Q: 4 pts Which of the following represents a valid resonance structure for the original cation shown?…
A: We will draw the valid reasonance structure for the given cation shown in the given question.
Q: Draw three resonance structures for OCN–. Carbon is the central atom in this anion. Indicate all…
A:
Q: 1.88/ The curved arrow notation introduced in Section 1.6B is a powerful method used by organic…
A:
Q: which of the following molecules can exhibit equivalent resonance structures? CH3OH O2 NO3- NF3
A: Equivalent Lewis structures are called resonance forms. They are used when there is more than one…
Q: Which of the following are possible resonance structures for HNO3? Select all that apply. ... :Ö:…
A:
Q: Decide whether each row is a set of resonance structures for a single molecule and select "yes" or…
A:
Q: 2. Both pairs below show arrangements of the same (fictional) formula: A2X. Which pair (a or b)…
A: Resonance is a hypothetical concept which involves delocalization of pi- electron or lone pair when…
Q: Which of the following has resonance? O a. SCI6 Ob. H20 Ос. Оз od. CH4 e. At least two of the…
A:
Q: Write Lewis structures for these compounds. Show all valence electrons. None of them contains a ring…
A: Valence electrons are the outermost electron present in an atom. Lewis structure represented the…
Q: Identify the pairs that are not resonance structures. and H3C-O-N=o: H3C-O=N-O: H3C-0-N=O: and H3C-…
A:
Q: 1. Draw Lewis Structures for the following compounds. (Be sure to minimize formal charges and draw…
A:
Q: 45. Write resonance forms that describe the distribution of electrons in each of these molecules or…
A: (a) In SO2 molecule the interaction between pi bond and lone pair of O atom induces two resonating…
Q: Draw a resonance structure that shifts a pi bond to a new position. Include all hydrogen atoms and…
A:
Q: ecide whether each row is a set of resonance structures for a single molecule and select "yes" or…
A: Given
Q: Which of the following statements is true regarding the nitrite ion? O The nitrite ion has two…
A: We have to find out which statement is correct about the nitrite ion Statement 1: nitrite ion has…
Q: 3a. A neutral nitrogen covalent bonds and has 3b) A negatively charged carbon atom typically forms…
A:
Q: Draw two resonance structures for the anion below. These resonance structures should be ones in…
A: Defination: when a Lewis can be represented by the movement of electrons through chain without…
Q: The curved arrow notation introduced in Section 1.6B is a powerful method used by organic chemists…
A: a) Please find below the curved arrows in Step [1] showing the movement of electrons
Q: Draw a resonance structure that places a pi bond in a different position. Include all lone pairs in…
A: The compound given is,
Q: 6. Which of the following does NOT depict resonance structures? (1) CH3-0-CH CH3 CH, 0 CH-CH, (2)…
A: Correct answer is (3) Resonating structures are correct in option (3) but arrow used should double…
Q: Which molecule or ion has 40 valence electrons? pyrylium cation, C5H50* oxalate anion, C2042-…
A: Interpretation: We have to calculate the valence electrons.
Q: H :0: :0: H-C-C b. HC
A: In organic chemistry certain molecular ions have the tendency of delocalization of electrons. The…
Q: Does CH3COCH3 has molecular resonance? If so, show all possible forms.
A: Resonating structures are formed by delocalization of electrons.
Q: Draw Lewis Structures for the following compounds. (Be sure to minimize formal charges and draw…
A: The Lewis dot structure define a molecular structure with the arrangement of lone pairs and bond…
Q: A bond between an atom with an electronegativity value of 3.3 & an atom with an electronegativity…
A: Electronegativity (EN) :- The tendency of an atom to attract the shared pair of electrons more…
Q: Draw a resonance structure that places a pi bond in a different position. Include all lone pairs in…
A:
Q: Which of the following is a resonance form of the species in the box? PCH P(CH2 P(CHb OPICH PICH II…
A: The resonance form of given structure mentioned in box is given in attached image.
Q: Which of the following compounds have a resonance structure? A. PO4^3- B. H2CO C. CO2 D.XeF4 E.…
A: In the formation of the chemical bond with the same or different elements, all elements tend to…
Q: Which resonance form contributes most to the overall structure of the molecule? TH. H. H. TH.
