Q: 12. Label A through H on the phase diagram below. P A H D B E F C G T
A: We have ask to label the phase diagram from A to H. The answer to this question is given in…
Q: Find the mass of chromium in the spoon
A: we have to calculate the mass of chromium in the spoon
Q: A quantity of 0.0522 mol of a gas initially at 0.0500 L and 20.0 °C undergoes a constant-temperature…
A:
Q: 2. Considering your response to Question 1, as well as other data in your table from Part 1 and 2,…
A: As the reaction proceeds, [Zn2+] in the anodic compartment increases as the zinc electrode…
Q: 10. Name the following molecules. a) b) c) CH3 CH3 BrCH₂CHCH₂CCH₂Br OH CH3CH2 Br & CI CH3…
A: Naming of the given compounds is done according to rules set by IUPAC.
Q: Calculate the volume of 50% (w/w) sodium hydroxide solution needed to prepare 0.5L. of a 0.05M…
A:
Q: A popular car has an engine that is reported to have a volume of 5840 cm³ the size in cubic inches ?…
A: Answer: Given that,
Q: Match the following types of mixtures: Heterogeneous, will not settle Heterogeneous, will settle…
A: Solutions are classified into 3 types 1)Solution: It is always transparent, light passes through…
Q: н 1.) (з 118 52 2.) DBU Li Т.) CH3BL 2) HT
A: 1,3 propanedithiol(C3H8S2) is used for the protection of aldehyde and Ketone.It is used for…
Q: Cooper, Zach, Nate did this experiment dissolving 2.55g of Cu(C2H3O2)2 x H20 and adding 5.85g…
A:
Q: A compound is composed of only C and H. If 42.8475 grams of the compound contain 36.03 grams of C,…
A: Given: Compound is composed of only C and H mass of compound = 42.8475 g mass of C = 36.03 g
Q: Write 7.08 x 10 -4 in standard notation.
A: The standard notation is also known as scientific notation and when the value is represented in the…
Q: Question 5 The solubility of O₂ in water is 5.7 mg/L at an atmospheric pressure of 1.00 atm and…
A: Henry's Law formula is KH = solubility in mole / Lpartial pressure of gas partial pressure = mole…
Q: 9. A 0.250 L sample of blood plasma was found to weigh 0.25625 kg. What is the density (in g mL-¹)…
A: “Since you have posted a question with multiple sub-parts, we will provide the solution only to the…
Q: Procedure and Data Density of Volatile Substance 1. Measure the mass of the empty glass beaker to…
A: 5. Given,Volume of Isopropyl alcohol = 5.00 mLMass of solvent from each addition = 4.54 g Trial 1…
Q: Calculate the number of hydrogen atoms in a 130.0 g sample of ammonia (NH3). Be sure your answer has…
A: Given data, mass of ammonia = 130 g Molar mass of ammonia = 17.03 g/mol We have to find out the…
Q: TO McGraw Hill Campus ||| Calculate the Molar Mass of Acet X +…
A: A mole is defined as the amount of a substance that contains 6.022 X1023 entities like particles,…
Q: Convert 36 kcal to J
A: Given : 36kcal 1 k = 1000 1 cal = 4.184 J Therefore 1kcal = 4184 J
Q: A sample of Krypton occupies 75.0 ml at 0.400 atm the temperature remained constant what volume…
A:
Q: A mixture of helium and argon gases, in a 9.92 L flask at 25 °C, contains 0.703 grams of helium and…
A: Answer: This question is based on Dalton's law which states that the partial pressure of a gas in a…
Q: A chemist adds 95.0 mL of a 0.26M aluminum sulfate (Al₂(SO4)3) solution to a reaction flask.…
A:
Q: Give typed solution not written How many milligrams in 1.26 scruples? 3 scruples = 1 dram 1…
A: How many milligrams are in 1.26 scruples
Q: mass of magnesium (g): Initial gas volume (ml): Final Volume (mL): Temperature (°C): Atmospheric…
A: The given data is- Atmospheric pressure = 30.59 inHg ∆h (cm of water) =12.98 Cm of water is a unit…
Q: Think about the reaction Na₂CO3(aq) + H₂SO4(aq) NaOH(aq) + H₂SO4(aq) NaCl(aq) + HC₂H₂O₂(aq) clear…
A:
Q: One likely application of nanotechnology is the construction of very small containers (Tony Stark’s…
A: Solutions- Let's call these small containers nanocells. The simplest of these cells would mimic red…
Q: What is the total concentration of ions in 0.20 M Al2(SO4)3 solution? O a. 0.5 M O b. 0.1 M C. 1.0 M…
A: we have to calculate the total concentration of ions in 0.20 M Al2(SO4)3 solution
Q: A 0.5700 g sample of impure Ca(OH)2 is dissolved in 50.00 mL of water. 20.00 mL of the resulting…
A: We have to use the mole concept to calculate the mass or number of moles of Ca(OH)2 neutralize by…
