A: Optimum pH is the pH at which the enzyme has maximum activity. Optimum rate is the maximum rate that…
Q: What is the likely problem with doing a diethyl ether/water extraction on the following reaction…
A: Water is a good solvent for those compounds which can form hydrogen bonding. Now the product E has 4…
Q: Show your work for calculating the equivalents of bromine used, be sure to find molar mass and…
A:
Q: A) 51 B) 45 C) 47 D) 46 E) 48 Determine the total number of valence electrons in SnCl62- and then…
A: Total number of bonded and non bonded electrons of outer most shell is called valance electrons.
Q: Draw and I palos a model of 4 molecules of H2O and label the strongest interactions that are present…
A: The strongest interaction that are present between separate H2O molecules= intermolecular H- bond
Q: Using the data in the table, determine the rate constant of the reaction and select the appropriate…
A: Given reaction: A + 2B → C + D We have to find the rate constant of the reaction using the given…
Q: What is the ideal H-N-H bond angle for that geometry? and What is the molecular geometry of NH3 ?
A:
Q: How much voltage is needed for electrolysis, when in the cell Co|Co2+ (6.40∙10-3M), Zn2+…
A: We have, Current,A = 0.078 Cell resistance,R = 5.00
Q: QUESTION 2 How many grams of O 2 would be needed to react with 4.272 moles of CS 2 in the reaction…
A: Number of moles is calculated by dividing given mass with molecular mass. In the given question, it…
Q: FUSION 2 8. H 1 9. 253 + Es 3 PARTICLE ACCELERATION TO PRODUCE A TRANSURANIUM ELEMENT + H 4 2 1 04…
A:
Q: What is the master formulae of the nicotine chewing gum? Please answer at your own easy words.
A: Nicotine chewing gum is a medicine which helps people to quit smoking.
Q: What is the approximate bond angle between 2 of the nitrogen hydrogen bonds in NH3?
A: We have been given a molecule NH3 and we have been asked the approximate bond angle between 2 of…
Q: Calculate the time required for electrolysis when a constant current of 0.852A is applied to produce…
A: We have to calculate the time needed for a constant current of 852 A to deposit 0.250 gm of cobalt…
Q: How many lone pairs are on phosphorus in BPr3 ?
A: In order to determine the lone pair on the phosphorous atom, it is very important to write the…
Q: 8 Two Steps Label step 2 only OH
A:
Q: 28. (a) In Figure P7.7, which sphere could represent a Na atom, Nation, and K atom? Provide an…
A: In P 7.7 = (a) represent Na , (c) represent Na+ and (b) represent K. As we decrease the no of…
Q: A 1.500-g sample of gasoline was burned in a bomb calorimeter whose total heat capacity is 13.20…
A: Given that the heat capacity of the bomb calorimeter is 13.20 KJ/ oC. Mass of the sample is 1.500 g…
Q: In an experiment when 50 g of C3H8 is combusted, 35 g of carbon dioxide resulted. What was the…
A: The combustion reaction for C3H8 : C3H8 + 5 O2 ==> 3CO2 + 4 H2O Given Mass of propane = 50 g…
Q: A74.12 g sample of a hydrocarbon (CH) has a mass of carbon that is equal to 62.90 g. Given this…
A: To find out the empirical formula, first we need to find out the moles of C and H. Round off that…
Q: How many grams of H 2 are needed if we want to make 95.44 grams of NH 3 by the reaction N2 + 3H2 2…
A:
Q: Supply the missing data in the tables below. Lactic Ka (K Acid 10-4) = 1.38 x 10 Iodic Ka = 1.7 x 10
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 3-methylbutanal Route X CI 1-chloropropane Et₂O + Mg MgCl propylmagnesium chloride i) Et₂O ii) H₂O…
A: The ratio of Stereoisomer of 2- methyl haptan-1- ol is 1:1 because there is only one chiral carbon(…
Q: certain temperature, the equilibrium constant for the chemical reaction she centration of AB is…
A:
Q: The van der Waals constants a and b are 5.489 L2 atm mole-1 and 0.0638 L mole-1 for C2H6. (a)…
A: Given, Vander Waals constant , a=5.489L2 atmmol2b=0.0638Lmol
Q: 13.07 ml of an Unknown Acid titrated with 24.03 ml of a Base standardized to 0.10000M. What is the…
A:
Q: For the following reaction, 9.45 grams of nitrogen gas are allowed to react with 5.81 grams of…
A: Given :- Mass of Nitrogen gas, N2 = 9.45 g Mass of Oxygen gas, O2 = 5.81 g Maximum mass of…
Q: For each pair of compounds, describe which instrumental technique (IR, MS, proton NMR, carbon NMR)…
A: Note: According to our guidelines we are supposed to answer only first three subparts. Kindly repost…
Q: Answer the questions below about the highlighted atom in this Lewis structure: H H + H-C- C=C-H │ |…
A: A sigma bond is a covalent bond created when two atomic orbitals axially overlap. The methane…
Q: هب الطلا Q2/Rhodium has an atomic radius of 0.1345 nm and a density of 12.41 g/cm³. Determine…
A: The formula to calculate the density of a unit cell is- Density, ρ=z×MV×NA Where z is the number of…
Q: Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic…
A: •Polar molecules : i) have polar bond. ii) have asymmetric structure. •Non - polar molecules :…
Q: 10.) The reaction of limestone with hydrolic acid is CaCO3 + 2HCl → CaCl₂ + CO₂ + H₂O If the…
A:
Q: What do CO, ethylene (C2H4), H2, and NO+ ligands all have in common? Group of answer choices they…
A: Ligand: An ion or molecule, which donates a pair of electrons to the central metal atom or ion to…
Q: A student collected 66.2 mL of hydrogen gas at 22 degrees Celsius. If the pressure of "dry"…
A:
Q: Question 1 Which of the following is NOT TRUE of the Schrödinger Equation? Ⓒ It is theoretically…
A: This is a simple problem of basics of quantum mechanics where we have apply the principle of…
Q: How many tetrahedral stereocenters are present in this compound? OH сH CH CHCHCHCHCH CH CH2CH
A: In order for a carbon center to have the tetrahedral geometry, the carbon center must be Sp3…
Q: How many KILOJOULES of heat is required to raise the temperature of the entire 1.0 Kilogram of steam…
A: Given, mass of steam = 1.0 kg =1000 g
Q: Why does HF have the lowest melting point compare to CH4, MgCl2, and c(s, diamond./
A: The hydrogen fluoride have lowest melting point among all because the bond between hydrogen and…
Q: 4) A redox titration: 20.00 mL of 0.00500 M Sn²+ solution below. Titration reaction: Bucony C Ce+ +…
A:
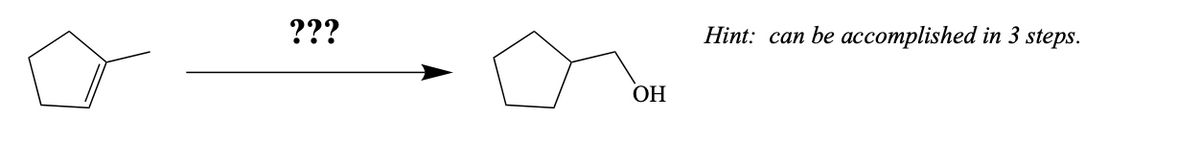
Step by step
Solved in 2 steps with 1 images