A: Answer - The correct options is (d) According to the question - Resonance form contribute most to…
Q: What is the formal charge on oxygen in the following structure? H H C-0-H H H O 2- О 1- О 1+ О 2+
A: Formal charge is obtained as – F.C = V - N - B2V = Number of valence electronsN = Non bonded…
Q: S Which of the following pairs are not resonance structures of one another? All lone pairs of…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: What is the calculated formal charge for carbon (C) in the CNO- ion's most stable resonance state?…
A: The formal charge on CNO- is -1 So Option A is correct.
Q: Draw resonance formulas for this molecule. Clearly indicate where any delta negative and delta…
A: Given structure:
Q: Draw major resonance structures, B and C, and one minor resonance structure, D. Be sure to include…
A: The structure given is,
Q: cide whetner each row is a set of resonance structures for a single molecule and select "yes in the…
A:
Q: which of these species have resonance or isomers? CH4, CH2Cl2,CH4O,H2O,H3O+,HF,NH3,H2O2,N2,P4
A: Resonance structures are groups of Lewis structures that explain the delocalization of electrons in…
Q: In HCN, the formal charge on nitrogen is Select one: a.+1 b.0 c.–1 d.None of these. For the…
A: Here we have to calculate formal charge of nitrogen of HCN and number of equienergetic contributing…
Q: 23. Write another resonance structure for ethyl acetate. Include formal charges. CH, ÖCH,CH,
A:
Q: Draw all resonance structures for the carbonate ion, C03.
A: The Carbonate ion CO32- Carbon has 4 valence electrons, each oxygen has 6 valence electrons, and…
Q: 1. Draw three resonance structures with separation of charges for the following compound: NM@2 Meo.
A: Since you have asked multiple questions, we will answer only first question for you. In order to get…
Q: Decide whether each row is a set of resonance structures for a single molecule and select "yes" or…
A: Resonance structure:If the Lewis structure of a molecule or ion cannot explain by a single structure…
Q: Consider the resonance structures below. Which resonance structure is the MAJOR contributor to the…
A: Given structures are : Which resonance structure is the MAJOR contributor of the true structure ?…
Q: Consider the resonance structures below. Which resonance structure is the MINOR contributor to the…
A: we have to identify the minor contributor from the resonance structures
Q: H3C. 2. NH2 CH3- N- C- NH2 H
A: Resonance :- The phenomenon in which a species can be represented in more than one structural form…
Q: The isocyanate ion (CNO1‐ ) has a skeleton of C‐N‐O. Draw three resonance structures. Circle the…
A: Isocyanate ion has a skeleton of N-C-O instead of C-N-O.
Q: Which of the following is the correct movement of electrons to account for a pair of resonance…
A: Answer :- 2. c
Q: Nido-CB5H9 Does this molecule have resonance structures? If so, draw each structure
A: Nido CB5H9 is isolobal to B6H10 which has nido structure. No any conjunction present in structure of…
Q: 2. Which of the following pairs represent resonance structures? H2N & *HN & NH NH2 & H3C CH3 H3C CH2…
A: Of the given pairs of compound, some are pair of resonance structures and some are isomers.…
Q: What is the formal charge of nitrogen in this structure? н ӧ: H-C-N-o: H a) +1 b) +2 c) -1 d) -2
A: Formal charge on an atom is calculated as: Formal Charge=Valence Electrons-Non bonding…
Q: 1. Consider the following Lewis structures (1-4) for the compound CHNO. Indicate the answer to the…
A:
Write A if the following compounds has a resonance form and B if they don't have.
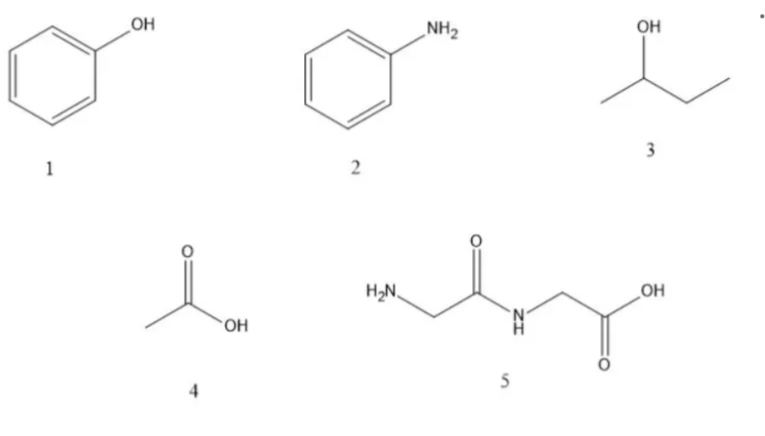
Step by step
Solved in 2 steps with 2 images


