Q: Enter electrons as e". The following skeletal oxidation-reduction reaction occurs under basic condit...
A:
Q: When it is treated with sodium ethoxide in deuterated ethanol (C2H5OD), cyclopentadiene exchanges al...
A: The structure of given cyclopentadiene is depicted as follows: The chemical formula of the above st...
Q: 4.62mL of .1510mL NaOH was needed to neutraliz 50.mL of H2SO4 solution. What is the concentration o...
A: In the given question we have to calculate the concentration of original sulphuric acid solution. th...
Q: Calculate the average H-S bond enthalpy in H2S(g) given that thestandard enthalpies of formation of ...
A: Given,
Q: 1. A cylinder containing 15.0 L of helium gas at a pressure of 165 atm is to be used to fill party b...
A: As per our guidelines we can only solve first question. Please resubmit the other questions as next ...
Q: Calculate the concentration of CO32- in an aqueous solution of 0.174 M carbonic acid, H2CO3 (aq). [...
A: Calculate the concentration of CO32- in an aqueous solution of 0.174 M carbonic acid, H2CO3
Q: Chlorine is widely used to purify municipal water supplies and to treat swimming pool waters. Suppos...
A:
Q: This question has multiple parts. Work all the parts to get the most points. a What are the IUPAC na...
A: Acetals or hemiacetal undergoes hydrolysis to from corresponding aldehyde or ketone and alcohols.
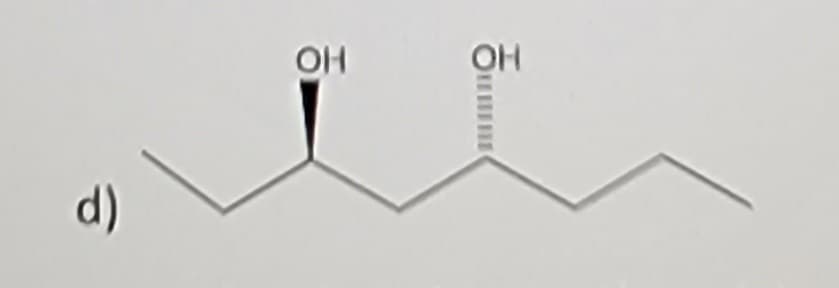
Q: Name the following alkene, including proper stereochemical designation.
A: Since you have posted multiple questions, we are entitled to answer the first only.
Q: 150 mL of polyethylene glycol has a density of 1.1333. What will be its weight?
A: Density is the amount of substance present per unit volume of solution. It can be expresses as- Den...
Q: Which of the statements below apply to the following reaction: 0. R C-OH + HNR" H. -C-NR + H,0 R- Ch...
A: Given reaction is : Options are : What are the correct options = ?
Q: Calculate the enthalpy change, AH, for the reverse of the formation of methane: Substance AH (kJ/mol...
A: Enthalpy change for any reaction is given as following formula Enthalpy change for the reaction =(su...
Q: The standard enthalpy of formation (AH) is the enthalpy change that occurs when exactly 1 mol of a c...
A: As we know enthalpy of formation of is defined as change in enthalpy associated with the formation o...
Q: Calculate the molarity of the two solutions. The first solution contains 0.300 mol of NAOH in 1.10 L...
A: Since the molarity of solution is calculated using formula Molarity = Moles of compoundVolume of sol...
Q: A 8.24 g sample of a compound contains 2.63 g of potassium, K, 2.38 g of chlorine, Cl, and oxygen, O...
A:
Q: Virtual Lau GQuesion3 . A student prepares a solution of hydrochloric acid that is approximately 0.1...
A: Applying M1V1 = M2V2 where M1 = Molarity of NaOH , V1= vol of Naoh M2 = Molarity of acid , V2= vol ...
Q: Give the noble gas abbreviated electron configuration for suifur using the box orbital method. S has...
A:
Q: The stock solution of KMnO4 has a concentration of 2.6 × 10-4 M. The pipette has a volume of 10.0 mL...
A: We have to calculate the amount of solution delivered.
Q: Show the bond angles, lone pairs and charges present of Tryptophan
A: Concept - Tryptophan (symbol Trp or W) is an α-amino acid that is used in the bio-synthesis of prote...
Q: 1. How much NaH2PO4 and Na2HPO4 in grams are needed to prepare 3.00L of 25.0 mM phosphate buffer pH ...
A: Given molarity of the buffer = [NaH2PO4] + [Na2HPO4] = 2.0 mM pH of the buffer = 6.5 pKa of H2PO4-...
Q: 7. NO2 → NO3 + NO, (Basic Solution)
A:
Q: Molecules can be paramagnetic or diamagnetic. Which of the following is INCORRECT? a. Mercury: diama...
A: Note : according to our guidelines we are supposed to answer only one question.kindly repost other p...
Q: Write the balanced chemical equation for the reaction shown. = NO NO %3D balanced equation:
A:
Q: If chlorine bleach is mixed with other cleaning products containing ammonia, the toxic gas NCl3(g) c...
A: Given the unbalanced equation is: NaClO(aq) + NH3(aq) -----> NaOH(aq) + NCl3(aq) While balanci...
Q: 2. Which of the two-cloud droplets or raindrops-would you expect to be more effective in scavenging ...
A: Answer - Cloud droplets Cloud droplets expect to be more effective in scavenging gases from the atmo...
Q: Write the balanced chemical equation for the reaction shown. = NO = NO, balanced equation:
A:
Q: A small volume within a 400 mL aqueous solution of methanol is shown in the image. Identify which im...
A: As we double the volume the molecules of methanol per unit area decrease as we have not added any...
Q: NH2 OH
A:
Q: In the IR Spectroscopy, describe the sample preparation techniques for: 1) solid, 2) liquid, 3) gas ...
A: The cells for the sampling in IR are made up of NaCl or KBr. The glass cells cannot be used for this...
Q: 3. What kind of ions are likely to form from following elements and how. a. Calcium b. Bromine c. Al...
A: Note : according to our guidelines we are supposed to answer only one question. Kindly repost other...
Q: 6. Н.О, + MnOд → Mn2+ + O2 (Acidic Solution)
A:
Q: Many metals react with halogens to give metal halides. For example, iron reacts with chlorine to giv...
A: To solve all the given questions we will use the stoichiometric ratio of the given balanced chemical...
Q: A NaCl-KCI mixture weighing 2.5005 g was dissolved in water and diluted to a total volume of 100 mL....
A: Solution -
Q: How many of the following molecules will rotate plane polarized light? H. CH, CH3 Me H. CH, CH,CH3 H...
A:
Q: A cereal contains 11.0 grams of sucrose (C12H22O11) per 60.0 grams of cereal. How many grams of cere...
A: Concept: here we can use molar concept As we know that Mole = weight/ M.W.
Q: what the three categories are. Are they talking about lone pairs, pi bonds, and sigma bonds?
A: Nucleophile is species that forms bonds with electrophiles by donating an electron pair.
Q: Q1a=> A flask contains a mixture of two gases: NO₂ and Ne. If the flask contains 0.448 mol of NO₂ an...
A: According to our policy, we should solve only one question (If any subparts of that question are pre...
Q: What is the purpose of the grignard reaction, preparation of triphenylcarbinol experiment.
A:
Q: How many moles of C3H8 contain 4.95 × 1024 hydrogen atoms? 3.09 moles C3H8 8.22 moles C3H8 1.03moles...
A:
Q: Complete the table by writing the correct formula (in the appropriate space) of the compound formed ...
A: The ionic compounds can be formed by cross multiplication of the electrovalencies of cations and ani...
Q: A width of 50 micron LDPE film with a density 0.925g/cm3 is required for producing plastic bags The ...
A:
Q: How much heat is released when 30 g of water at 96°C cools to 25°C? The specific heat of water is 4....
A:
Q: 16) Balance the following Oxidation Reduction reaction using either the half reduction method or the...
A: Redox reactions are those in which both oxidation and reduction takes place.
Q: A compound is 54.53% C, 9.15% H, and 36.32% O by mass. What is its empirical formula? Insert subscri...
A:
Q: 12. MnO, + Se?- + → MnO2 + Se (Basic Solution)
A: Solution - Step 1. Write down the unbalanced equation ('skeleton equation') of the chemical reactio...
Q: Which of the following statements is INCORRECT? O Filtration banks on the different sizes of mixture...
A: Filtration , distillation and chromatography are separation techniques.
Q: The rate of the reaction N205(g) → 2 NO2(g) + 1/2 O2(g) is measured at different temperatures, with ...
A: Arrhenius equation is the equation that gives the effect of temperature on rate constant of the reac...
Q: 23, and so on Which of the following was one of Empedocles elements (from ancient Greece)? Water O S...
A: According to the greek philosopher , Empedocles , all matters was generally composed from four eleme...
Q: Titanium metal requires light with a maximum wavelength of 286 nm to emit electrons. What is the min...
A: Given: The maximum wavelength of light required to emit electrons from Titanium metal is 286 nm.
Q: Choose the shape that matches each of the following descriptions Drag the appropriate items to their...
A: Hybridization depends on the number of lone pair + number of bond pair
Step by step
Solved in 2 steps with 3 images

- Explain why the pH of a 0.25 M solution of formic acid (HCO2H) has a lower pH than a solution that is 0.25 M in both formic acid and sodium formate (NaCO2H). Please include chemical reactions.the acidic dissociation of methyl ammonium hydrochloride, CH3NH3ClI need help with this problem please. The unknown acid type is H3A

