Q: Question 15 The decomposition of HI(g) to H₂(g) and I, (g) at 600 K is 2nd order with a rate…
A: 2nd order reaction Rate constant, k = 5.82 × 10-4 M-1 min-1[HI]o = 0.725 MWhat is the concentration…
Q: Consider the structure Y-X-Y If the symbol X represents a central atom, Y represents outer atoms,…
A: According to VSEPR Theory, Steric Number= no. of Sigma bonds + no. Of lone pairs Steric no.0 L.P.1…
Q: Buffer Question 4: 250.0 mL of 0.50 mol/L sodium formate is added to 250.0 mL of 0.75 mol/L formic…
A: Volume of HCOONa = 250.0 mlMolarity of HCOONa = 0.50 mol/LVolume of formic acid= 250.0 mlMolarity of…
Q: List the products of the reaction of acetaldehyde and acetone with sodium ethoxide in ethanol.…
A: Due to their various chemical structures, acetaldehyde and acetone react with sodium ethoxide in…
Q: A mixture of 2-pentanol, pentane, 2-pentanone and pentanoic acid are placed in a distilling flask.…
A: Fractional distillation of 2-pentanol, pentane , 2-pentanone and pentanoic acid.
Q: a) Formulate the 2nd law of thermodynamics. b) Consider the following statements concerning changes…
A: A question based on second law of thermodynamics. Two subparts are given that are to be individually…
Q: Predict the ideal bond angles around each central atom in this molecule. :N=C-N-H H
A: To know the bond angle of the atoms in the following component we first have to recognize…
Q: 5. What is the molality of a solution containing 50g of HCL dissolved in 125 g water?
A: It is based on the concept of concentration termHere we are are required to find the molality of…
Q: The heat of combustion (per CH₂) of several cycloalkanes is listed below. Based on the data given,…
A: Heat of combustion of several cycloalkanesHeat of combustion…
Q: 6) Which of the following compounds is meso? H₂C HC-C-H OH H₂C HO CH₂ H CH3 F HS SH Br & ||||| Br
A: Meso compound is a stereoisomer with two or more chiral centers but no optical activity due to an…
Q: he equilibrium constant, Kp, for the reaction: PCl3 (g) + Cl2 (g) ⇌ PCl5 (g) is 0.0870 at 300°C.…
A: The direction in which the reaction proceeds to reach equilibrium can be predicted by determining…
Q: Draw a structural formula for butanoic acid.
A: To draw the structural formula -Determine the number of carbon atomsIdentify the functional group…
Q: How many structures are possible for a trigonal bipyramidal molecule with a formula of AX₂ Y₂?…
A: Lewis Structure is a very simplified representation of the valence shell electrons in a molecule. It…
Q: The reaction N2O4↽−−⇀2NO2 is allowed to reach equilibrium in a chloroform solution at 25 ∘C .…
A:
Q: Label each chiral carbon in the molecule below as having R or S configuration. HO₂C H F H Kor H3C…
A: The carbon atom of a molecule that is connected with four different atoms or groups is called a…
Q: DUE ASAP: Please choose the letter of the best and correct answer. No need for detailed explanation.…
A: Depending on the particular body of water and the local climatic circumstances, different nitrogen…
Q: what is the definiton of diffusion? a) gas molecules mix unequally. b) average distance between…
A: Diffusion is the movement of molecules in fluid from areas of high concentration to areas of low…
Q: Identify the major product in the following reaction sequence. Br₂ / light DBN NBS HBr peroxide…
A:
Q: 7. Label each chiral carbon in the molecule below as having R or S configuration. HO ₂01 H H3C H…
A: We have to locate the chiral centers in the given compound and name those centers as R or S.
Q: name this molecule
A: We have to name the given molecule.
Q: The Lewis structures of four compounds are given. OS H + :CH- -ci: Which of these molecules are…
A: In this question, we will select those molecules which are polar in nature. ( Lewis structures for…
Q: 4. Which Newman projection corresponds to point A on the graph of potential energy vs. rotation…
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: A sample of pure CaCO3 (FW= 100.1 g/mol) weighing 0.350g was dissolved in concentrated HCI (MW=…
A: A question based on concentration terms. Using the set of data available, molarity of EDTA and…
Q: M(NO3)3 2 and A certain metal M forms a soluble nitrate salt M(NO3). Suppose the left half cell of a…
A: A galvanic cell which contains same substance of different concentrations in both of its…
Q: OH 1st attempt 1. Proton transfer involving HBr 2. SN2 involving Br Each box should contain two…
A: In the given question, a reaction of alcohol with HBr is given and we have to write the product with…
Q: * * H3C. Но… Н Но... Н н н НО CH3 ...... I CH₂OH CH₂OH Н. I H CH3 ОН ОН Н НАЕ Н CH3 ...... ОН CH2OH…
A: Isomers: Same molecular formula but different structure Constitutional Isomers: Same formula but…
Q: Aqueous Pb(ClO3)2 and aqueous Nal are mixed together. What mass of precipitate, in grams, will form…
A: The question is based on the concept of reaction stoichiometry.we need to calculate mass of…
Q: Which compounds are bases in aqueous solution according to Brønsted-Lowry theory? H-N H H-C H H H…
A: The Brønsted-Lowry hypothesis is a principal idea in the field of corrosive base science, proposed…
Q: Complete the Avogadro's Statement for the element hydrogen: 1 mole of hydrogen hydrogen atoms =…
A: A mole is defined as that amount of the substance which has mass equal to gram atomic mass if the…
Q: Part D. Do the two structures A and B of each pair drawn below represent the same molecule,…
A: Isomers: Same molecular formula but different structure. Constitutional Isomers: Same molecular…
Q: مه h. H H3C Br H CH3 Br CH3 CH3 and and H H3C H Br Br CH3 CH3 CH3
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: the answer key, g is a configurational isomer.
A: Stereoisomers : The compounds having same molecular formula but differ in 3-dimensional arrangement.…
Q: 1. There are 2 possible ways for CuBr₂ to release Br₂: i) CuBr₂ → CuBr + 1/2 Br₂ or ii) CuBr₂ → Cu +…
A: CuBr₂ is a compound consisting of copper (Cu) and bromine (Br). The colors of CuBr₂, CuBr, and Cu…
Q: Predict the ideal bond angles around each central atom in this molecule. H H oxygen: carbon:
A:
Q: 96. A sample of (S)-(+)-lactic acid was found to have an enantiomeric excess of 72%. How much R…
A: Given that a sample of (S)-(+)-lactic acid was found to have a enantiomeric excess of 72%, that…
Q: The most stable carbocation from the following is: O a. IV O b. I O c. 11 Od. III B IV 11 III
A: A carbocation is a species in which carbon is carrying positive charge. In the given question it has…
Q: 1. Which of the following compounds will not rotate the plane of polarization of plane- polarized…
A: Optically active compounds:-Optically active compounds are those which are able to rotate plane…
Q: 7. a. Circle the compound below that corresponds to the IR spectrum shown. & 2.5 100 80 60 40 20…
A: The question is based on the concept of organic spectroscopy . we need to analyse the spectral data…
Q: 1. Is the molecule shown below chiral or achiral? ОН ОН
A: A question based on introduction to organic chemistry. An organic structure is given whose chirality…
Q: -3 The densities of pure water and ethanol are 997 and 789 kg. m respectively. The partial molar…
A: To calculate the change in volume relative to the pure components when preparing a solution with a…
Q: 3. A polynomial fit to measurements of the total volume of a binary mixture of A and B is Vtotal =…
A: A polynomial fit to measurements of the total volume of a binary mixture of A and B isVtotal =…
Q: Part C. The constitution of ectocarpene a volatile, sperm-cell-attracting material released by the…
A: Condensed formula of ectocarpene Structural formula of ectocarpene ?
Q: 8. For the reaction 2H₂O(1) + 2e → H₂(g) + 2OH(aq), calculate the volume of "dry" hydrogen gas…
A:
Q: c) i) d) e) a) h) This reaction was monitored for 2000 seconds starting with an initial H₂O2…
A: The concentration-time data of decomposition of H2O2 reaction is given.
Q: Consider the following equilibrium: 2NO₂(g) N₂O₂ (g) AG= = -5.4 kJ Now suppose a reaction vessel is…
A: Recall the given reaction, Pressure of dinitrogen tetroxide Temperature
Q: 6. What alkene should be used to synthesize the following alkyl bromides? a) b) H3C H3C Br CH 3 CH3…
A:
Q: 3. Balance the following reaction using the Oxidation Number Method 12 + HNO3 HIO3 + NO2 + H₂O
A: The oxidation state of iodine of I2 is 0. The oxidation state of iodine in HIO3 is +5.As oxidation…
Q: Question 9 Consider the ranked polarity of all fictitious compounds with different functional groups…
A: Thin-layer chromatography has two phases a stationary phase and a mobile phase. The silica gel…
Q: We measure the absorbance of a protein solution with the concentration 0.006M, which absorbs at…
A: Concentration (c) of protein solution = 0.006 MPath length (l) of cuvette = 1 cm molar absorptivity…
Q: What is the IUPAC name of the following compound?
A:
Which is more acidic? Why?
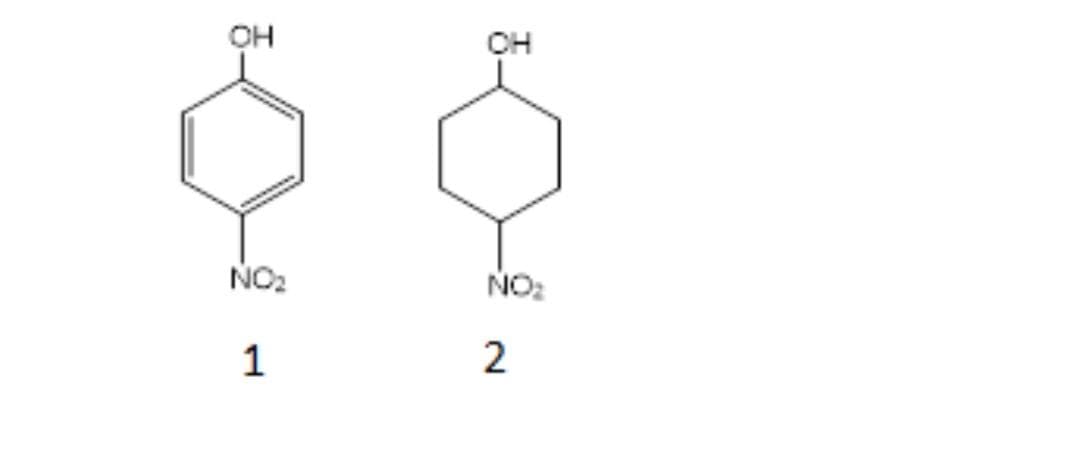
Step by step
Solved in 3 steps with 2 images


