Q: Very confused between balanced redox equation and reduction potential equation. Write a balanced r...
A: Reduction:- It is the gain of electrons by an atom or ion. Oxidation:- It is the loss of electrons ...
Q: Question from at-home practice questions: Use the IR spectra attached below. Is a carbonyl present? ...
A:
Q: Determine the solubility of the ions that is calculated from the Ksp for Ag2CrO4. A) 2S³ B) S3 C) 4S...
A: Ksp of Ag2CrO4
Q: If (+)-(2R)-methyl-(1S)-cyclohexanol with a specific rotation of +28 degree. What is the observed ro...
A: The specific rotation of (+)-(2R)-methyl-(1S)-cyclohexanol is = +28 degree The enantiomeric excess o...
Q: The concentration of Ba(103)2 in H2O at 35°C (Ksp = 3.00 x 10- %3D Ba(103)2 (s) - Ba²* + 2103 5.23 x...
A: Ksp = 3.00 × 10-9 Concentration of Ba (IO3)2 = ?
Q: To determine the amount of sodium citrate that gives the maximum solubility of one gram Aspirin O a....
A: To determine the amount of sodium citrate that gives the maximum solubility of one gram aspirin
Q: An unknown weak acid with a concentration of 0.091 M has a pH of 1.80. What is the Ka of the weak ac...
A: Weak Acid - These are the acids that dissociate partially in water. Given:- pH = 1.80 concentration...
Q: Distance Traveled Rf value Active Ingredient Analgesic Drug Distance Traveled by Spot (mm) by Solven...
A:
Q: 5. Reactivity to EAS CHO TEN BEN MEN DECREASING REACTIVITY TO EAS:
A:
Q: Which of the following is a correc mass balance equation for a solution that is 0.15 M in H3ASO4: (H...
A: Mass balance equation of 0.15 M triprotic acid H3AsO4
Q: What is the major product of the reaction between 1-chloro-2-methylcyclopentane andsodium hydroxide ...
A:
Q: What is the pH of a buffer made from 0.580 mol of CH₃NH₂ (Kb = 4.4 × 10⁻⁴) and 0.120 mol of CH₃NH₃I?
A: In this question, we want to determine the Calculate the pH of the buffer solution. You can see det...
Q: What is the change in the overall standard entropy (S°) for the following chemical reaction. 31 (aq)...
A:
Q: Calculate the overall AG°ƒfor the chemical reaction at 100°C in units of kJ/mole to 2 decimal places...
A:
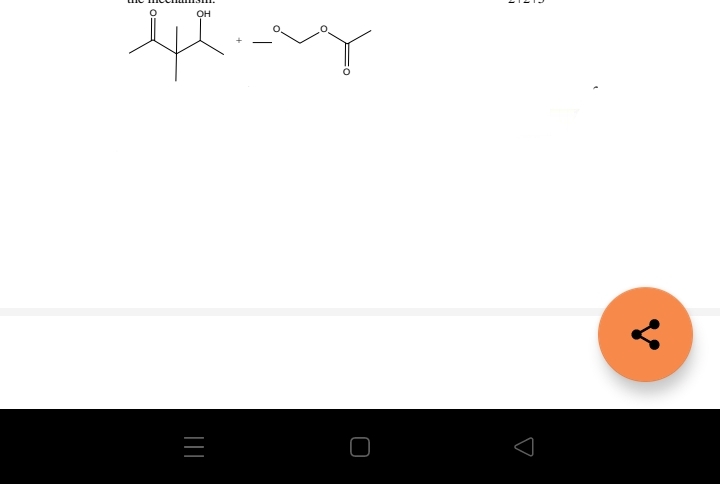
Q: Determine the mechanism if it is SN1, SN2, E1 or E2. Indicate all possible mechanisms for the given ...
A: 1- In this reaction, CH3CH2O- ion act as base and it will abstracted H+ ion from the most Steric bet...
Q: Which of the following would best indicate the internal kinetic energy of a substance? temperature ...
A: We know that , overall internal energy is the sum of kinetic energy and the potential energy . From ...
Q: The major product of the following * :reaction is CH, HC CH + HBr (D) CH, CH, (C) (B) CH3 HyC. H;C (...
A: This is the addition reaction between alkene and HBr. According to markonikows rule, H+ will attack...
Q: The following data are for the rearrangement of cyclopropane to propene at 500 °C. (CH2)3 CH3CH=CH...
A: We are given concentration(M) versus time(min) data for the rearrangement reaction of cyclopropane t...
Q: The least stable of the following carbocation intermediates is: * O II III IV
A: Stability of carbocation depends upon many factors in following order :- Aromaticity > Mesomeric ...
Q: 14 The alkane CH3CH2C(CH3)2CH2CH(CH3)2 has how many 1°, 2° and 3° carbon atoms? * 10:4, 20: 2, 3°: 2...
A: To find out the different degrees of carbon atom present in the given compound. We need to first dra...
Q: Which of the following statement is incorrect regarding buffer solutions: O Maximum buffer capacity ...
A: Buffer The buffer solution are prepared from conjugate acid / base pair . With addition of small a...
Q: Calculate the overall AH°ffor the following chemical reaction in kJ/mole to 2 decimal places. 13 (aq...
A: ∆Hof reaction = ∑n∆Hof products - ∑m∆Hof reactants
Q: The activity coefficient of Ni2+ ion (a Ni2+ =0.6 nm) in a solution that has an ionic strength of 0....
A:
Q: Which of the following compounds contains an ether functional group? CH3-CH2- C-CH3 CH3- C-H CH3 CH3...
A: Ether are a class of organic compounds that contain an ether group- an oxygen atom connected to alky...
