Q: Draw in all hydrogens and lone pairs on the charged carbons in each ion. a. b. d.
A:
Q: 6.|Questions pertain to the structure shown below: CH H с. HC H3C. .CH H3C° H3C 6.1 Which of the…
A: Two carbon atoms in a carbon-carbon triple bond are sp hybridized. In a carbon-carbon triple bond,…
Q: a.) Draw all the important resonance contributors for the structure shown below. Use curved arrows…
A: While drawing the resonance structures, the lone pair electrons are converted to bond pair electrons…
Q: Answer true or false. (a) In organic compounds, carbon normally has four bonds and no unshared pairs…
A: Given statements, (a) In organic compounds, carbon normally has four bonds and no unshared pairs of…
Q: 1.35 Draw a Lewis dot structure for each of the following compounds: CH CH OH
A:
Q: Which model, the ball-and-stick or the space-filling, more effectively showsthe angles between bonds…
A: Ball and stick model is a 3-D (three dimensional) model which represents position of the atoms (via…
Q: Choose the most stable structure. + 5
A: The above given structures are chair conformation of dimethyl decalin. Decalin can exist in two…
Q: 3- The bonds that would be expected for atoms forming H2O molecule and joining H2O molecules…
A: What type of bond formed in H2O and joining H2O molecules are ?
Q: What is the difference between H+ and H-?
A: The atomic number of hydrogen is 1 , this atom has 1 proton and 1 unpaired electron.Essentially…
Q: Consider the compounds below: i) CH3CHO Cii) CH3CH2CN (a) Draw complete Lewis structures for each…
A:
Q: 3. Draw a 3-dimensional structure showing the molecular geometry for each of the following. Circle…
A: inorganic compound structures can be determined by using valence bond theory and hybridization…
Q: Part B: Draw the correct structure for th appropriate number of hydrogens. (2 m
A:
Q: :0: н :0: I| || -C. H- -H —н a b H H
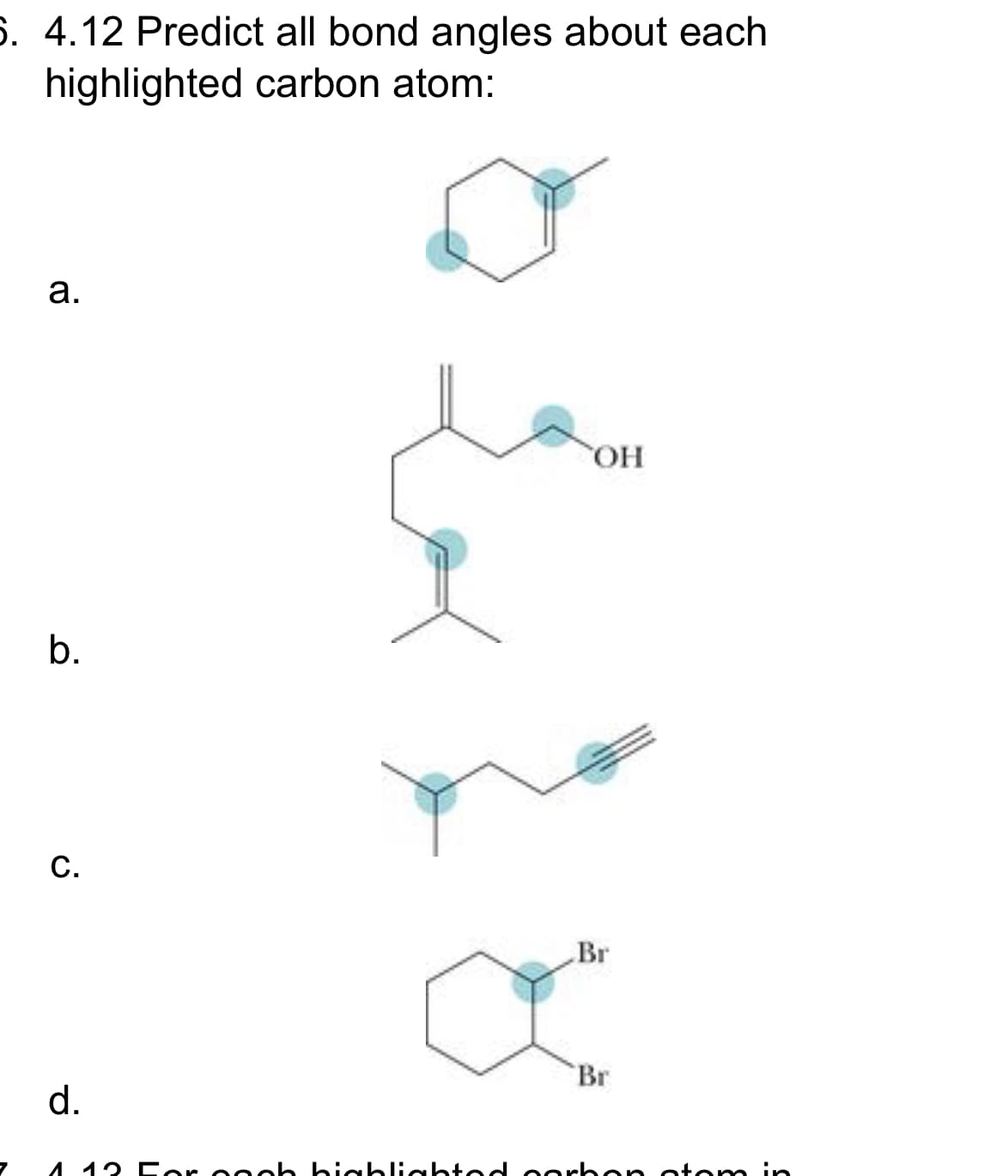
A: Angle can be determined by finding hybridization. For sp3, bond angle = 109.5° For sp2, bond angle =…
Q: 2) Draw a second resonance structure for the following structures and then an overall resonance…
A:
Q: 6b. Convert the line-angle formula below into condensed formula: Br
A: The compounds given are,
Q: How many moles of bonds between which pairs of atoms are broken during the combustion of 3 moles of…
A: The balance chemical reaction of combustion of methane gas is
Q: (a) Classify the carbon atoms in each compound as 1°, 2°, 3°, or 4°. (b) Classify the hydrogen atoms…
A: Depending on the type of attachment carbon atoms can be classified as 1°, 2°, 3°, and 4°.
Q: 5. Convert the line formulas into condensed formulas a. b.
A:
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. [3] a.…
A: Since you have posted a question with multiple sub parts, we will solve first three sub parts for…
Q: Where is the chloroethane molecule below most positive? (Carbon is grey, hydrogen white and chlorine…
A: The chemical formula of chloroethane molecule is C2H5Cl .
Q: Draw in all hydrogens and lone pairs on the charged carbons in each ion ?
A: A molecule in which carbon has a positive charge along with usually three groups attached is known…
Q: a b c CH3-CH2-0-H
A: What are the numbers of unshared pairs at atoms a, b, and c?
Q: QUESTION 4 Which of the following molecules are polar (they possess molecular dipole moment)? е. d.…
A: Answer 4.) A polar molecule is a molecule in which one end of the molecule is slightly positive,…
Q: 1) Draw the resonance structures A, B, AND C below. 1) A B + H3C
A: Complete delocalization of pie electron, in a system , it is a interaction of lone pair and pie…
Q: Structure of compound A: H CH3
A: To find : several small molecules from the compound and to give the reason for the breakage of bond.…
Q: In their neutral forms shown below, how many lone pairs are present in (a) (b) and (c) (a) (b) (c)
A: Given Neutral forms of compound.
Q: Draw a complete structure for a molecule with the molecular formula N2H2 and CH2Cl2. Include all…
A: Structure for a molecule with the molecular formula N2H2 and CH2Cl2 ?
Q: write the condensed formula for each molecules please answer 4 and 5
A: How to write to condensed formula: In compound name first substituents will come then parent chain…
Q: 2. Answer parts a-e using the skeletal structure of the molecule shown below. NB # -NC=N; NÁ a. Add…
A:
Q: MCQ 154: Double bonds, as compared to single bonds are A. shorter B. longer C. stronger D. weaker
A: Ans A) shorter C) stronger
Q: (1) a. What is the IUPAC name of the following molecule: Br CI, CI I b. Draw the bond dipole(s) in…
A:
Q: MCQ 18: The bonding pairs of clectrons in cach carbon atom A. repel B. attract C. remain intact D.…
A: In this problem, option A is correct and B, C, and D are incorrect. The order of energy is given by:…
Q: 3. The structure of Tamiflu, an antiinfluenza drug, is shown below (Organic Lett. 2007, 259). Circle…
A: Ester ,amine ,amide.,Ether and alkene five functional groups are present . And there are toatal 3…
Q: Draw a circle the compound below with the strongest C-CI bond and draw a rectangle around the…
A: The compound is mostly stabilized by the resonance effect . More is the resonance effect , more will…
Q: Calculate the number of double bond equivalencies each formula has. Blank 1: C17H16NBr Blank 2:…
A: Double bond equivalent(DBE) is also termed as degree of unsaturation. DBE is the number of molecules…
Q: Answer the following questions about erlotinib and terbinafine. Erlotinib,sold under the trade name…
A: Hybridization is a conceptual process in which atomic orbitals are combined together to furnish a…
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. a. What…
A: Hey, since there are multiple subparts posted, we will answer first three subparts. If you want any…
Q: Organic Chemistry Organic Chemistry Problems Molecular Shape and Polarity Problem 4: What is the…
A:
Q: Which model, the ball-and-stick or the space-filling, more effectively showsthe angles between bonds…
A: There are different ways to depict a molecule. For instance here, methane is shown using different…
Q: What is the approximate H−C−O bond angle in formaldehyde, H2C=O? a. 90° b. 109° c.…
A:
Q: а. CI Br b.
A: Condensed formula for the above given compound are
Q: Classify the bond shown in each molecule below as polar or non-polar: (а) CH3- CI (b) CH3- CH3 (c) В…
A: If there is electronegativity difference between the two atoms and it is in the range of 0.5 to 1.6,…
Q: a) Draw the complete structure of propane. (Draw all hydrogen atoms.) b) Draw the structure of…
A: Propane is an alkane, so it contains all single bonds. The general molecular formula of an alkane is…
Q: CI Cl2
A:
Q: Why is the H–N–H angle in NH3 smaller than the H–C–H bond angle in CH4? Why is the H–N–H angle in…
A:
Q: 1. Arrange in the order of increasing bond C - C lengths (lowest to highest) C2H2, C2H4, C2H6…
A: 1. Answer - The correct option is (e) According to the question The given compound are - ethyne ,…
Q: Convert each skeletal structure to a complete structure with all atoms drawn in. a. edit…
A:
Q: 4. Draw dipoles for each of the polar bonds in the molecules below: a. b. Но Но
A: When there is a Diffarence in electronegativity of atoms ,then the bond formed by those atoms are…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- CH3+ and CH3 − are two highly reactive carbon species.a.) What is the predicted hybridization and geometry around each carbonatom?b.) Two electrostatic potential plots are drawn for these species. Whichion corresponds to which diagram and why?Represent the bonding in each molecule or ion by drawing the orbitals (hybridized and unhybridized) of each atom in the bond. Label the σ and π bonds and label each bond by the orbitals that it is made from. For instance, the C–H bond in CH4 would be made from the overlap of: C (sp3) – H (1 s). You may draw the hybridized sigma orbitals as sticks and the unhybridized p-orbitals as lobes for clairity and ease. a. HONO b. CH3 CCH c. C3 H4 d. C2 O4 2-Polarity (a 3-d representation with bond and molecular dipoles annotated) of ClO4- , PF3, H2O, PF5 , SF4
- Consider the incomplete orbital representation of O2 , below right. a. Identify which lobes are hybrid orbitals (identify the type) and which lobes arep orbitals. b. Use dotted lines to show any bonds. c. Use up or down arrows to show electron occupation of each hybrid orbital or bond.- What orbital(s) are the lone pairs of electrons on the O atom placed? - What orbital is the lone pair of electrons on the N atom placed? - What type of bond(s) are formed between Ca1 – C? - What type of bond(s) are formed between C = O? - What type of bond(s) are formed between C – N? - What type of bond(s) are formed between N – Ca2? - What type of bond(s) are formed between N – H? - What is the angle between Ca1 – C = O? - What is the angle between Ca1 – C – N? - What is the angle between O = C – N? - What is the angle between C – N – Ca2?pls help ASAP, thanks! “rank the following bond angles from least to greatest”
- Assign Re and Si face to an SP2 hybridized carbon (C=O or C=C) Can you explain how to get it and provide a example?Complete the following table by providing theVSEPR-name and hybridisation (sp, sp2 or sp3) ofthe atom indicated by the arrow.A)mark any formal charges on the structures below, and provide the hybridization for any atom with an arrow pointed to it. B) below draw the various Newman projections listed if looking down the C2--> C3 bond
- CH3+ and CH3- are two highly reactive carbon species. a. What is the predicted hybridization and geometry around each carbon atom? b. Two electrostatic potential plots are drawn for these species. Which ion corresponds to which diagram and why?How many Cs are sp2-hybridized in the molecule shown below? (kindly show explanation or illustration. thank you!) Choices: A. 6B. 8C. 10D. 9Methanethiol, CH3SH, has a substantial dipole moment (μ = 1.52) even thoughcarbon and sulfur have identical electronegativities. Explain.

