Q: A gas tank contains 213 torr ofN2.692torr ofO2was added to the tank. What is the mole fraction of…
A:
Q: Benzoic acid of mass 1.20 g is reacted with oxygen in a constant volume calorimeter to form H₂O(1)…
A: we have to calculate the calorimeter constant
Q: How would you synthesize the following molecule (1,4-dibromobutane) starting with…
A: This problem is based on general organic chemistry reaction and its mechanism. The solution is given…
Q: What is the PH at the half-stoichiometric point for the titration of 0.20M NH3(aq) with 0.50 M…
A: This problem is based on titration of acid and bases. Here a salt of strong acid and weak base is…
Q: A what type of covalent mage Sketch the two amino acids on page 126. Contrast the polarity of the…
A: Answer - Protein - Proteins are large biomolecules and macromolecules that comprise one or more long…
Q: Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid…
A:
Q: D. What is the initial cor SO₂Cl2 in the reaction shown? P-2.3 : : 0.1002 M c. Calculate the rate…
A:
Q: Draw a structural diagram (of any kind) of the following: 3-ethyl-1,1,2,2-tetramethylcyclopentane
A: IUPAC nomenclature used for the systematic naming of the organic molecules. According to IUPAC…
Q: Which is not used to determine an electron configuration for an atom? Pauli exclusion principle…
A: We have to select the option which is not used to determine the electron configuration of atom
Q: Give detailed Solution with explanation needed..complete the following reaction
A: We have been given an organic reaction and we have been asked to complete given organic reaction.…
Q: 2. Which one of each of the following pairs can form intermolecular hydrogen bonds? Explain your…
A: A hydrogen bond between two organic compounds is formed if they contain a hydrogen atom bonded to an…
Q: 196. °C. For many purposes we can treat nitrogen (N₂) as an ideal gas at temperatures above its…
A: Given : Initial temperature of gas = 89.0°C = 362 K Final temperature of gas = 143.0 °C = 416 K…
Q: Н. HO-C-H HO-C-H H-C-OH H-C-OH H₂C -OH 2 Cu2 31 но - с - н но - C-H H OH OH +Cu,0
A: Benedict's test is used for identification of reducing sugar. Reducing sugar having free aldehyde…
Q: Explain why it makes sense that most energies of formation on the data sheet are almost all negative…
A: The energy of formation, also known as the heat of formation, is the amount of energy released or…
Q: A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains…
A:
Q: What happens to the reaction below when B is removed from the reaction mixture? A (g) + 3B (g) ⇌ 4C…
A:
Q: Be sure to answer all parts. A 35.00-ml solution of 0.2500 M HF is titrated with a standardized…
A: Note - Since you have posted a question with multiple sub-parts, we will solve first three subparts…
Q: 14. 20(g) 30₂(g) O (A) K₂ O 30₂(g) K₁ 20(g) K₂ The correct relationship between the two equilibrium…
A: The equilibrium constant is defined as the ratio of the product of the concentration of product to…
Q: What is the value of ΔG when ΔH = -24 kJ and ΔS = -44 J/K at 283 K?
A:
Q: Enter your answer in the provided box. The pH of a sodium acetate-acetic acid buffer is 4.60.…
A: Henderson equation for buffer solution is pH = pKa + log [salt][acid] [salt] = [CH3COO-][acid] =…
Q: A solution consists of 1.50 M Ni(NO3)2 and 1.25 M Fe(NO3)2. Which species will precipitate first as…
A: Precipitation depends on the Ksp ( solubility product value). If the Ksp is lower in value, that…
Q: When the following bridged bicyclic alkene undergoes hydroboration-oxidation, how many produ…
A: Hydroboration-oxidation reaction is a two step hydration reaction to get an alcohol from an alkene.…
Q: A chemist measures the energy change AH during the following reaction: C₂Hg(g)+50₂(g) → 3 CO₂(g) +…
A: Exothermic reaction : ΔH = -ve or heat energy is released. Endothermic reaction: ΔH = +ve or heat…
Q: Deduce the starting materials for the synthesis of imines A and B. B. `N H -OCH 3 Deduce the…
A:
Q: OEt A Me B OTS Me C NaOEt Me Me E Me
A: We know for E2 elimination both H and leaving group must be anti periplanar to each other i.e at…
Q: Suppose a 500. mL flask is filled with 1.4 mol Of OC12, 2.0 mol of BrOCI and 0.60 mol of BrC1. The…
A:
Q: 2. Complete the table for the following isomers. Molecular Formula C3H6Br2 (isomer 1) C3H6Br2…
A: Isomers are those that have same molecular formula but different structural formula .
Q: Student Name: Experimental: Part I: (standardization of NaOH) Mass of KHP: Moles of KHP: Initial…
A: “Since you have posted multiple questions with multiple subparts, we will provide the solution…
Q: Macmillan Learning Arrange the compounds by boiling point. hexane: pentane: H₂C-CH₂-CH₂-CH₂-CH₂…
A: Since, On increasing carbon chain, boiling point will be increases.This is because on increasing…
Q: temperature of 30.0 °C is found to occupy a volume of 28.5 liters. The pressure of this gas sample…
A:
Q: Draw a structural diagram (of any kind) of the following: 1,1-diiodo-2-methyl-3-pentylcyclohexane
A:
Q: Determine the pH of a buffer solution by constructing an ICE table, writing the equilibrium constant…
A:
Q: If there are two weak acids, one with a pK, of 4 and another with a pK, of 9, an equimolar solution…
A: When an acid is dissolved in water, it can dissociate into its conjugate base and a hydrogen ion.…
Q: Which rule or principle states that orbitals of equal energy fill with one unpaired electron each…
A:
Q: What mass of gold is produced when 14.9 AA of current are passed through a gold solution for 28.0…
A:
Q: 2. Draw a Haworth structure for the B-pyranose form of the monosaccharide shown (Fischer projection)…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: -OH Na₂Cr₂O7, acetone H₂SO4
A: The product of the given oxidation reaction: An aqueous solution of sodium dichromate in acidic…
Q: OH Ag(NH3)2OH H2O
A:
Q: a. Using the average atomic mass of carbon, calculate the mass (in amu) of 138 carbon atoms. amu b.…
A: Note: As per the guidelines solution of 1st question has been made. For the expert solution of…
Q: Nrite a complete name for each compound: old IUPAC format for a/c, new IUPAC format for b/d. 1. مفید…
A: Rules for IUPAC naming:- Select the longest chain which contains carbon-carbon single bonds. Given…
Q: Think of an experiment you could perform to determine if the iron(III) thiocyanate formation…
A: Endothermic and exothermic are terms used to describe whether a chemical reaction or physical…
Q: 1.00 mol of perfect gas molecules at 27°C is expanded isothermally from an initial pressure of 3.00…
A: Given that, for an isothermal process of an ideal gas The number of moles is n = 1 mol, The…
Q: Decide whether the Lewis structure proposed for each molecule is reasonable or not. molecule…
A: Please find your solution below : Lewis structure is a representation of valence shell electrons in…
Q: 1. Predict the major product. a. b. C. CHO 8.8.8 N NH₂ NH₂ 1. EtOH, HCI 2. NaOH, H₂O NaBH3CN, HCI H…
A: We have to give the major product of the given reactions Introduction: Imine formation reaction,…
Q: What is the correct IUPAC name for the compound shown here? Br Br
A:
Q: Calculate the density of krypton gas at a pressure of 749 mmHg and a temperature of 31 °C. Express…
A: The formula of the density for ideal gas is =>density(d) = PM/RT where, P is the pressure M is…
Q: Consider the electrophilic addition of Br₂ to cyclohexene. The intermediate is a hybrid of a…
A: This question belongs to organic reaction mechanism.