Q: Cl2O7(g) + Ca(OH)2(aq)à Ca(ClO4)2(aq) + H2O(l) how do i balance this equation
A: The given chemical reaction is as follows: Cl2O7g + CaOH2aq → CaClO42aq + H2Ol
Q: Draw the form of L-tryptophan which is present at biological pH.
A: In this question, we will draw the structure of L-Tryptophan at biological pH You can see details S...
Q: The equilibrium constant is given for one of the reactions below. Determine the value of the missing...
A: Equilibrium constant is a relationship that shows how concentration of reactants vary with concentra...
Q: 20 of 48 For the equilibrium, H2(g)+Cl2(g)=2HCI(g), which stress will cause it to go in reverse? dec...
A: The given reaction represents a combination reaction. H2(g) + Cl2(g) <=> 2HCl(g) Here H2 and ...
Q: IV- C-C and C-H bond have different lengths * I and II I and III II, III, and IV I, II, and III
A:
Q: Define covalent bond
A: Chemical bond is a force of attraction which binds two atoms or ions together . Chemical bonds are g...
Q: In the following oxidation-reduction chemical reaction which chemical species is getting reduced? Hi...
A: In an oxidation-reduction reaction, the species whose oxidation number increases is oxidized and the...
Q: thermometer A sample of aluminum, which has a specific heat capacity of 0.897 J'g '.°c ', is put int...
A: Given the initial temperature of Al sample = 91.4 oC Initial temperature of water = 19.0 oC Final te...
Q: What is the relationship between strength of intermolecular forces and capillarity
A: When intermolecular forces are stronger, then the difference in energy increases between gas and liq...
Q: What is the IUPAC name of the following structures?
A: The question is based on the concept of IUPAC naming of the compound. we have to write correct names...
Q: Arrange the hybridizations of carbon based on the highly electronegative * sp > sp3 > sp2 sp > sp2 >...
A: Given Hybrid Orbitals sp3 sp2 sp Order based on Higher electronegative = To be determined
Q: Which of the following statements are correct? I-Halogenation of alkane is based on heterolytic clea...
A: Here some statements are given. We have to identify which statements are correct.
Q: Which of these gives mostly the meta product when treated with Br2 / * ?Fe a. b. HN d. O-CH,
A: The question is based on the concept of organic reactions. We have to identify the product formed ...
Q: Write the name for the enantiomer of (+)-(2R)-methyl-(1S)-cyclohexanol.
A:
Q: Which of the following conformational structures is 2,2-dimethylpentane? H. H CH H3C CH3 H3CH H CH3 ...
A:
Q: For the following complexes: see image Determine the oxidation states, coordination number, charge ...
A:
Q: Butane, CHo, is an easily liquefied gaseous fuel. Calculate the density of butane gas at 0.557 atm a...
A: Formula for the ideal gas density PM = ρRT Where, P = pressure in atm. M = molar mass in gram/ mol ρ...
Q: Identify the atomic groups with different functional groups within the same molecules.
A: A question based on IR spectroscopy, which is to be accomplished.
Q: Under the influence of common-ion effect which is a mass-action effect, the correct statement regard...
A: In presence of common ion effect , dissociation of weak electrolyte having that common ion , is supp...
Q: What is the major product of the reaction between 1-chloro-2- methylcyclopentane andsodium hydroxide...
A: The question is based on the concept of organic reactions. We have to identify the product formed ...
Q: Which one of the following statements is correct O The base is proton donor O Weak electrolyte does ...
A:
Q: 4. 4. purple How many elements are in sodium thiosulfate? yellow Na,s,O3 7. orange 5. 3 orange How m...
A: Given that, We have to identify the number of elements in the following compounds. Na3S2O3 C12H22O11
Q: When 2,2-dimethylbutane (1 mole) is allowed to react with Br2 (1 mole)in the presence of light or he...
A:
Q: The reactants A and B are mixed, and the reaction is timed until a colour change occurs. The data ar...
A:
Q: Identify the acetal and ketal group in the following disaccharide. Then decide whether the compound ...
A: The solution is given below -
Q: The molar solubility of the metal hydroxide (M(OH)3) in water is: M(OH)3 = M3+ + 3OH M3+ + 30H Ksp =...
A: The solubility reaction given is, => M(OH)3 (s) --------> M3+ (aq) + 3 OH- (aq) ...
Step by step
Solved in 2 steps with 2 images

- 15cComplete each of these reactions by filling in the blanks. Predict whether each reaction is product-favored or reactant-favored, and explain your reasoning. (a) _________ (aq) + Br(aq) NH3(aq) + HBr(aq) (b) CH3COOH(aq) + CN(aq) ________ (aq) + HCN(aq) (c) ________ (aq)+H2O () NH3(aq) + OH(aq)Q7 chapter 15 titration: multimedia activities Solve Asap
- Please complete the calculations related to pH, pOH, [H3O+], and [OH-]. Remember there are special rules related to significant digits when working with pH, pOH, [H3O+], and [OH-]. A sample of household vinegar was measured to have a hydronium ion concentration of 5.8 x 10-3mol/L. Determine the pH of the sample. Hand soap is effective at preventing bacterial contamination. Calculate the hydronium ion concentration in a brand of hand soap with a pH of 10.1Human saliva contains enzymes that start the process of digestion. Human saliva typically has a hydroxide ion concentration of 1.2 x 10-8 mol/L. Determine the pOH of saliva. Human blood is slightly basic. Calculate the hydroxide ion concentration in a sample of blood with a pOH of 6.55The concentration of hydronium ion in a sample is 3.5 x 10-7 mol/L. What is the pH of the above solution? The question should be answered to the proper number of significant digits.Help with question 7 c)

