Q: How many chiral centers are present in the molecule below? OH A.7 B. 5 C. 6 D. 8 H H
A:
Q: Which of the following hydrogen bonds would be the weakest? (Explain your answer) a. An N-H ... O…
A: This question is related to hydrogen bond. A hydrogen bond is an electrostatic interaction between…
Q: H2C CH H2 C. CH .C. H. CI H2 H2 HN. CH3 HC O =C
A:
Q: Look at the image shown. What does this image represent? Linear molecule with one domain Linear…
A: The given image represents what has to be given.
Q: TLC Questions 1. If polar compounds elute the lowest, what compound is what 2. Give an example of…
A: Thin-Layer chromatography: It is a chromatographic technique used for separation, purification, and…
Q: Which structure is identical to Br Cl CH3 O a. Br CH ČI Ob. Br CH3 Oc. Br CH3
A: In the cyclohexane ring, the group present above the plane are above and the groups present below…
Q: In the blanks provided, indicate whether each double bond is À =( CI -OH >=1
A: Here we have to predict for which of the following compounds E and Z notation is applicable. E and Z…
Q: (d) HÖ (a) yes H₂N: ОН (e) No (b) 8: Identify what type of intermolecular force the following…
A:
Q: What is the most polar area/functionality for this compound?
A: Polarity of a bond or functional group depends on the electronegative difference between two atoms…
Q: 1.Determine the number of lone pairs (if any) on each oxygen, nitrogen, and charged carbon atoms in…
A: To find the lone pair on oxygen and nitrogen atom we will use the following formula. FC= V – (N + B)…
Q: [1] CH;MgBr [2] H,0 draw structure ...
A: Given
Q: Which one of the following compounds could have exactly one ring and two C3DO double bonds? O…
A: Ans a
Q: For which compounds can a second resonance structure be drawn? Draw an additional resonance…
A: Resonance structures are the Lewis structures having the same placement of atoms but a different…
Q: See the Attachment & Answer the following question? Ques: Draw a resonance structure of…
A: Nicotine- Nicotine is obtained from nightshade family of plants. It is used as stimulant and…
Q: O IZ Im ·N- S HO
A:
Q: Describe the bonding interactions between the Carbon atoms in the following molecule.
A: Pi-orbitals are formed by the overlapping of un-hybridized p-orbitals.
Q: Name the functional groups present in compounds 1,2 and 3 and what kind of noncovalent bonding…
A: A question based on chemical bonding, which is to be accomplished.
Q: e) How many asymmetric carbon atoms are present in the following compound? . NHCH3 A) 0 B) 1| C) 2…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: Circle and name any and all functional groups present in each molecule (hint #1: two molecules have…
A: Functional groups are groups of atoms that shows a particular characteristics.
Q: HO 0. |
A: Answer
Q: 4.12 Predict all bond angles about each highlighted carbon atom: a. OH b. С. Br Br d.
A: A carbon having 4 sigma bonds in its surrounding is sp3 hybridized and the bond angle is 109o28' A…
Q: Draw
A: Ammonia is a gas at room temperature but it will easily to converted into liquid form by…
Q: 1. Each molecule below contains three functional groups. Identify them. Refer to your functional…
A: Functional group present in the given compound is,Amide has a carbonyl group that is bonded with a…
Q: Indicate whether hydrogen bonding between like molecules would be important in the following (Yes or…
A: Hydrogen bonding A Hydrogen bond is a low kind type of dipole-dipole bond that exists between an…
Q: provide the maximum number of hydrogen-bridge bonds each compound can have by providing the numeric…
A: H- bridge bonds are formed when H is attached with highly electronegative atoms like O and F. then…
Q: Draw dashed arrows to show two hydrogen bonds that can form between these two molecules. Be sure the…
A: In a molecule when hydrogen attached with electronegative atom then hydrogen gain partial positive…
Q: 2. A molecule with dual polarity has an ionic “head” and a double nonpolar “tail” is often…
A: Benzene is a non-polar compoud.
