Q: Choose the correct functional group present in each structure below that would dictate how it would ...
A: Given, some functional groups
Q: Explain the origin of quantization from Max Planck and Einstein. Give necessary formulae
A:
Q: 18. The equilibrium constant for the following reaction is 0.18 at a set temperature. Find the equil...
A:
Q: A solution of 2.165 grams of compound Y per 1000 grams of water at 20C has a vapor pressure of 2.165...
A:
Q: Titration is lab method of find the Hydrogen ion concentration of several acids and basis. A 35 .00m...
A:
Q: What is the molality of 14.5 g of MgSO4 in 285.5 mL of water at 40 °C?
A: given mass of MgSO4 = 14.5 g volume of water = 285.5 ml
Q: A swimming pool measures 45.0 meters by 30.0 meters. How many grams of water are needed to fill the ...
A: The density of water is = 1.00 g/mL The dimension of the pool is = 45.0 m × 30.0 m × 7.20 feet The m...
Q: 13. Identify the weakest acid. HCN Ka = 6.2 × 10-10 Ka = 1.8 x 104 а. b. HCOOH с. HNO2 d. HF Ka = 7....
A: more the value of Ka higher will be the acidity of that molecule and inverse is true for weak acid. ...
Q: molecular form
A:
Q: Part A Two substances, A and B, initially at different temperatures, come into contact and reach the...
A: Mass of substance A = 6.13 g Initial temperature of substance A = 20.1oC Mass of substance B = 25.8 ...
Q: What would be the equilibrium potential for the ion Ca2+ in a cell be if (Ca2+Jout=15 mM and [Ca2+Ji...
A: The equilibrium potential of ion is given by Vion = RT/nF ln[ion]out/[ion]in here, Vion = equilibriu...
Q: Which of the following compounds is INSOLUBLE? O potassium acetate O lithium carbonate O magnesium b...
A: Given, Potassium acetate, lithium carbonate, magnesium Bromide, aluminium sulfide all are ionic com...
Q: Sample A weighs 30.00 g and displaces 15.00 mL of water. Sample B weighs 20.00 g and displaces 30.00...
A: Given, Mass of sample A = 30.00 g Volume of sample A = 15.00 mL Mass of sample B = 20.00 g Volume ...
Q: How do snow crystals form chemically?
A: A snowflake begins to form when an extremely cold water droplet freezes onto a pollen or dust partic...
Q: 12.At the applications have high vibration.. is most useful 13. Sphere or nodular graphite cab be pr...
A:
Q: Which is expected to have the lowest vapor pressure? A) CH, B) SiH, C) NH3 D) PH3 E) H,0
A: When the molecules in liquid sate has strong intermolecular forces of interaction, then they evapora...
Q: Without doing a calculation, predict whether your estimate in part (a) is more negative or less nega...
A: Most of the elements or compounds have generally three states such as gaseous state, liquid state a...
Q: What is the classification of the compound shown below? ÇH,OH H-C-OH CH,OH A) L В) D C) A sugar this...
A: The given compound consist of -OH functional group. In the given compound, -OH group is present in t...
Q: Following is the structure of pyridoxamine. CH2NH2 CH2OH но. H3C N. a) Which nitrogen atom of pyrido...
A:
Q: 7- Find kinetic eneray of T He in flsion dlactions 15 12 4. N+H C+ 'He
A: Nuclear masses of reactants and products : Reactants : Mass of N-15 = 15.000109 u Mass of H-1 = 1...
Q: Part A Mothballs are composed primarily of the hydrocarbon naphthalene (C10 Hg). When 1.094 g of nap...
A:
Q: 32. The illustration below graphs energy during the formation of a covalent bond. Where can you iden...
A: When we give heat or light energy to any process, then the process will be endothermic.
Q: All changes save W 31. Why does increasing the temperature of a chemical reaction speed up the react...
A: The speed of a reaction is known as its rate. The rate is dependent on many factors such as concentr...
Q: Referring to the attached information, select the TRUE statements: (I) The bromide ion acts as a cat...
A: The TRUE statements are (I) The bromide ion acts as a catalyst (IV) The maximum point of the graph...
Q: First, add curved arrow(s) to show the resonance using the following pattern: a pi bond between two ...
A: Resonating structures are used to express the stabilities of compounds having alternate double bonds...
Q: Clements, Compounds, and Mixtures irections: Classify each of the pictures below by placing the corr...
A: Element means atoms of single type. Compounds are formed by combination of atoms obtained by chemic...
Q: Classification of Reactions Part 1: Classify the following reactions as synthesis, decomposition, si...
A: Different types of reaction: a) Precipitation reaction: In precipitation reaction an insoluble salt ...
Q: In the given heat curve below, which of the following shows the heat capacity of the gas? gas liquid...
A: Heat capacity of gases is defined as the amount of heat required to raise the temperature of one gra...
Q: Which of the following molecules has a net molecular dipole? X X X F F. F H H F. F F H H H н н F F 1...
A: The following is a depiction of the provided molecules:
Q: What amount of heat, in kJ, is required to vaporize 115.00 g of ethanol (C,H,OH)? (AHvap = 43.3 kJ/m...
A: The given weight of ethanol = 115 gram DelHvap = 43.3 kJ/mol
Q: How much heat (in kilojoules) is evolved in converting 1.00 mol of steam at 130.0 °C to ice at -45.0...
A: Number of moles of steam = 1 mol Mass of steam = 18 g Initial temperature of steam = 130oC Final tem...
Q: TRUE/FALSE. Write 'T' if the statement is true and 'F' if the statement is false. 16) The carbohydra...
A: Disacharides are the carbohydrates which is composed of two monosaccharide that are bonded to each o...
Q: bottle. The bottle contains 3.78 L of ethanol. The bottle contains 2 What is the density of ethanol ...
A:
Q: s. Ethane reacts with oxygen in a combustion reaction described by the unbalanced equation C,H6le) +...
A: Balanced equation means number of each atom in both side are present in equal number.
Q: You may find additional useful data in the ALEKS Data tab. Does the following reaction absorb or rel...
A: When we add an electron to the outermost shell of a gaseous atom then heat is released and that heat...
Q: Calculate the potential in the solution in the titration of 50.0 mL of 0.100 M Fe2+ in 1.00 M HCl4 w...
A:
Q: In the heating curve below, the heat flow into the sample in the segment will yield AHvap of this su...
A: When we add the amount of heat to change a liquid to the gaseous phase then that energy will be the ...
Q: A compound containing carbon, oxygen, and hydrogen was found to have an empirical formula of CH,0. T...
A: Empirical Formula is CH2O Formula mass of CH2O = 1 x Atomic mass of C + 2 x Atomic mass of H + 1x At...
Q: Given in the table the molecular mass and dipole moment of the five compounds, which should have the...
A: A higher dipole moment can mean a larger attraction between molecules if the size of the molecules s...
Q: 39. What amount of phosphorus produces 8.80 mol of phosphorus trichloride? P4l6) + 6Cle) →4PC1316) а...
A:
Q: 4. Predict whether each of the following substitution reactions is likely to be SN 1or SN2. CI OAC C...
A:
Q: In which region of the IR spectrum would a ketone give a very characteristic band? A) 4000-3000 cm1 ...
A: 4000- 3000 cm-1 .In this region possible groups are -OH ( alcohol) ~ 3600- 3200 cm-1 -NH( amine ) ~...
Q: Study the reactions for the formation of compounds from their elements. 1. C (s) + O2 (g) → CO2 (g) ...
A:
Q: Symbol 35 Cl Protons 26 50 Neutrons 30 66 Electrons 49 86 Mass no. 222 119
A:
Q: 5) In the adsorption of N2 at 90.1 K on a certain solid, the following volumes of gas, reduced to st...
A: The BET equation is given by, where p and p0 are the equilibrium and the saturation pressure of ads...
Q: RiSK CONTROL CONTROL HIERAROY COTRL ELIMINATE YOU SYSTEMS HAZARD ISOLATE t HALMD CONTRILS USE PPE RI...
A: Risk control refers to some set of rules that are used by us to evaluate the potential damage that c...
Q: Explain the 2 trends of atomic radius in the periodic table. state the trend and explain why
A: Atomic radius decreases on moving from left to right ( across a period). Because on moving from left...
Q: Round this addition of measurements to proper sig figs: 23 cm + 111.24cm + 0.0045 cm
A: At the time of addition , round off to minimum decimal number.
Q: Question 32 ( state the trend and explain why Explain the 2 trends of atomic radius in the periodic ...
A: atomic radius is defined as the distance between the centre of nucleus of an atom and outermost shel...
Q: Classify the following as a type of matter: Liquid Gold
A:
Convert the line structure to a Newman projection. Select the Newman projection that matches your answer.
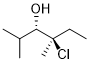
Step by step
Solved in 2 steps with 2 images


