ОН О OH ОН О H. B For the "crossed" aldol reaction that lead to product C, did the kind of base used make any difference to the reaction outcome? If so, what happened?
ОН О OH ОН О H. B For the "crossed" aldol reaction that lead to product C, did the kind of base used make any difference to the reaction outcome? If so, what happened?
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter26: Aldol And Claisen Reactions
Section: Chapter Questions
Problem 51CTQ
Related questions
Question
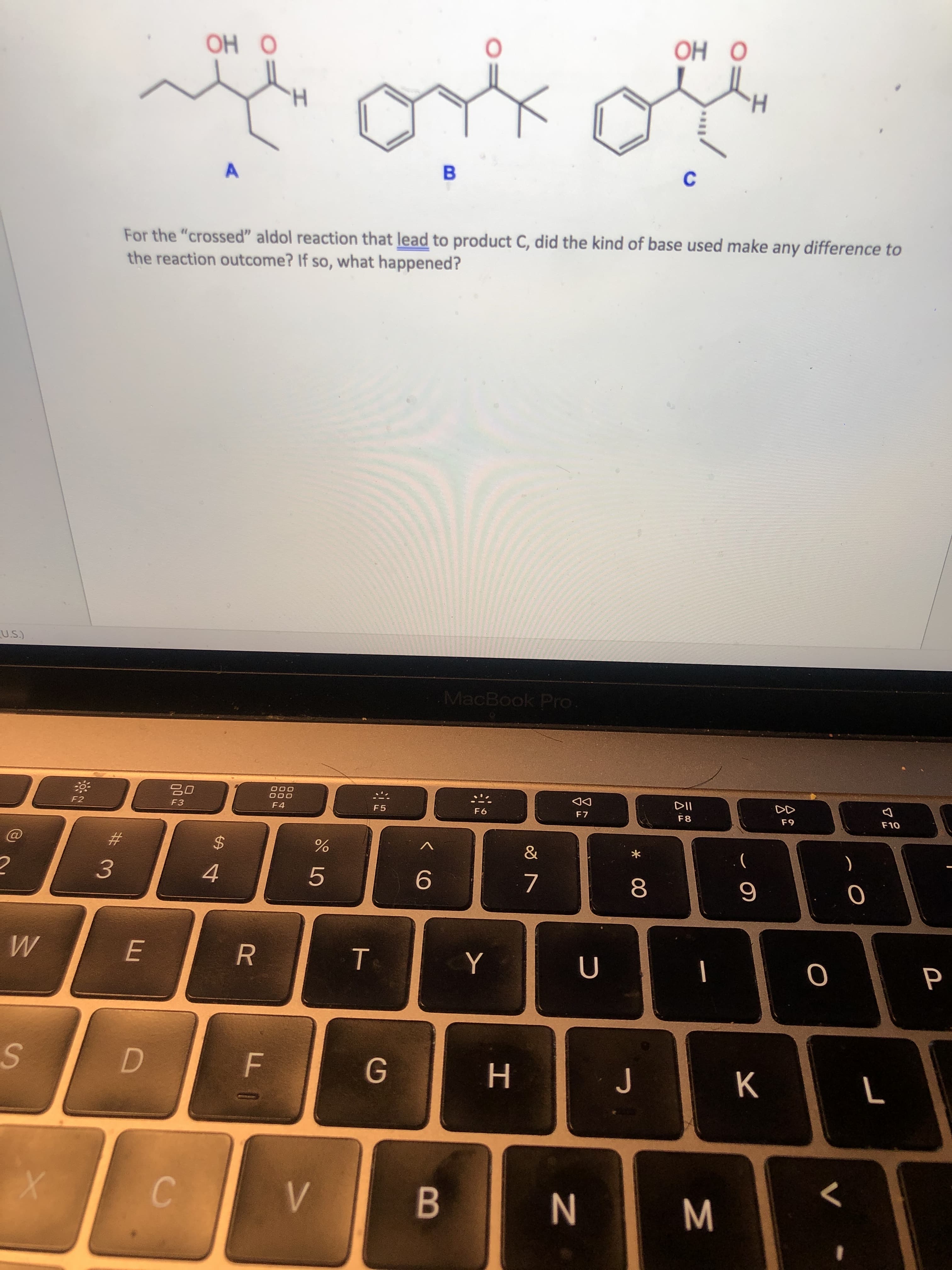
Transcribed Image Text:ОН О
OH
ОН О
H.
B
For the "crossed" aldol reaction that lead to product C, did the kind of base used make any difference to
the reaction outcome? If so, what happened?
Expert Solution
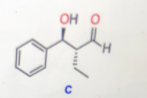
Step 1
The structure of product C is represented as follows:
Step 2
The reaction in which two same or different carbonyl compounds containing alpha hydrogen are used with a base leads to removal of acidic hydrogen and forms an enolate ion which acts as a nucleophile for other carbonyl compound which is acting as an electrophile is termed as cross aldol reaction. If same carbonyl groups are used then there will be formation of 2 products but if different carbonyl groups are used then there will be formation of four different products.
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning
