Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are easily decomposed. Hexanitroethane, C2(NO2)6, decomposes according to the equation C2(NO2)6 2NO2+ 4NO + 2CO2 The reaction in CCI4 solvent is first order with respect to C2(NO2)6. At 79.0°C, k = 0.000230 s1 and at 111.9 °C, k = 0.0116 s1. (a) What is the half-life of C2(NO2)6 at 79.0 °C? i seconds eTextbook and Media (b) What is the half-life of C2(NO2)6 at 111.9 °C? i seconds 7:46 P 19-Apr-2
Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are easily decomposed. Hexanitroethane, C2(NO2)6, decomposes according to the equation C2(NO2)6 2NO2+ 4NO + 2CO2 The reaction in CCI4 solvent is first order with respect to C2(NO2)6. At 79.0°C, k = 0.000230 s1 and at 111.9 °C, k = 0.0116 s1. (a) What is the half-life of C2(NO2)6 at 79.0 °C? i seconds eTextbook and Media (b) What is the half-life of C2(NO2)6 at 111.9 °C? i seconds 7:46 P 19-Apr-2
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.55PAE: The following rate constants were obtained in an experiment in which the decomposition of gaseous...
Related questions
Question
26
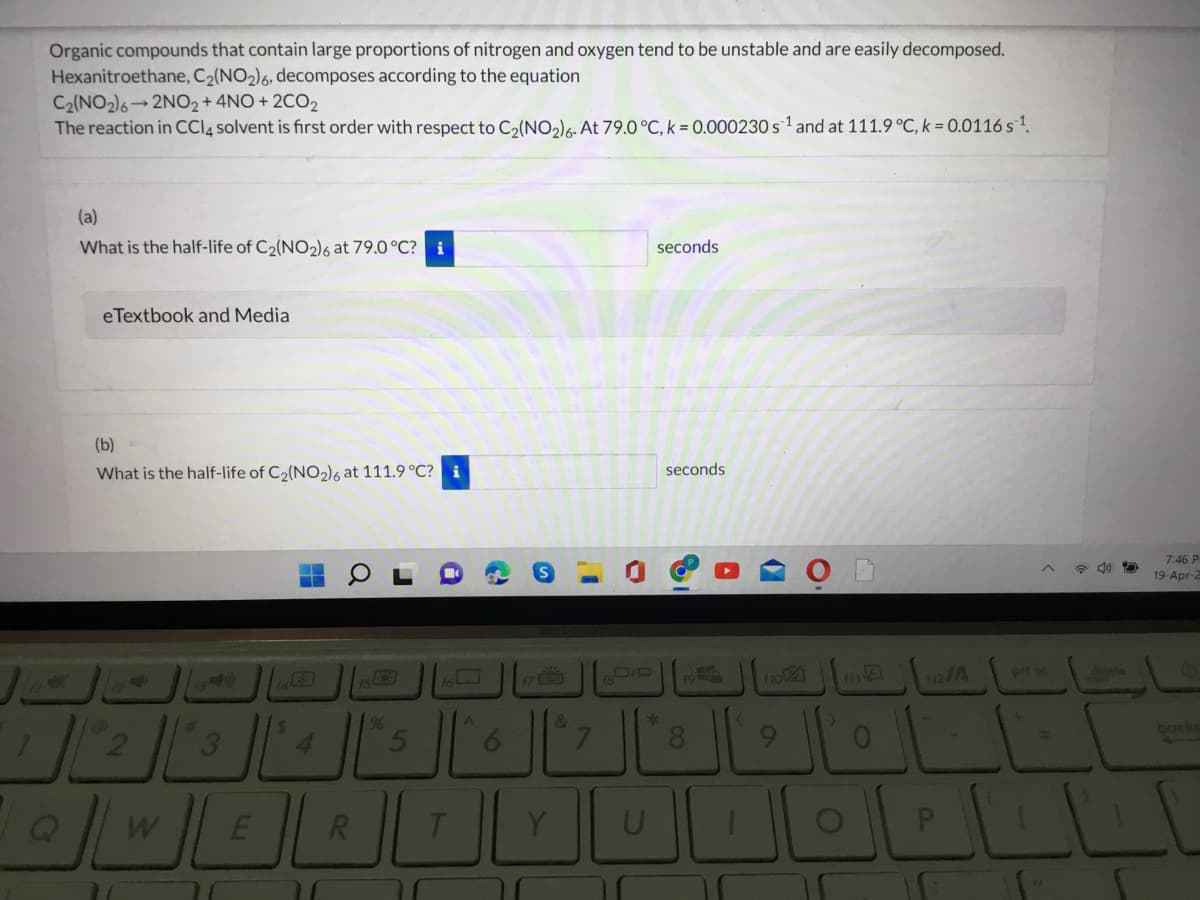
Transcribed Image Text:Organic compounds that contain large proportions of nitrogen and oxygen tend to be unstable and are easily decomposed.
Hexanitroethane, C2(NO2)6, decomposes according to the equation
C2(NO2)6 2NO2+ 4NO + 2CO2
The reaction in CCI4 solvent is first order with respect to C2(NO2)6. At 79.0°C, k = 0.000230 s1 and at 111.9 °C, k = 0.0116 s1.
(a)
What is the half-life of C2(NO2)o at 79.0 °C? i
seconds
eTextbook and Media
(b)
What is the half-life of C2(NO2)6 at 111.9 °C? i
seconds
7:46 P
19-Apr-2
17
19A
FO
12/A
prt sc
delete
18
LeLl
%23
3
7\
8.
9.
R.
T.
Y.
2\
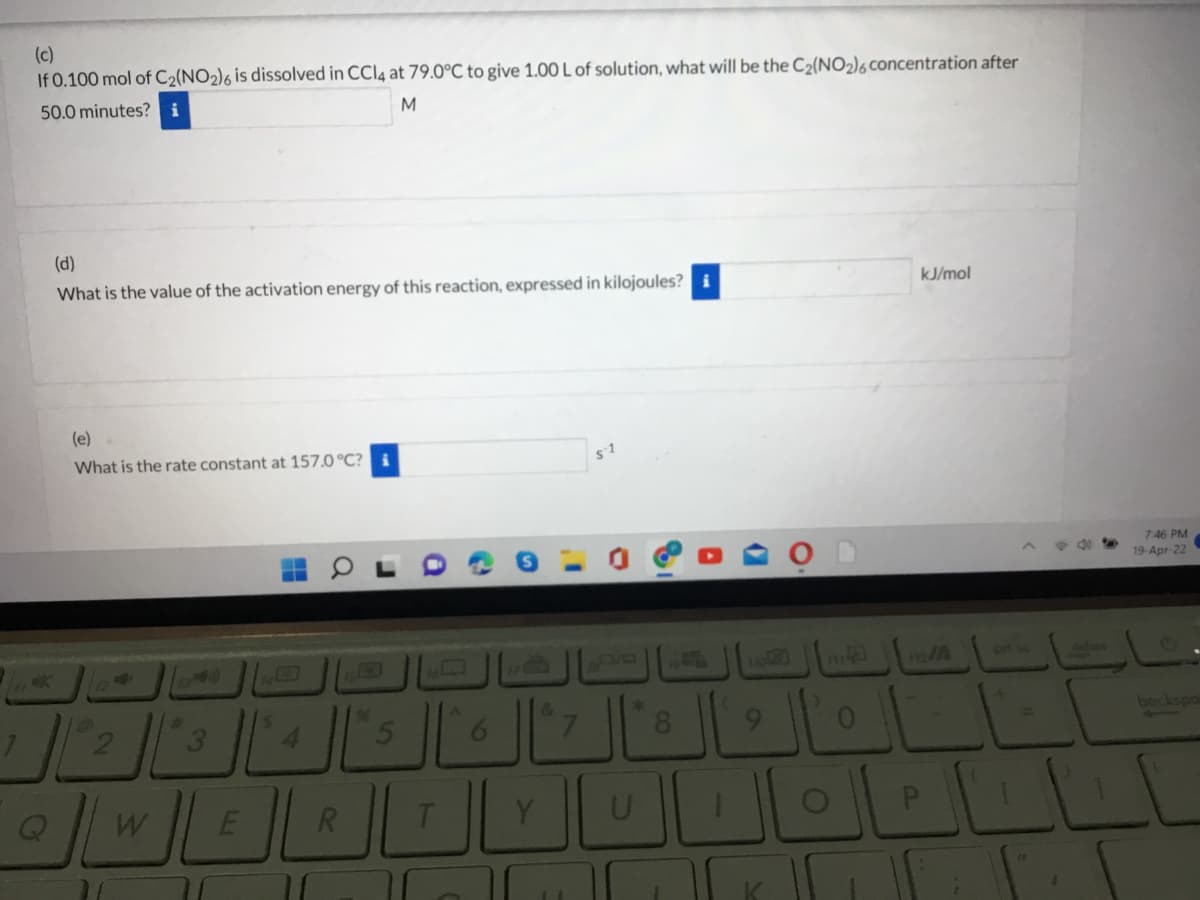
Transcribed Image Text:(c)
If 0.100 mol of C2(NO2)6 is dissolved in CCI4 at 79.0°C to give 1.00 L of solution, what will be the C2(NO2)6 concentration after
50.0 minutes? i
(d)
What is the value of the activation energy of this reaction, expressed in kilojoules?
kJ/mol
(e)
What is the rate constant at 157.0 °C? i
746 PM
19-Apr-22
ort s
delate
%23
96
bockspo
4.
8.
9.
W
E
R
LLI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning