Q: Draw the structure of organic compound m-t-butylanisole Name and Draw the structures of all possible...
A: In this question, we have to find out the correct answer of given problem by the help of the con...
Q: Use the MO diagrams to calculate the bond order for Li2+ and Li2−
A: Bond order is calculated as Bond order = NO of e in ABMO- NO of electron in BMO2
Q: Given the reduction half-reactions Fe?*(aq) + 2e- Fe(s) E° = -0.45 V > Mn2+(aq) + 2e- Mn(s) E° = -1....
A: Given reaction is Fe(s) + Mn2+(aq) = Mn(s) + Fe2+(aq)
Q: Consider the reactionCO(g) + 3H2(g)CH4(g) + H2O(g)Using standard thermodynamic data at 298K, calcula...
A: Given: The temperature is 298 K. The number of moles of CO is 2.38 g. Introduction: The entropy of t...
Q: Plz do Asap....!
A: Given data ∆G°=-107 KJ/moleR=8.314 J/mole·KK=1.02×1016 ---------------------------------------- Rela...
Q: Use the Rydberg equation to calculate the wavelength (in nm) of the photon emitted when an electron ...
A: Givenni = 7nf = 2R = Rydberg Constant = 1.09 * 107 m-1
Q: 2 Mg(s) + 3 N2 (g) --> 2 MgN3 (s) How many moles of N2 will be used to produce 115 grams of MgN3?
A: The given reaction is, 2 Mg(s) + 3 N2 (g) --> 2 MgN3 (s) We have, mass of MgN3 = 115 ...
Q: What are the [H+] and [OH-] for the following pH values? 10. = 2.33 =
A: pH is a scale to measure the concentration of hydrogen ions. The negative logarithm of hydrogen ion ...
Q: QUESTION 12 The carbon atoms in a C2H2 molecule are: sp hybridized sp2 hybridized sp3 hybridized unh...
A: Answer
Q: 2# Element Symbol Charge +, 0, Protons Neutrons electrons Carbon-13 6 Hydrogen-2 He 2 Nitrogen Oxyge...
A: An element is represented as XAz here A is the symbol of element X is Atomic mass( mass number)...
Q: A 0.88-L sample of helium is heated from 68°F to 68°C. At constant pressure, what volume (L) does th...
A: From charls law = first volume = second volume = first temperature = second tempera...
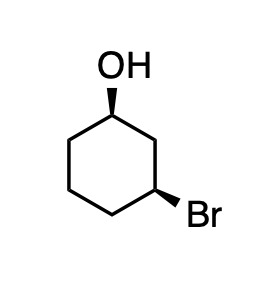
Q: Complete the following for the compound shown below. a) Draw the line structure using the wedge/dash...
A: We only answer up to 3 sub-parts. Please resubmit the question with other sub-parts (up to 3) to be ...
Q: Sodium bisulfate, NaHSO4, is used in some household cleansers as a source of HSO4- ion, a weak acid....
A: PH of the solution can be calculated with help of concentration of H+ ions in the solution.
Q: A gas cylinder contains 1.70 mol He , 1.80 mol Ne , and 1.80 mol Ar . If the total pressure in the c...
A: We have given that No.of mole of He gas = 1.70mole No.of mole of Ne gas = 1.80mole No.of mole of ...
Q: topic- around the world boiling point and vapor pressure.
A: Changes in atmospheric pressure will ulter the temperature at which water boils.
Q: Help which one is correct?
A: A Bronsted- Lowry base is that species in the reaction, that accepts a proton from the donor species...
Q: The ice fish lives under the polar ice cap where thewater temperature is 24°C. This fish does not fr...
A:
Q: For the following reaction at equilibrium, which choice gives a change that will shift the position ...
A: 1. Increase the temperature
Q: Quantum number Allowed values What does it represent? n- principal 1, 2, 3, 4, 5, 6, 7 e - angular m...
A: Quantum No Allowed Values What Does it represents n - Principal 1,2,3,4...... l - Angular mome...
Q: 5.58 A 2.30-g snmple of white solid was vaporized in a 345-mL vessel. If the vapor has a prezsure of...
A:
Q: What symbols are used to denote alpha and beta particles ? Where do these particles come from ?
A: Given Particle Alpha Particle Beta Particle Symbols of particles = To be determined
Q: topic- around the world boiling point and vapor pressure. Q- describe the motion of the water molec...
A: Interpretation - To describe the motion of water molecules as the vapor pressure increases on the b...
Q: Ka for hypochlorous acid, HCIO, is 3.0x10-8. Calculate the pH after 10.0, 20.0, 30.0, and 40.0 mL of...
A: Given: Concentration of NaOH= 0.100 M Concentration of HClO= 0.100 M Ka=3.0×10-8 To find: The pH of ...
Q: Consider the following half-reactions: Half-reaction E° (V) I2(s) + 2e- 2I-(aq) 0.535V Ni2+(...
A:
Q: 1) A gas at a pressure of 10.0 Pa exerts a force of A) 55 B) 0.55 C) 5.5 D) 1.8 E) 18 N on an area o...
A: The pressure of the gas is = 10.0 Pa The area of the surface is = 2.2 m2 The force in N is =?
Q: Question 7
A:
Q: In the picture below, the egg on the left was placed in a distilled water for 7 days. The egg on the...
A: In the left figure the egg placed in the distilled water and in distilled water there is no any ions...
Q: Discuss if the following compound will experience solvation and/or dissociation. C6 H12 O6
A: Solvation is the process in which the solute particles and solvent particles interact with each othe...
Q: Balance the following equation in acidic solution. When balanced with the smallest set of whole numb...
A: Given unbalanced reaction ; MnO4− + Br− ----> Mn2+ + Br2
Q: QUESTION 17 Which method would be most appropriate for the separation of a mixture of diethyl ether ...
A: Diethyl ether and propane - 1,3 diol both are liquids. Diethyl ether is low boiling liquid while pro...
Q: Question: The students have some wool samples that they have dyed. They want to find out how much of...
A: The concentration of coloured samples and the intensity of different wavelength or colors in them ca...
Q: Which monovalent hydroxide below supports the following data: A 0.515 g sample dissolved in 24.7 gra...
A:
Q: If AG°rxn is 10.9 kJ/mol, determine AGxn at 525 K, when TICI4(g) = 0.250 atm. %3D TICIĄ(liq) TiCl4(g...
A:
Q: If the energy in a serving of food can heat up 450,000 g of water 1 degrees C how many calories are ...
A: Calories served can be calculated using the following equation: Q = m.s.ΔT Where Q = energy per serv...
Q: The prefix hetero- meansa. same.b. different.c. both.d. solid.e. below
A: A group of letters added to the start of a word to form a new word is termed prefix. Prefixes are a ...
Q: Use the References to acce important valuen if needed for this question Does a reaction occur when a...
A: This reaction will not occur.
Q: Suggest a possible mechanism for decarboxylation of quinaldic acid?
A: Quinaldic acid, also known as quinaldate or chinaldinsaeure, belongs to the class of organic compoun...
Q: 5.55 The density of the vapor of a compound at 90°C and 720 mmHg is 1.434 g/L. Calculate the molecul...
A: The ratio of mass to volume is called density. The unit of density is g/L. The sum of the masses o...
Q: Help!
A: Ir spectrum gives information about functional groups that are present in given compound. From funct...
Q: 4) lonic vs Covalent: If ionic, give ionic formula (showing charges) & if covalent , give structural...
A: Ionic bond : The ionic bond is a type of chemical bond that involves the electrostatic force an attr...
Q: AB4(aq) + 4CD(aq) --> AD4(s) + 4CB(aq) How many grams of AD4(s) (MW= 231.11 g/mol) would be made ...
A:
Q: 1. Drag the name to the correct picture: Filtration Evaporation Distillation Decantation
A: We'll answer the first question, since the exact one wasn't specified. Please submit a new question ...
Q: Please solve Redox Reaction using molecular method MnS + HCl + HNO3 -----> MnCl2 + NO + S + ...
A: There are two methods by which we can balance the redox reaction. The methods are ion method and oxi...
Q: Please balance using the half reaction method MnS + HCl + HNO3 è MnCl2 + NO + S + H2O HMnO4+ Bi...
A: Balancing via half balance method include- Step 1: Write the unbalanced ionic equation. Step 2: Writ...
Q: Using the general solubility rules, predict the following substances as soluble or insoluble in wate...
A: Some Substances are water soluble while of other are insoluble in water. It is because of the ions p...
Q: A 335 mL sample of Oxygen gas at 25oC is heated to 50.0oC. If the pressure remains constant, what is...
A: Given, Initial Volume = 335 mL Initial Temperature = 25.0°C Final temperature = 50.0 °C Final Vol...
Q: Which of the following has the lowest entropy? a. none - all of these have the same entropy b. H2O (...
A: Entropy of the system is the measure of randomness and disorder in the system, this concept was give...
Q: Does a reaction occur when aqueous solutions of zinc chloride and potassium sulfide are combined? O ...
A: the reaction will take place and this is an example of precipitation reaction.
Q: Starting with benzene synthesize the amino acid phenylalanine 1. using the strecker approach 2. by r...
A:
Q: The electrolysis of molten AlCl 3 for 1.50 hr with an electrical current of 12.0 A produces of alu...
A: Given: Time = 1.50 hr electrical current = 12.0 A

Step by step
Solved in 2 steps with 2 images

- Fill in the blanks: cis-1,3-Dimethylcyclohexane has two different chair conformations: one withboth methyl groups in __________ positions and one with both methyl groups in ____________ positions.In the next chapter we'll look at cycloalkanes—saturated cyclic hydrocarbons—and we’ll see that the molecules generally adopt puckered, nonplanar conformations. Cyclohexane, for instance, has a puckered shape like a lounge chair rather than a flat shape. Why?a model of cyclohexane in a chair conformation, and explain why the names “axial” and“equatorial’ are appropriate.
- Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out of the paper and going behind the paper, respectively.Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.Draw the two chair conformations of cis-1-chloro-4-methylcyclohexane. Which of the conformers is more stable?
- What is the least and stable conformer among the choices below?Take a look at the butane conformers below. Identify: (a)Which is an anti conformation in Newman? (b)Which is a Gauche conformation? (c)Which is the more stable Sawhorse conformer? (d)Which has the same potential energy/strain with ALS?Perspective drawings of Chair and Boat Conformers fo Cyclohexane (C6H12). Note it is impossible to place all the carbons in the same plane without straining the bonds. Take two opposite carbons and pull both of them up to make one conformation adn then pull one of them down to make the other conformation. Can you inercovert one conformer int othe other without breaking any bonds? Explain why these represent conformers and not isomers.