Q: Draw the skeletal (line-bond) structure of diethyl ether. Select to Draw
A: A line bond structure is a type of structural representation used in organic chemistry to show the…
Q: If an average carbon atom weighs 12.01 amu, how many carbon atoms are contained in 6.36 x 10¹3 amu…
A: We have been asked to calculate number of carbon atoms in 6.36 × 1013 amu of carbon and mass of…
Q: Calculate the root mean square (rms) average speed of the atoms in a sample of neon gas at 0.14 atm…
A:
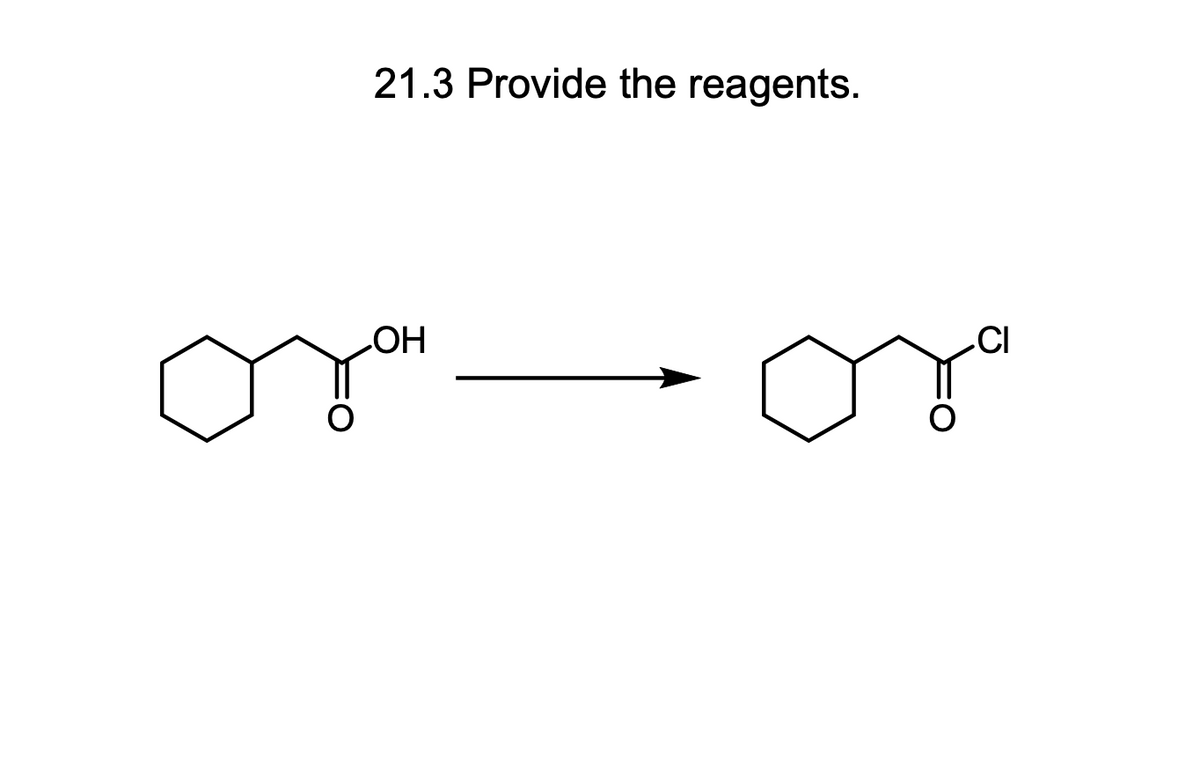
Step by step
Solved in 3 steps with 3 images

- Show how the synthetic scheme developed in Problem 23.67 can be modified to synthesize this triiodobenzoic acid X-ray contrast agent.provide the missing reagents as appropriateDevise a synthesis of benzocaine, ethyl p-aminobenzoate(H2NC6H4CO2CH2CH3), from benzene, organic alcohols, and any neededorganic or inorganic reagents. Benzocaine is the active ingredient in thetopical anesthetic Orajel
- Devise a synthesis of benzocaine, ethyl p-aminobenzoate (H2NC6H4CO2CH2CH3), from benzene, organic alcohols, and any needed organic or inorganic reagents. Benzocaine is the active ingredient in the topical anesthetic Orajel (Section 18.15C).Devise a stepwise mechanism for the following reaction, a key step in the synthesis of the antibiotic abyssomicin C. Abyssomicin C was isolated from sediment collected from almost 1000 ft below the surface in the Sea of Japan.provide the reagents necessary to complete the following information.
- Compound A is a derivative of the carbohydrate perosamine, which is found in the antibiotic perimycin. When A is treated with acetic anhydride in methanol, a monoacyl derivative B (C9H17NO5) is obtained in 73% yield. What is the structure of compound B?Devise a synthesis of benzocaine, ethyl p-aminobenzoate (H2NC6H4CO2CH2CH3), from benzene, organic alcohols, and any needed organic or inorganic reagents. Benzocaine is the active ingredient in the topical anesthetic Orajel.Amobarbital is a sedative marketed under the trade name Amytal. Propose a synthesis of amobarbital, using diethyl malonate and urea as two ofthe starting materials.



