Q: The change in internal energy for the combustion of 1.0 mol of octane at a pressure of 1.0 atm is 50...
A: Given : Change in internal energy = 5084.3 kJ Change in enthalpy = 5074.1 kJ Pressure = 1 atm No. of...
Q: :Using the following data .7 Fe3+ +3e- Fe E° = -0.036 V Fe2+ +2e-- Fe E° = -0.447 V The standard cel...
A:
Q: Equation writing. balance the following redox reactions using change in oxidation state method (Vale...
A:
Q: 1.1 and 1.2
A: Molar mass can be calculated by getting each element atomic mass and multiply to the index then add ...
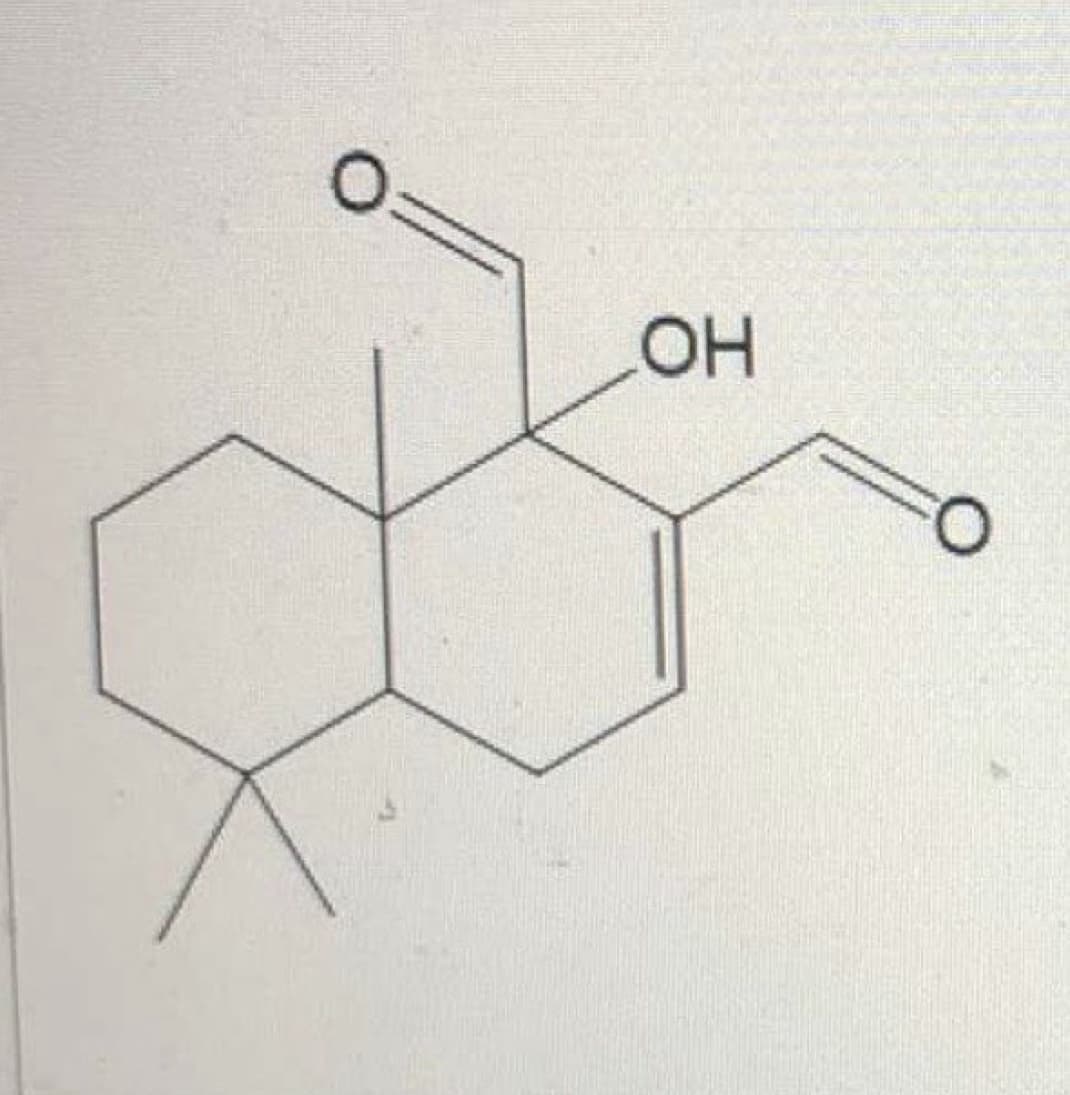
Q: What is the correct IUPAC name of the structure presented.(Follow this format “Branch-Carbon Prefix-...
A: Solution- IUPAC name is given below of the above given structure -
Q: Can someone answer this?
A: Given: The mass of the vinegar=5.00 g The concentration of NaOH=0.108 M The volume of NaOH=39.1m L=3...
Q: Consider the following system at equilibrium at 698 K: 2 HI (g) H2 (g) + I2 (g) When some H2 (g) is ...
A: Given chemical reaction is - 2HI(g) = H2(g) + I2(g) When extra H2(g) is added then the reaction wil...
Q: Consider the reaction: 4 K(s) + O2(g) 2 K2O(s) The molar mass of K is 39.10 g/mol, and that of 0, is...
A: Limiting reagent: It is defined as the reactant in a chemical reaction which is present in lesser am...
Q: What is the mass of 3.30 x 1023 atoms of silver, Ag? 25.8 g 59.1 g 85.7 g 197 g 0.549 g
A: We will use formula of mole and mass to get answer. Number of atom = 3.3×1023
Q: How many mg of magnesium chloride are needed to supply 2.2 MEQ of positive ions (mg+2) 1 mol + charg...
A: Solution- 1mol + charge=1000mEq Mg2+1.7mEq Mg2+=2.21000 mol Mg2+ =2.2×10-3 mol m...
Q: Draw the most stable conformer of the following molecule. (A solid wedge points out of the plane of ...
A: The carbon ( C ) atom which are bonded to the four other groups is called asymmetric C-atom. CHIRAL ...
Q: What is a suspension, and how does it differ from acolloid?
A: A suspension is a heterogeneous mixture of a finely distributed solid in a liquid. The solid is not ...
Q: Redox Reaction BALANCE THE EQN. show solution Mn2O3(s) + MnO4(aq) --> MnO3 (s) + Mn+2(aq)
A:
Q: The coefficients on balancing the following reaction will be. Pb(OH)2 + HCI H2O + PbCl2 a) 1,2,2,1 b...
A: For a balanced chemical equation no. of elements in reactant side must be equal to product side . So...
Q: In the reaction A -> B, the plot of 1/[A] versust resulted in a straight line with equation (1/[A] =...
A: Since we know that - In 2nd order equation - plot of 1/At vs t gives straight line graph
Q: 1. what does a low enthalpy of fusion for a solid tell you about the solid (its bonding or type)?2. ...
A: Enthalpy of fusion is the enthalpy change that is associated with phase change of a substance from ...
Q: In this reaction, the limiting reagent is Question 6 options: None Al...
A: Dear student I have given answer to your question in the image format.
Q: A 1-kg cylinder of aluminum and 1-kg jug of water, both at room temperature, are put into a refriger...
A: The specific heat of a substance is the ratio of the quantity of heat required to raise the temperat...
Q: Calculate the solubility of silver acetate.1 (CH;COOA9) in water at 25 °C, assuming * .the DHLL to a...
A: The solubility product constant is the equilibrium constant for the dissolution of a solid substance...
Q: The following reaction can potentially produce three different cyclized products. 1. NaOMe, MeOH MeO...
A: Cyclic Product formation 8 - Membered 6- Membered and 6 Membered
Q: Q5. Draw the Lewis structure, determine the electron domain geometry, and predict the molecular geom...
A: Lewis structure is a representation of valence shell electrons in a molecule.
Q: Astanderdigetion of NGOH dolution Trial I Trial 2 بلمن Deight of KHP,g 0. 1012 O. 1009 ০:০৭৭৪ Inidia...
A:
Q: A chemistry graduate student Is given 450. mL of a 1.00M chlorous acid (HCIO,) solution. Chlorous ac...
A:
Q: Which one of the following statements about the following reaction is false? CH4(g) + 2O2(g) CO2(g)...
A: We have given the balance chemical equation CH4(g) + 2O2(g) → CO2(g) +...
Q: What is the freezing point of a water solution with the total volume of 1.00 L, that is made by diss...