Q: Draw the organic product formed when cyclopentene reacts with iodobenzene in the presence of…
A: This is an exaple of Heck reaction . The Heck reaction is a type of chemical reaction that couples…
Q: When 224. mg of a certain molecular compound X are dissolved in 45.0 g of cyclohexane (C6H₁2), the…
A: As we know, Depression in freezing point, Tf° - Tf = i * Kf * mWhere Tf° & Tf are freezing…
Q: 6 29) What is the major organic product obtained from the following sequence of reactions? Br- -NH₂…
A: Aromatic amine reacts with sodium nitrite and hydrochloric acid at 0oC temperature to form a…
Q: Predict which of the following substances would have the highest boiling point. HBr, Cl2, Kr…
A: Boiling point: The temperature at which the vapor pressure of a liquid becomes equal to the…
Q: The force constant for the diatomic H2 is 575 N m-1. Calculate the fundamental vibrational frequency…
A:
Q: # H 1.) H₂ (CH₂)₂ SH 2)DBULI action 1.) C3 HB 52 2.) BULI Cs H₂o O Ht 1.) C5 H₁0O H30
A: Given first reagent is 1,3-dithiol , which is used for protection of aldehyde and ketone and after…
Q: 40) What is the formal charge of nitrogen in a) 1 b) 2 c)-2 45 d) 3 e) -1
A:
Q: $ 1.04 ▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬▬ 1 L (number) (number) (unit) (unit) 11 A /gallon
A: $ 1.04 / 1 L x ( 3.79 L / 1 gal ) = $ 3.94 / gal
Q: What is the pH of a 0.02 M solution of dipotassium phosphate, K₂HPO4?
A: Concentration of K2HPO4 =0.02M
Q: Write the correct empirical formula for hydrogen peroxide, H₂O2
A: Given: molecular formula of hydrogen peroxide is H2O2. As we know, empirical formula of a compound…
Q: A compound is composed of only C and H. If 42.8475 grams of the compound contain 36.03 grams of C,…
A: Compound contains C and H atoms. Compound is a hydrocarbon. % composition of C = mass of C in…
Q: From the information that follows, write the isotopic notation for each atom, then decide which one…
A: We have been given isotopic notations for each atom and we have been asked to decide which one is…
Q: The gas in the discharge cell of a laser contains (in mole percent) 11% CO2, 5.3% N2, and 84% He.…
A: We know density can be calculated using the formula - DRT=PM Where D=Density(g/L)…
Q: Vanadium crystallizes in a body centered cubic structure and has an atomic radius of 131 pm.…
A: Atomic radius of Vanadium = 131 pm Vanadium crystallizes in a body centered cubic structure Density…
Q: What is true for this chemical reaction: 3CO(g) + Fe₂O3(s) = 3CO₂(g) + 2Fe(s)? Select an answer and…
A: This problem is based on Thermodynamics. Here we will see the feasibility of the reaction and then…
Q: The synthesis reaction producing mathane gas from its elements.
A: Given : production of methane gas
Q: wynom grivieto sa shilqizib bns triguor omor se gold Product Agcl(s) + 2NH₂ [Hg (NH₂)₂] +Cli (P2)…
A: Qualitative analysis determines the identity of substances present in the mixture, whereas…
Q: 2. Measure the mass of each coin separately and record the masses below. Why were several coins used…
A: This problem is based on the analytical chemistry. Here we will be calculating density of the coins.…
Q: if the titration of a weak acid with a base was done, the solution at the point would be? a) neutral…
A: The equivalence point is the titration point at which the volume of titrant added is enough to…
Q: CH3CH₂CH₂CN • Draw in all the hydrogens for the following structure HO NH₂ CI
A: -> Expanded structural formula shows all of the bonds connecting all of the atoms in the…
Q: What is the relationship between the following pair of molecules? s s Identical Enantiomers O…
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: From the data collected for reaction Aproducts determine: a. Order with respect to A b. Rate…
A: We will determine order of reaction first, then using it find rate constant.
Q: NaOH 11,0 Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and…
A: NaOH is a strong base. Where Br is a good leaving group.Thus in presence of NaOH, Br group leaves…
Propose a synthetic pathway for the transformation shown.
Please only provide typed solution no handwritten solution needed allowed
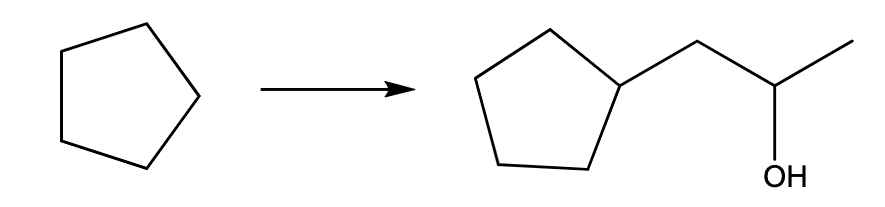
Step by step
Solved in 2 steps with 1 images


