One of the ways in which an L a-amino acid can be synthesized is to carry out an Sy2 reaction between an a-bromo acid and ammonia. (The wavy line indicates that the bond could be a dash or a wedge.) Draw the stereoisomer of the a-bromo acid that would be necessary Br. H3N. R An a-bromo acid An L a-amino acid to produce L-alanine, in which the R group is CH3.
One of the ways in which an L a-amino acid can be synthesized is to carry out an Sy2 reaction between an a-bromo acid and ammonia. (The wavy line indicates that the bond could be a dash or a wedge.) Draw the stereoisomer of the a-bromo acid that would be necessary Br. H3N. R An a-bromo acid An L a-amino acid to produce L-alanine, in which the R group is CH3.
Chapter26: Biomolecules: Amino Acids, Peptides, And Proteins
Section26.SE: Something Extra
Problem 28MP
Related questions
Question
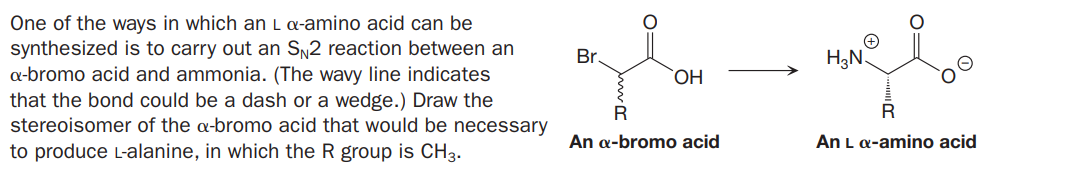
Transcribed Image Text:One of the ways in which an L a-amino acid can be
synthesized is to carry out an Sy2 reaction between an
a-bromo acid and ammonia. (The wavy line indicates
that the bond could be a dash or a wedge.) Draw the
stereoisomer of the a-bromo acid that would be necessary
Br.
H3N.
R
An a-bromo acid
An L a-amino acid
to produce L-alanine, in which the R group is CH3.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 4 images

Recommended textbooks for you

