Q: 4. Calculate the K, given the solubility: Ag.S has a solubility of 3.4 x 10-¹7 M. Calculate K for…
A:
Q: Draw the perspective form of the newman projection from the viewpoint indicated below by the arrow
A: The compound that shows wedge (above the plane) and dash (below the plane) bonds is called as…
Q: Draw/write out the peptide bond formation mechanism. Be sure to incorporate the group that acts as a…
A: A peptide bond is a covalent bond that links amino acids together in a protein chain. It forms…
Q: Determine whether each of the five compounds in the picture attached below is predominantly…
A: Polar molecule is the one which has polar bonds like -C=O, C-Cl, C-N, N-H. Non-polar molecules are…
Q: Rank the following organic molecules in order of decreasing acidity. That is, select '1' in the…
A: Acidic strength of carboxylic acids increases with the presence of electron withdrawing group…
Q: ct Question 16 16. Which proton is least shielded? A) CHCl3 B) CH₂Cl₂ C) CHF3 D) CHBr3 B D
A: Which proton is least shielded?The protons adjacent to most electronegative atoms are least…
Q: a solution that contains 18.467 g of sucrose and 45.61 mL of water freezes at -2.21 °C. calculate…
A: Given,mass of sucrose = 18.467 gvolume of water = 45.61 mLFreezing point of the solution…
Q: Compound 1 Choose the molecule that is NOT a constitutional isomer to the other compounds in this…
A: Constitutional isomers : These are also known as structural isomers which have the same molecular…
Q: Give the IUPAC name for the following compound: Нас - 0- C-H -СН3 ±-0-1 I-U-I - I-N-I т
A: Alcohols are the compounds having -OH functional group.Ethers are the compounds having -O-…
Q: Give clear detailed Solution with explanation needed with structure..don't give Handwritten answer
A: The given structure is shown belowWe have to select all the functional groups that are present in…
Q: Draw the major product of this elimination. Consider regiochemistry and stereochemistry. Ignore…
A: This reaction is the E1 elimination reaction.At first presence of acid, protonation occurs at the…
Q: Write the empirical formula for at least four ionic compounds that could be formed from the…
A: Answer :--Here, we have to determine the charges of the ions are :-CN- :- The cyanide ion carries a…
Q: Which of the following accurately depicts the resonance for a carbonyl (A) w[a --g] (B) (C) (D) 요 오
A: In resonance, electrons are considered to be delocalized, meaning they are not confined to a single…
Q: 1. How many grams of sucrose (C12H22011) are required to make 50mL of a 1M solution? (Remember that…
A: Mass of sucrose required to prepare 50 mL , 1 M sucrose solution = 17.12 grams We have to write the…
Q: Predict what will be observed in each experiment below. experiment A student sees tiny bubbles…
A: The carbonated soft drink in the bottle contains carbon dioxide gas that is dissolved under…
Q: tate the oxidation number of the following molecules (show working and explain your answer naming…
A: Oxidation number denotes the oxidation state of an element in a compound ascertained according to a…
Q: 3. Explain in words the process of how the student can then make a 0.5 M sucrose solution from the…
A: Molarity is a concentration term. It is defined as the number of moles present in one litre of a…
Q: Cyclohexane derivatives exist primarily in the most stable of the available chair conformations.…
A: The given cyclohexane compound is a disubstituted derivative.In this compound one is small…
Q: Calculate the wavelength, in nanometers, of the spectral line produced when an electron in a…
A:
Q: A solution is prepared by dissolving 21.7 g of ammonium sulfate in enough water to make 100.0 mL of…
A: The molecular formula of Ammonium sulfate is (NH4)2SO4.So, the molar mass of Ammonium sulfate is =…
Q: Applications of nuclear chemistry
A: Given:Nuclear ChemistryWrite applications of nuclear chemistryNuclear chemistry is a branch of…
Q: what volume of a 3.9 m HNO3 must be used to make 100.0 ml of 0.75 m HNO3 solution?
A: Given : Concentration of stock solution of M1=HNO3 = 3.9 MConcentration of final diluted HNO3…
Q: The density of a solid with a melting point of 42° to 44 °C was determined to be 0.87 ‡ 0.02 g/mL.…
A: Lauric acid is a saturated fatty acid with a formula of C12H24O2. It is a white, waxy solid with a…
Q: 100 4000 4000 A 3371 2971 3299 1000 2956 2997 3000 B BAVENUMBER 2132 2000 1500 1500 C 1000 1000 D nn
A: IR spectroscopy is used to determine the functional groups present in the sample. Different…
Q: Draw the Newman projections for one staggered and one eclipsed conformation of 3,3-dichlorohexane,…
A:
Q: Identify and explain whether MgCl2 and SO2 are ionic or covalent compounds.
A: Ionic Compounds:Ionic compounds are chemical compounds formed by the electrostatic attraction…
Q: Which of these expressions correctly expresses the solubility-product constant for Ag,PO, in water?…
A:
Q: For each solute, click the button under the better solvent. H H solute H I Q H (2 CaCl₂ H H |HO— CH,…
A: Here we need to find which solute is dissolved better in which solvent. We know that 'Like dissolves…
Q: 30. A compound (C7H₁4O) has a strong IR absorption at 1715cm¹. The ¹HNMR spectrum has two signals at…
A: Given that, the molecular formula is C7H14O.The major peak in IR is at 1715 cm-1.There are two peaks…
Q: Use the following data to estimate the value of ASrxn for the represented process? HINT: watch sign…
A: Change of Entropy ∆Srxn :- the change in the state of disorder of a thermodynamic system that is…
Q: Rank these acids from most to least acidic, using the dropdowns. H₂N. A B OH C OH H-OO D
A: Given some organic molecules.Rank the acids from most to least acidic.
Q: A saline solution contains 0.770 g of NaCl (molar mass = 58.55 g/mol) in 118 mL of solution. The…
A:
Q: Using the Beer's Law plot constructed for TZ at 430 nm, please report the effective molar…
A: According to Beer's law when a monochromatic light is passed through a transparent solution, then…
Q: Multiply or divide the following measurements. Be sure each answer you enter contains the correct…
A: Answer :----1. 803.7 m / 0.89 s= 903.0337 m/s (Keep all significant digits in the division)2. 78.1…
Q: Curved arrows are used to illustrate the flow of electrons Follow the curved arrows and draw the…
A: The flow of electrons in conjugation typically refers to the movement of electrons through a system…
Q: 525 mL of water was added to 40.0 mL of 0.550 M MgCl₂ solution. Assume the volumes are additive.…
A: • The values provided in the question are:- i) Initial concentration of MgCl2 solution, M1 =…
Q: Draw the major product of this solvolysis reaction. Ignore any inorganic byproducts. CI CH3CH₂OH [B]
A: Solvollysis reaction is a type of reaction in which the solvent acts as an one of the reactant of…
Q: For the following structure, draw and indicate the low and high-energy chair conformations AND…
A:
Q: The pH of urine has been found to be correlated with diet. Acidic urine results when meat and dairy…
A: Answer:Any specie that loses H+ ion in its solution is called as Bronsted-Lowry acid and any specie…
Q: 1.Differentiate cations from anions. 2. What is the main difference between an ionic bond and a…
A: 1. Answer :---- Cations :-- Here, these are positively charged ions and they form when an atom…
Q: Fill in the electrons in the orbital box diagrams shown below for the atoms or ions specified. Note:…
A: Answer:In the given problem, we have to find out the electronic configuration of boron atom and…
Q: 2.8 The following diagram represents an ionic compound in which the red spheres represent cations…
A: Answer:Given picture of ionic compound tells that red spheres represent cation and the blue spheres…
Q: Consider the below chair equilibrium. Which chair configuration will be favored in the equilibrium?…
A: There are two chair conformations and they are remain in equilibrium. Lower the energy of…
Q: 2. Name the following aromatic compounds: NH₂ HO H
A:
Q: 1. Draw the structures with correct bonding and geometry for the molecules in the figure. 2.…
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts…
Q: Use the following data to estimate the value of AH rxn for the represented process? HINT: use the…
A: The slope of the graph can be calculated using the following formula:```slope = (y2 - y1) / (x2 -…
Q: Draw a skeletal (line-bond) structure of sec-butylcyclohexane.
A: Skeletal (line-bond) structure:-In this only bonds between carbon atoms are represented by lines…
Q: Which element is oxidized in the reaction below? Fe(CO)5 (1) + 2HI (g) → Fe(CO)412 (8) + CO (g) + H2…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: Fill in the missing information: symbol 3+ Cr atom or ion? check all that apply neutral atom neutral…
A: Any atom is made up of subatomic particles viz electron , proton and neutron . Electron carries…
Q: which atomic orbital corresponds to this set of quantum numbers in a ground state atom of boron? (2,…
A: Quantum numbers : According to quantum mechanics, there are four types of quantum numbers. The…
Give typed full explanation
draw all possible resonance structures and circle the most stable one
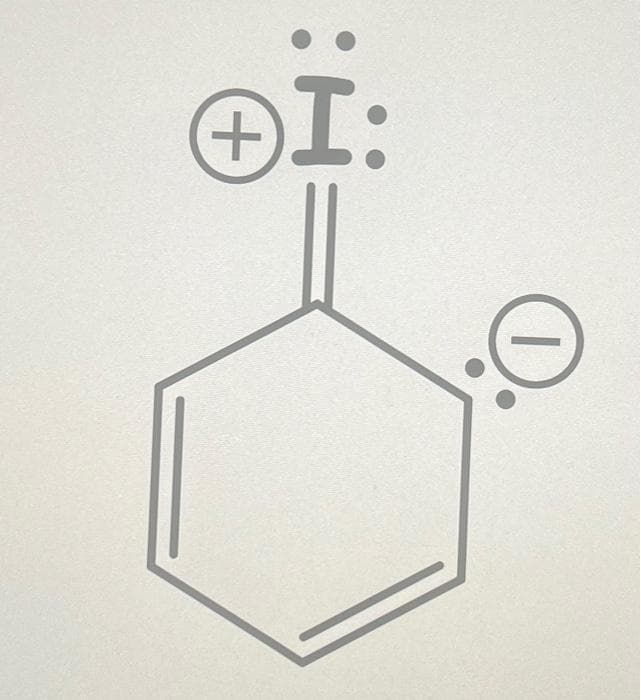
Step by step
Solved in 3 steps with 1 images