A: Normal freezing point of water = 00 C Density of water = 1Kg/L
Q: Show complete calculationsCalculate the pOH, H3O+, OH- if the pH is: 1. pH = 3.75 2. pH = 2.5
A:
Q: Explain When fluorine gas combines with water vapor, the followingreaction occurs.2 F 2 (g) + 2 H 2 ...
A: If we assume that all the gases are ideal gases, we can describe the coefficients of this reaction a...
Q: What is the compound produced before dehydration step in hydrate experiment? Select one: O a. Anhydr...
A: We have to find the what is the compound produced before dehydration step in hydrate experiment. Ans...
Q: How much work (in J) is required to expand the volume of a pump from 0.0 L to 2.5 L against an exter...
A: The formula used to calculate the work required to expand the volume of gas is: w = -pext × (V2 - V1...
Q: Avogadro's number: a. Changes depending on the quantity that's being measured b. Is equal to the num...
A: Avagadro number is equal to 6.022 x 1023 (atoms or ions or molecules) per mole of that ( element or ...
Q: Can you plz help me with these thankyou so much
A: In this question, we have to find out the correct answer of given problem by the help of the con...
Q: Reaction of butane (CH 3CH 2CH 2CH 3) with Cl 2 in the presence of light forms two different alkyl c...
A: Interpretation - Reaction of butane with Cl2 in the presence of light forms two different alkyl chlo...
Q: The electronic configuration of elements of Group IV (4A) ends with * O ns2 np2 O ns2 nd7 O ns2 nd5 ...
A: Solution
Q: Match the items. а. b. С. 1. continuous spectrum a 2. emission spectrum a 3. absorption spectrum a >...
A: Spectrum: The range of all types of electromagnetic radiation is known as the electromagnetic spectr...
Q: 1. What changes in color occur when bromine reacts with an alkene?
A: Using concept of bromine test .
Q: a. Calculate the pH for a buffer with 0.20 M NH3 and 0.20 M NH4Cl, then calculate the pH after 10.0 ...
A: b.
Q: What is the difference between a solute and a solvent?
A: Solution- solvent - Major part of the solution are called solvent also we can say that the substan...
Q: Calculate the pH for a buffer with 0.20 M NH3 and 0.20 M NH4Cl, then calculate the pH after 10.0 mL ...
A:
Q: Chemistry
A: Atomic radius decreases along the period and increases along the group.
Q: What are the similarities between Arrhenius, Brönsted-Lowry, and Lewis Theory for acid-base? use you...
A: ARRHENIUS THEORY Arrhenius's theory defines acids as substances that dissociate in water to give hyd...
Q: The quantum yield of light-induced chemical reactions (called photochemical reactions) measures the ...
A: Interpretation - The maximum amount (in moles) of CH3X that breaks apart is to be explained. Giv...
Q: 25. At 40°C, H,O,(aq) will decompose according to the following reaction: 2H,0, (aq) 2H,0(1) + 0,(g)...
A: Average rate for decomposition is a negative value and for formation is positive.
Q: List the characteristics of chemicalequilibrium.
A: Characteristics of chemical equilibrium : 1.Chemical equilibrium is dynamic in nature. 2. Chemical ...
Q: What theory did John Dalton formulate?
A: John Dalton is an English Chemist, known as Father of atomic theory.
Q: Enter the sequence of one-letter codes (all upper-case, capital letters with no spaces!) which descr...
A: The question is based on the concept of biomolecules. we have to write the name of the pentapeptide ...
Q: The LiH dipole torque is 1.964 * 〖10〗^(-30) Cm and the bond length is 1.596 A. Find the percentage o...
A: Given Dipole Torque = 1.964 * 10-30 CmBond length = 1.596 A° = 1.596 * 10-10 m
Q: What is the value of k for order reaction whose ha 2.37 minutes if [A]O= O.1 O k=0.1 sec-1
A: We will use relation between t1/2 and K
Q: For the following categories of elements, state the possiblenumber(s) of electrons in their outermos...
A: The electron configuration of an element describes how electrons are distributed in its atomic orbit...
Q: Describe the characteristic movement of the particles of solid, liquid and gas and arranged them i...
A:
Q: What is the value of k for a first order reaction whose half life = 3.27 minutes if [A]O= 0.1 M * O ...
A: We have to calculate k(rate constant). This above formula is used for first order reactions. H...
Step by step
Solved in 2 steps with 1 images