Q: For which compounds can a second resonance structure be drawn?Draw an additional resonance structure…
A:
Q: 2. Indicate the direction of the polarity for each of the bonds indicated except for all carbon to…
A:
Q: Rank the following compounds from HIGHEST to LOWEST melting point and explain why they are in that…
A: The temperature at which the compound melt is the melting point of that substance. There are several…
Q: The chemical structure of oxamide NH, is shown below. Highlight each atom that is an amino group.…
A:
Q: CH3 35. In several of our experiments, compounds with the functional groups listed below were…
A:
Q: Draw the following compounds ( put in all H’s) 2,2 – dichloro – 3 – methylpentane
A:
Q: which compounds exhibit hydrogren bonding forces? CH3CH2OH XeCl2 CH4 CH3NH2 cO SF4
A: Hydrogen bonding is a type of dipole-dipole interaction of molecules when the hydrogen is bonded to…
Q: The lone pair of electrons on the indicated N atom in the following structure occupies aorbital. ...…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Which carbon will have the least partial positive charge (8+)? A) B) CI F-C-F O=C=0 CI NH C) D) .C.…
A: Carbon have the least partial positive charge with that element with which electronegativity…
Q: Identify the most polar bond in each molecule. Q) HSCH2CH2OH
A: In modern periodic table element are arranged in the increasing order of their atomic number. S…
Q: 5. Determine the number of t bonds in the molecule shown HO Tt bonds но- CH2CH2NH2
A: In double bond π bond = 1 In triple bond π bond = 2
Q: compound hydrogen-bonding force Between Between molecules of formula or Lewis the compound and…
A: Hydrogen atom is very less electronegative element. When it is bonded with more electronegative…
Q: Draw the simplest possible set of curved arrows that shows how the structure on the left could be…
A:
Q: MCQ 1: Diamond fom of Carbon does not conduct electricity due to the absence of A. spectators…
A: Allotropes are various structural forms of the same element and can show considerably different…
Q: 1. For each of the following molecules, draw a clearly labeled diagram showing the hydrogen bonding…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What type of orbital overlap is responsible for the n bond between carbon and oxygen in the molecule…
A: The given molecule is The electronic configuration for Carbon is [He]2s22p2. Thus, there are 4…
Q: H H H H H H H H
A: Given : We have to edit the structure of butane to make butanoic acid.
Q: 4. For the following compound, (1) draw the complete Lewis structure, including any missing…
A: The Lewis structure, hybridization, and functional groups present in the given compound has to be…
Q: Vhich of the following would be most likely to form a so Oa. oil and water Ob. wax and water O C.…
A: In the given question Solute and solvent are non polar Non polar solute and solvent would be most…
Q: Name the following %3D |3| C-OH H2N:
A: Since you have posted multiple questions, we will solve only one question for you. To get the…
Q: 4. How would the compound shown below affect plane polarized light? COOH NH2 H.
A: Chiral carbon – Asymmetric centre is a molecule having a special arrangement of ligands which is…
Q: Draw 2‑methylpropanal. Include all hydrogen atoms.
A: according to IUPAC nomenclature:
Q: a. Propane, CH,CH,CH; b. Heptane, CH;(CH,);CH; c. Propanol, CH;CH,CH,OH d. Heptanol, CH;(CH,),OH 2.…
A: Note: according to our guidelines we are supposed to answer only one question
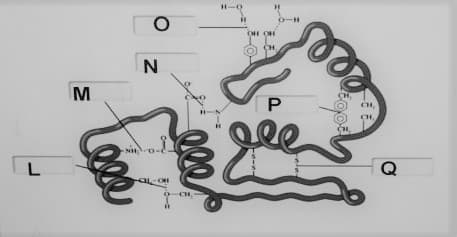
Kindly identify the noncovalent interaction between the groups on the indicated letter in the picture.
Step by step
Solved in 3 steps with 3 images

- A substance disolves well in water whcih of the following can be concluded about the substanceHand written soln only.Zn + 2 HCl ------> ZnCl2 + H2 2) When HCl(aq) is poured over the zinc metal, the liquid begins to fizz with small bubbles. What are the bubbles made of? Combustion Tests of Hydrogen –
- __ Cu(NO3)2 (aq) + __ NaOH (aq) → Type of Reaction:A student measures the conductivity of three different aqueous solutions of KNO3 using a connductivity probe. The SI units for conductivity is siements/merter (S/M). They find that the conductivity of solutions #3 is higher than the conductivity of Solutions #1, and that the conductivity of solutions #2 is the highest. What conclusion are you able to make about solutions#3?What is polarity in chemistry

