On the diagram of the filter paper below, create a particulate representation for CuCro4 after watching the demonstration video run for a period of time. Note that the positive and negative terminals of the battery are represented in the diagram by + and - symbols. Question 2 +
On the diagram of the filter paper below, create a particulate representation for CuCro4 after watching the demonstration video run for a period of time. Note that the positive and negative terminals of the battery are represented in the diagram by + and - symbols. Question 2 +
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter2: Matter And Energy
Section: Chapter Questions
Problem 83E: Particles in the illustration below undergo a chemical change. Which among the remaining boxes, a...
Related questions
Question
100%
The Structure of lonic Compounds
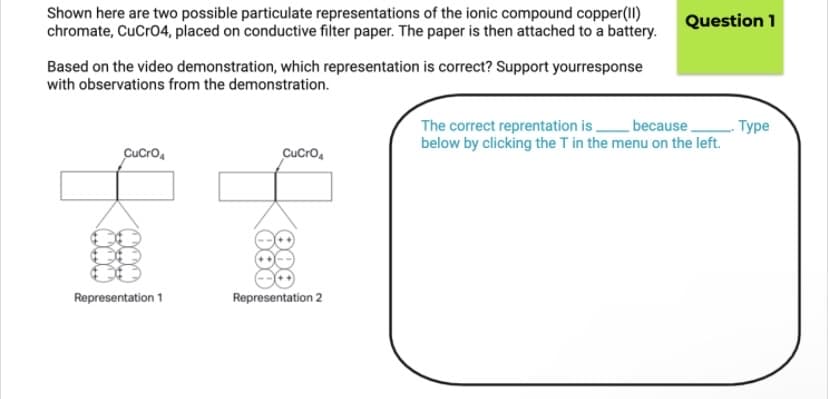
Transcribed Image Text:Shown here are two possible particulate representations of the ionic compound copper(II)
chromate, CuCro4, placed on conductive filter paper. The paper is then attached to a battery.
Question 1
Based on the video demonstration, which representation is correct? Support yourresponse
with observations from the demonstration.
because,
The correct reprentation is .
below by clicking the T in the menu on the left.
. Туре
CuCro,
CuCro,
Representation 1
Representation 2

Transcribed Image Text:On the diagram of the filter paper below, create a particulate representation for
CuCro4 after watching the demonstration video run for a period of time. Note that
the positive and negative terminals of the battery are represented in the diagram
by + and - symbols.
Question
2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning
